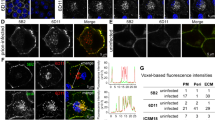
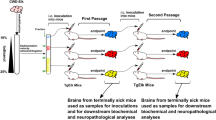
Abstract
The occurrence of multiple strains of prions may reflect conformational variability of PrPSc, a disease-associated, aggregated variant of the cellular prion protein, PrPC. Here we used luminescent conjugated polymers (LCPs), which emit conformation-dependent fluorescence spectra, for characterizing prion strains. LCP reactivity and emission spectra of brain sections discriminated among four immunohistochemically indistinguishable, serially mouse-passaged prion strains derived from sheep scrapie, chronic wasting disease (CWD), bovine spongiform encephalopathy (BSE), and mouse-adapted Rocky Mountain Laboratory scrapie prions. Furthermore, using LCPs we differentiated between field isolates of BSE and bovine amyloidotic spongiform encephalopathy, and identified noncongophilic deposits in prion-infected deer and sheep. We found that fibrils with distinct morphologies generated from chemically identical recombinant PrP yielded unique LCP spectra, suggesting that spectral characteristic differences resulted from distinct supramolecular PrP structures. LCPs may help to detect structural differences among discrete protein aggregates and to link protein conformational features with disease phenotypes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Prusiner, S.B. Novel proteinaceous infectious particles cause scrapie. Science 216, 136–144 (1982).
Fraser, H. & Dickinson, A.G. The sequential development of the brain lesion of scrapie in three strains of mice. J. Comp. Pathol. 78, 301–311 (1968).
Fraser, H. & Dickinson, A.G. Scrapie in mice. Agent-strain differences in the distribution and intensity of grey matter vacuolation. J. Comp. Pathol. 83, 29–40 (1973).
Bruce, M.E., McBride, P.A. & Farquhar, C.F. Precise targeting of the pathology of the sialoglycoprotein, PrP, and vacuolar degeneration in mouse scrapie. Neurosci. Lett. 102, 1–6 (1989).
Bessen, R.A. & Marsh, R.F. Biochemical and physical properties of the prion protein from two strains of the transmissible mink encephalopathy agent. J. Virol. 66, 2096–2101 (1992).
Polymenidou, M. et al. Coexistence of multiple PrPSc types in individuals with Creutzfeldt-Jakob disease. Lancet Neurol. 4, 805–814 (2005).
Hill, A.F. et al. Distinct glycoform ratios of protease resistant prion protein associated with PRNP point mutations. Brain 129, 676–685 (2006).
Safar, J. et al. Eight prion strains have PrP(Sc) molecules with different conformations. Nat. Med. 4, 1157–1165 (1998).
Peretz, D. et al. A change in the conformation of prions accompanies the emergence of a new prion strain. Neuron 34, 921–932 (2002).
Aguzzi, A. Understanding the diversity of prions. Nat. Cell Biol. 6, 290–292 (2004).
Weissmann, C. A 'unified theory' of prion propagation. Nature 352, 679–683 (1991).
Nilsson, K.P.R., Herland, A., Hammarstrom, P. & Inganas, O. Conjugated polyelectrolytes: conformation-sensitive optical probes for detection of amyloid fibril formation. Biochemistry 44, 3718–3724 (2005).
Nilsson, K.P.R. et al. Conjugated polyelectrolytes-conformation-sensitive optical probes for staining and characterization of amyloid deposits. ChemBioChem 7, 1096–1104 (2006).
Nilsson, K.P., Rydberg, J., Baltzer, L. & Inganas, O. Self-assembly of synthetic peptides control conformation and optical properties of a zwitterionic polythiophene derivative. Proc. Natl. Acad. Sci. USA 100, 10170–10174 (2003).
Nilsson, K.P.R., Rydberg, J., Baltzer, L. & Inganas, O. Twisting macromolecular chains: self-assembly of a chiral supermolecule from nonchiral polythiophene polyanions and random-coil synthetic peptides. Proc. Natl. Acad. Sci. USA 101, 11197–11202 (2004).
Fischer, M. et al. Prion protein (PrP) with amino-proximal deletions restoring susceptibility of PrP knockout mice to scrapie. EMBO J. 15, 1255–1264 (1996).
Sigurdson, C.J. et al. Strain fidelity of chronic wasting disease upon murine adaptation. J. Virol. 80, 12303–12311 (2006).
Bruce, M.E., McConnell, I., Fraser, H. & Dickinson, A.G. The disease characteristics of different strains of scrapie in Sinc congenic mouse lines: implications for the nature of the agent and host control of pathogenesis. J. Gen. Virol. 72, 595–603 (1991).
Ding, L., Jonforsen, M., Roman, L.S., Andersson, M.R. & Inganas, O. Photovoltaic cells with a conjugated polyelectrolyte. Synth. Met. 110, 133–140 (2000).
Ho, H.-A. et al. Colorimetric and fluorometric detection of nucleic acids using cationic polythiophene derivatives. Angew. Chem. Int. Edn. Engl. 41, 1548–1551 (2002).
Andersson, M.R. et al. Improved photoluminescence efficiency of films from conjugated polymers. Synth. Met. 85, 1383–1384 (1997).
Berggren, M. et al. Controlling inter-chain and intra-chain excitations of a poly(thiophene) derivative in thin films. Chem. Phys. Lett. 304, 84–90 (1999).
Nilsson, K.P.R., Andersson, M.R. & Inganas, O. Conformational transitions of a free amino-acid-functionalized polythiophene induced by different buffer systems. J. Phys. Condens. Matter 14, 10011–10020 (2002).
Petkova, A.T. et al. Self-propagating, molecular-level polymorphism in Alzheimer's beta-amyloid fibrils. Science 307, 262–265 (2005).
Jimenez, J.L. et al. The protofilament structure of insulin amyloid fibrils. Proc. Natl. Acad. Sci. USA 99, 9196–9201 (2002).
Apetri, A.C., Vanik, D.L. & Surewicz, W.K. Polymorphism at residue 129 modulates the conformational conversion of the D178N variant of human prion protein 90–231. Biochemistry 44, 15880–15888 (2005).
Bocharova, O.V., Breydo, L., Parfenov, A.S., Salnikov, V.V. & Baskakov, I.V. In vitro conversion of full-length mammalian prion protein produces amyloid form with physical properties of PrP(Sc). J. Mol. Biol. 346, 645–659 (2005).
Casalone, C. et al. Identification of a second bovine amyloidotic spongiform encephalopathy: molecular similarities with sporadic Creutzfeldt-Jakob disease. Proc. Natl. Acad. Sci. USA 101, 3065–3070 (2004).
Asante, E.A. et al. BSE prions propagate as either variant CJD-like or sporadic CJD-like prion strains in transgenic mice expressing human prion protein. EMBO J. 21, 6358–6366 (2002).
Meyer-Luehmann, M. et al. Exogenous induction of cerebral beta-amyloidogenesis is governed by agent and host. Science 313, 1781–1784 (2006).
Acknowledgements
This study was supported by grants from the European Union (Tseur to A.A. and Understanding Protein Misfolding and Aggregation by NMR (UPMAN) to K.W.), the Swiss National Science Foundation, the Swiss National Competence Centers for Research on Neural Plasticity and Repair (A.A.) and on Structural Biology (K.W.), US National Institutes of Health K08-AI01802 (C.J.S.), the Foundation for Research at the University of Zürich (C.J.S.), the US National Prion Research Program (C.J.S. and A.A.), the Knut and Alice Wallenberg foundation (K.P.R.N.), the Swedish Foundation for Strategic Research (P.H.), the Wenner-Gren Foundations and the Swedish Research Council (P.H.), and the Natural Sciences and Engineering Research Council of Canada (M.L.). We thank P. Vilkman, S. Fransson and C. von Schroetter for providing technical support, P. Tittmann (Electron Microscopy Center, ETH Zurich) for providing electron microscopy services, the staff at the confocal microscopy centers at the ETH Zurich and at the University of Zurich for help with spectral collection, B. Seifert for help with statistical analysis, and C. Casalone (Istituto Zooprofilattico Sperimentale del Piemonte), F. Ehrensperger (University of Zürich), M. Miller (Colorado Division of Wildlife) and K. O'Rourke (United States Department of Agriculture) for BASE, BSE, CWD and sheep scrapie brain samples, respectively.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
K.P.R.N. is part owner of a company commercializing the LCPs and the methods described in this article.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–4, Supplementary Tables 1–4, Supplementary Methods (PDF 746 kb)
Rights and permissions
About this article
Cite this article
Sigurdson, C., Nilsson, K., Hornemann, S. et al. Prion strain discrimination using luminescent conjugated polymers. Nat Methods 4, 1023–1030 (2007). https://doi.org/10.1038/nmeth1131
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmeth1131
This article is cited by
-
Therapeutic development of polymers for prion disease
Cell and Tissue Research (2023)
-
Prion-like strain effects in tauopathies
Cell and Tissue Research (2023)
-
Dual-probe fluorescence spectroscopy for sensitive quantitation of Alzheimer’s amyloid pathology
Acta Neuropathologica Communications (2022)
-
Two distinct conformers of PrPD type 1 of sporadic Creutzfeldt–Jakob disease with codon 129VV genotype faithfully propagate in vivo
Acta Neuropathologica Communications (2021)
-
Helical polymers for biological and medical applications
Nature Reviews Chemistry (2020)