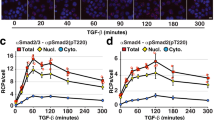
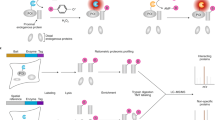
Abstract
Cellular processes can only be understood as the dynamic interplay of molecules. There is a need for techniques to monitor interactions of endogenous proteins directly in individual cells and tissues to reveal the cellular and molecular architecture and its responses to perturbations. Here we report our adaptation of the recently developed proximity ligation method to examine the subcellular localization of protein-protein interactions at single-molecule resolution. Proximity probes—oligonucleotides attached to antibodies against the two target proteins—guided the formation of circular DNA strands when bound in close proximity. The DNA circles in turn served as templates for localized rolling-circle amplification (RCA), allowing individual interacting pairs of protein molecules to be visualized and counted in human cell lines and clinical specimens. We used this method to show specific regulation of protein-protein interactions between endogenous Myc and Max oncogenic transcription factors in response to interferon-γ (IFN-γ) signaling and low-molecular-weight inhibitors.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Fredriksson, S. et al. Protein detection using proximity-dependent DNA ligation assays. Nat. Biotechnol. 20, 473–477 (2002).
Gullberg, M. et al. Cytokine detection by antibody-based proximity ligation. Proc. Natl. Acad. Sci. USA 101, 8420–8424 (2004).
Baner, J., Nilsson, M., Mendel-Hartvig, M. & Landegren, U. Signal amplification of padlock probes by rolling circle replication. Nucleic Acids Res. 26, 5073–5078 (1998).
Grandori, C., Cowley, S.M., James, L.P. & Eisenman, R.N. The Myc/Max/Mad network and the transcriptional control of cell behavior. Annu. Rev. Cell Dev. Biol. 16, 653–699 (2000).
Oster, S.K., Ho, C.S., Soucie, E.L. & Penn, L.Z. The myc oncogene: MarvelouslY Complex. Adv. Cancer Res. 84, 81–154 (2002).
Patel, J.H., Loboda, A.P., Showe, M.K., Showe, L.C. & McMahon, S.B. Analysis of genomic targets reveals complex functions of MYC. Nat. Rev. Cancer 4, 562–568 (2004).
Pelengaris, S., Khan, M. & Evan, G. c-MYC: more than just a matter of life and death. Nat. Rev. Cancer 2, 764–776 (2002).
Hu, C.D., Chinenov, Y. & Kerppola, T.K. Visualization of interactions among bZIP and Rel family proteins in living cells using bimolecular fluorescence complementation. Mol. Cell 9, 789–798 (2002).
Venetsanakos, E. et al. Induction of tubulogenesis in telomerase-immortalized human microvascular endothelial cells by glioblastoma cells. Exp. Cell Res. 273, 21–33 (2002).
Grinberg, A.V., Hu, C.D. & Kerppola, T.K. Visualization of Myc/Max/Mad family dimers and the competition for dimerization in living cells. Mol. Cell. Biol. 24, 4294–4308 (2004).
Winqvist, R., Saksela, K. & Alitalo, K. The myc proteins are not associated with chromatin in mitotic cells. EMBO J. 3, 2947–2950 (1984).
Mateyak, M.K., Obaya, A.J., Adachi, S. & Sedivy, J.M. Phenotypes of c-Myc-deficient rat fibroblasts isolated by targeted homologous recombination. Cell Growth Differ. 8, 1039–1048 (1997).
Alroy, I. & Yarden, Y. The ErbB signaling network in embryogenesis and oncogenesis: signal diversification through combinatorial ligand-receptor interactions. FEBS Lett. 410, 83–86 (1997).
Bouchard, C., Marquardt, J., Bras, A., Medema, R.H. & Eilers, M. Myc-induced proliferation and transformation require Akt-mediated phosphorylation of FoxO proteins. EMBO J. 23, 2830–2840 (2004).
Adhikary, S. & Eilers, M. Transcriptional regulation and transformation by Myc proteins. Nat. Rev. Mol. Cell Biol. 6, 635–645 (2005).
Öberg, F., Larsson, L.G., Anton, R. & Nilsson, K. Interferon gamma abrogates the differentiation block in v-myc-expressing U-937 monoblasts. Proc. Natl. Acad. Sci. USA 88, 5567–5571 (1991).
Guzhova, I. et al. Interferon-gamma cooperates with retinoic acid and phorbol ester to induce differentiation and growth inhibition of human neuroblastoma cells. Int. J. Cancer 94, 97–108 (2001).
Bahram, F., Wu, S., Öberg, F., Lüscher, B. & Larsson, L.G. Posttranslational regulation of Myc function in response to phorbol ester/interferon-gamma-induced differentiation of v-Myc-transformed U-937 monoblasts. Blood 93, 3900–3912 (1999).
Eilers, M., Picard, D., Yamamoto, K.R. & Bishop, J.M. Chimaeras of myc oncoprotein and steroid receptors cause hormone-dependent transformation of cells. Nature 340, 66–68 (1989).
Littlewood, T.D., Hancock, D.C., Danielian, P.S., Parker, M.G. & Evan, G.I. A modified oestrogen receptor ligand-binding domain as an improved switch for the regulation of heterologous proteins. Nucleic Acids Res. 23, 1686–1690 (1995).
Solomon, D.L., Philipp, A., Land, H. & Eilers, M. Expression of cyclin D1 mRNA is not upregulated by Myc in rat fibroblasts. Oncogene 11, 1893–1897 (1995).
Yin, X., Giap, C., Lazo, J.S. & Prochownik, E.V. Low molecular weight inhibitors of Myc-Max interaction and function. Oncogene 22, 6151–6159 (2003).
Arabi, A. et al. c-Myc associates with ribosomal DNA in the nucleolus and activates RNA polymerase I transcription. Nat. Cell Biol. 7, 303–310 (2005).
Beckstead, J.H. A simple technique for preservation of fixation-sensitive antigens in paraffin-embedded tissues. J. Histochem. Cytochem. 42, 1127–1134 (1994).
Acknowledgements
We thank A.-C. Andersson for technical assistance; C. Wählby for image analysis; M. Taussig and S. Fredriksson for valuable comments on the manuscript; J.M. Sedivy and M. Eilers for providing cells; A.-C. Steffen and J. Carlsson for providing EGFR and Her-2 antibodies; and G.R. Adolf for providing IFN-γ. We obtained frozen human tissues from the Fresh Tissue Biobank at the Department of Clinical Pathology, Uppsala University Hospital (supported by the SWEGENE/Wallenberg Consortium North Biobank Program). This project was supported by the Wallenberg Foundation, the EU Integrated Project MolTools, the Research Councils of Sweden for natural science and for medicine (to U.L.); and the Swedish Cancer Foundation, the Swedish Children Cancer Foundation and the Human Frontiers Science Program (to L.-G.L.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
U.L., M.G. and J.J. are cofounders of the company Olink AB, which exploits this technology and holds the right to patents US6558928, US6878515, US7074564, US09/785657, US 10/496385, US 11/011438, and corresponding patents and patent applications in other geographies describing the technology. The inventions described in the patents have been jointly or separately invented by U.L. and Simon Fredriksson.
Supplementary information
Rights and permissions
About this article
Cite this article
Söderberg, O., Gullberg, M., Jarvius, M. et al. Direct observation of individual endogenous protein complexes in situ by proximity ligation. Nat Methods 3, 995–1000 (2006). https://doi.org/10.1038/nmeth947
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmeth947
This article is cited by
-
TRIM28-mediated nucleocapsid protein SUMOylation enhances SARS-CoV-2 virulence
Nature Communications (2024)
-
Refining the lens of DNA microscopy
Nature Computational Science (2024)
-
The phosphatase DUSP22 inhibits UBR2-mediated K63-ubiquitination and activation of Lck downstream of TCR signalling
Nature Communications (2024)
-
The CRISPR-Cas13a Gemini System for noncontiguous target RNA activation
Nature Communications (2024)
-
Soluble and multivalent Jag1 DNA origami nanopatterns activate Notch without pulling force
Nature Communications (2024)