Abstract
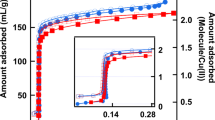
Nanoporous materials find widespread applications in our society: from drug delivery to environmentally friendly catalysis and separation technologies1,2,3,4,5,6. The efficient design of these processes depends crucially on understanding the mass transfer mechanism. This is conventionally determined by uptake or release experiments, carried out with assemblages of nanoporous crystals, assuming all crystals to be identical. Using micro-imaging techniques7, we now show that even apparently identical crystals (that is, crystals of similar size and shape) from the same batch may exhibit very different uptake rates. The relative contribution of the surface resistance to the overall transport resistance varied with both the crystal and the guest molecule. As a consequence of this crystal diversity, the conventional approach may not distinguish correctly between the different mass transfer mechanisms. Detection of this diversity adds an important new piece of evidence in the search for the origin of the surface barrier phenomenon. Our investigations were carried out with the zeolite SAPO-34, a key material in the methanol-to-olefins (MTO) process8, propane–propene separation9,10 and adsorptive heat transformation11.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Horcajada, P. et al. Porous metal-organic frameworks nanoscale carriers as a potential platform from drug delivery and imaging. Nature Mater. 9, 172–178 (2009).
Ruthven, D. M., Farooq, S. & Knaebel, K. S. Pressure Swing Adsorption (VCH, 1994).
Kärger, J., Ruthven, D. M. & Theodorou, D. N. Diffusion in Nanoporous Materials (Wiley-VCH, 2012).
Weitkamp, J. & Puppe, L. Catalysis and Zeolites (Springer, 1999).
Cejka, J., Corma, A. & Zones, S Zeolites and Catalysis: Synthesis, Reactions and Applications (Wiley-VCH, 2010).
Humplik, T. et al. Nanostructured materials for water desalination. Nanotechnology 22, 292001 (2011).
Kärger, J. et al. Microimaging of transient guest profiles to monitor mass transfer in nanoporous materials. Nature Mater. 13, 333–343 (2014).
Stöcker, M. Methanol-to-hydrocarbons: catalytic materials and their behavior. Micropor. Mesopor. Mater. 29, 3–48 (1999).
Ruthven, D. M. & Reyes, S. C. Adsorptive separation of light olefins from paraffins. Micropor. Mesopor. Mater. 104, 59–66 (2007).
Hedin, N., DeMartin, G. J., Roth, W. J., Strohmaier, K. G. & Reyes, S. C. PFG NMR self-diffusion of small hydrocarbons in high silica DDR, CHA and LTA structures. Micropor. Mesopor. Mater. 109, 327–334 (2008).
Henninger, S. K., Jeremias, F., Kummer, H., Schossig, P. & Henning, H.-M. Novel sorption materials for solar heating and cooling. Energy Procedia 30, 279–288 (2012).
Yasuda, Y. Frequency response method for investigation of gas/surface dynamic phenomena. Heterogen. Chem. Rev. 1, 103–124 (1994).
Eic, M. & Ruthven, D. M. A new experimental technique for measurement of intracrystalline diffusivity. Zeolites 8, 40–45 (1988).
Zhang, L. et al. Direct assessment of molecular transport in mordenite: dominance of surface resistances. Chem. Commun. (2009)fhtsjw.
Gobin, O. C., Reitmeier, S. J., Jentys, A. & Lercher, J. A. Role of the surface modification on the transport of hexane isomers in ZSM-5. J. Phys. Chem. C 115, 1171–1179 (2011).
Higgins, J. B. et al. The framework topology of zeolite beta. Zeolites 8, 446–452 (1988).
Robson, H. & Lillerud, K. P. Verified Syntheses of Zeolitic Materials (Elsevier, 2001).
Karwacki, L., Stavitski, E., Kox, M. H. F., Kornatowski, J. & Weckhuysen, B. M. Intergrowth structure of zeolite crystals as determined by optical and fluorescence microscopy of the template-removal process. Angew. Chem. Int. Ed. 46, 7228–7231 (2007).
Gueudré, L., Jolimaître, E., Bats, N. & Dong, W. Diffusion in zeolites: is surface resistance a critical parameter? Adsorption 16, 17–27 (2010).
Mitchell, S., Michels, N.-L., Kunze, K. & Pérez-Ramírez, J. Visualization of hierarchically structured zeolite bodies from macro to nano length scales. Nature Chem. 4, 825–831 (2012).
Buurmans, I. L. C. & Weckhuysen, B. M. Heterogeneities of individual catalyst particles in space and time as monitored by spectroscopy. Nature Chem. 4, 873–886 (2012).
Crank, J. The Mathematics of Diffusion (Clarendon, 1975).
Kärger, J., Pfeifer, H., Rauscher, M. & Walter, A. Self-diffusion of n-Paraffins in NaX Zeolite. J. Chem. Soc. Faraday Trans. I 76, 717–737 (1980).
Wragg, D. S., Johnson, R. E., Norby, P. & Fjellvag, H. The adsorption of methanol and water on SAPO-34: in situ and ex situ X-ray diffraction studies. Micropor. Mesopor. Mater. 134, 210–215 (2010).
Briend, M., Vomscheid, R., Peltre, M. J., Man, P. P. & Barthomeuf, D. Infuence of the choice of the template on the short- and long-term stability of SAPO-34 zeolite. J. Phys. Chem. 99, 8270–8276 (1995).
Heinke, L., Gu, Z. & Wöll, C. The surface barrier phenomenon at the loading of metal-organic frameworks. Nature Commun. 5, 4562 (2014).
Fjermestad, T., Svelle, S. & Swang, O. Mechanism of Si island formation in SAPO-34. J. Phys. Chem. C 119, 2086–2095 (2015).
Mores, D. et al. Space- and time-resolved in-situ spectroscopy on the coke formation in molecular sieves: methanol-to-olefin conversion over H-ZSM-5 and H-SAPO-34. Chem. Eur. J. 14, 11320–11327 (2008).
Qian, Q. et al. Single-particle spectroscopy on large SAPO-34 crystals at work: methanol-to-olefin versus ethanol-to-olefin processes. Chem. Eur. J. 19, 11204–11215 (2013).
Qian, Q. et al. Single-particle spectroscopy of alcohol-to-olefins over SAPO-34 at different reaction stages: crystal accessibility and hydrocarbons reactivity. ChemCatChem 6, 772–783 (2014).
Remy, T. et al. Adsorption and separation of C1-C8 alcohols on SAPO-34. J. Phys. Chem. C 115, 8117–8125 (2011).
Chmelik, C. & Kärger, J. In situ study on molecular diffusion phenomena in nanoporous catalytic solids. Chem. Soc. Rev. 39, 4864–4884 (2010).
Heinke, L., Kortunov, P., Tzoulaki, D. & Kärger, J. The options of interference microscopy to explore the significance of intracrystalline diffusion and surface permeation for overall mass transfer on nanoporous materials. Adsorption 13, 215–223 (2007).
Chmelik, C. et al. Mass transfer in a nanoscale material enhanced by an opposing flux. Phys. Rev. Lett. 104, 085902 (2010).
Heinke, L. Significance of concentration-dependent intracrystalline diffusion and surface permeation for overall mass transfer. Diffus. Fundam. 4, 1–11 (2007).
Karge, H. G. & Kärger, J. in Adsorption and Diffusion (eds H. G., Karge & Weitkamp, J.) 135–206 (Springer, 2008).
Barrer, R. M. Zeolites and Clay Minerals as Sorbents and Molecular Sieves (Academic, 1978).
Garg, D. R. & Ruthven, D. M. The effect of the concentration dependence of diffusivity on zeolitic sorption curves. Chem. Eng. Sci. 27, 417–423 (1972).
Titze, T. et al. Microimaging of transient concentration profiles of reactant and product molecules during catalytic conversion in nanoporous materials. Angew. Chem. Int. Ed. 54, 5060–5064 (2015).
Acknowledgements
J.C.S.R. is grateful to the Agency for Innovation by Science and Technology in Flanders (IWT) and the Research Foundation—Flanders (FWO—Vlaanderen) for their financial support. Graphical content was supported by Visuality (BE). Deutsche Forschungsgemeinschaft and Fonds der Chemischen Industrie are acknowledged for establishing the micro-imaging system in Leipzig. Valuable discussions with D. M. Ruthven are particularly appreciated.
Author information
Authors and Affiliations
Contributions
J.C.S.R., C.C., J.F.M.D. and J.K. contributed to the development of the experimental approach. J.C.S.R. and A.L. conducted the interference microscopy experiments; C.C. and A.L. performed the infrared microscopy measurements; J.C.S.R., C.C. and J.K. contributed to the initial ideas and interpreting experimental data; J.C.S.R., G.V.B. and J.F.M.D. supported the material characterization. J.C.S.R. provided the batch uptake simulations. I.V. and H.T. ensured the performance of the FE-SEM, AFM, XPS and EDX measurements, which were realized by J.C.S.R. All authors participated in the writing of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 2475 kb)
Rights and permissions
About this article
Cite this article
Remi, J., Lauerer, A., Chmelik, C. et al. The role of crystal diversity in understanding mass transfer in nanoporous materials. Nature Mater 15, 401–406 (2016). https://doi.org/10.1038/nmat4510
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmat4510
This article is cited by
-
Elemental zoning enhances mass transport in zeolite catalysts for methanol to hydrocarbons
Nature Catalysis (2023)
-
SiO2 Deposition to Regulate Surface Barriers and Its Impact on ZSM-5 Catalyzed Reactions with Distinct Molecular Sizes
Catalysis Letters (2023)
-
Diffusion and reaction in pore hierarchies by the two-region model
Adsorption (2021)
-
Diffusion in nanopores: inspecting the grounds
Adsorption (2021)
-
Remarks on adsorbent surface barrier to adsorbate mass transport
Adsorption (2021)