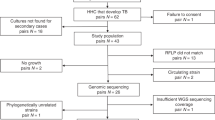
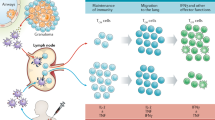
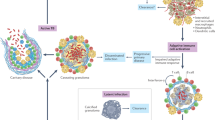
Abstract
Mycobacterium tuberculosis is a globally successful pathogen due to its ability to persist for long periods of time unrecognized by the human immune system. The panoply of genes that allows the organism to enter latency and then re-emerge later during endogenous reinfection are now being elucidated. Novel antimicrobials and vaccines will need to target these mycobacterial pathogenic mechanisms to suceed against tuberculosis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Steve Horwitz
Similar content being viewed by others
References
Medlar, E.M., Bernstein, S. & Steward, D.M. A bacteriologic study of resected tuberculous lesions. Am. Rev. Tuberc. 66, 36–43 (1952).
Hobby, G.L. et al. The late emergence of M. tuberculosis in liquid cultures of pulmonary lesions resected from humans. Am. Rev. Tuberc. 70, 191–218 (1954).
Opie, E.L. & Aronson, J.D. Tubercle bacilli in latent tuberculous lesions and in lung tissue without tuberculous lesions. Arch. Pathol. Lab. Med. 4, 1–21 (1927).
Comstock, G.W., Baum, C. & Snider, D.E., Jr. Isoniazid prophylaxis among Alaskan Eskimos: a final report of the bethel isoniazid studies. Am. Rev. Respir. Dis. 119, 827–830 (1972).
Cole, S.T. et al. Deciphering the biology of Mycobacterium tuberculosis from the complete genome sequence. Nature 393, 537–544 (1998); erratum: 396, 190 (1998).
DeMaio, J. et al. A stationary-phase stress-response σ factor from M. tuberculosis. Proc. Natl. Acad. Sci. USA. 93, 2790–2794 (1996).
Chen, P. et al. Construction and characterization of a mycobacterium tuberculosis mutant lacking the alternate sigma factor gene, sigF. Infect. Immun. 68, 5575–80 (2000).
Chen, P., Gomez, J. & Bishai, W.R. in Mycobacterial Transcription Regulation in Stationary Phase, in Molecular Genetics of Mycobacteria (eds. Hatfull, G.H & Jacobs, W.R.) 149–156 (ASM Press, Washington DC, 2000).
McCune, R.M. et al. Microbial Persistence II. Characteristics of the sterile state of tubercle bacilli. J. Exp. Med. 123, 469–486 (1966).
McCune, R.M. et al. Microbial Persistence I. The capacity of tubercle bacilli to survive sterilization in mouse tissues. J. Exp. Med. 123, 445–468 (1966).
Scanga, C.A. et al. Reactivation of latent tuberculosis: variations on the Cornell murine model. Infect. Immun. 67, 4531–4538 (1999).
Miyazaki, E., Chaisson, R.E. & Bishai, W.R. Analysis of rifapentine for preventive therapy in the Cornell mouse model of latent tuberculosis. Antimicrob. Agents Chemother. 43, 2126–2130 (1999).
Wayne, L.G. Synchronized replication of Mycobacterium tuberculosis. Infect. Immun. 17, 528–530 (1977).
Wayne, L.G. & Lin, K.Y. Glyoxylate metabolism and adaptation of M. tuberculosis to survival under anaerobic conditions. Infect. Immun. 37, 1042–1049 (1982).
Wayne, L.G. & Hayes, L.G. An in vitro model for sequential study of shiftdown of M. tuberculosis through two stages of nonreplicating persistence. Infect. Immun. 64, 2062–2069 (1996).
Wayne, L.G. Dormancy of Mycobacterium tuberculosis and latency of disease. Eur. J. Clin. Microbiol. Infect. Dis. 13, 908–914 (1994).
McKinney, J.D. et al. Persistence of M. tuberculosis in macrophages and mice requires the glyoxylate shunt enzyme isocitrate lyase. Nature 406, 735–738 (2000).
Glickman, M.S., Cox, J.S. & Jacobs, W.R., Jr. A novel mycolic acid cyclopropane synthetase is required for coding, persistence, and virulence of M. tuberculosis. Mol. Cell 5, 717–727 (2000).
Ramakrishnan, L., Federspiel, N.A. & Falkow, S. Granuloma-specific expression of mycobacterium virulence proteins from the glycine-rich PE-PGRS family. Science 288, 1436–1439 (2000).
Wayne, L.G. & Sramek, H.A. Metronidazole is bactericidal to dormant cells of Mycobacterium tuberculosis. Antimicrob. Agents Chemother. 38, 2054–2058 (1994).
Dhillon, J. et al. Metronidazole has no antibacterial effect in Cornell model murine tuberculosis. Int. J. Tuberc. Lung Dis. 2, 736–742 (1998).
Stead, W.W. Pathogenesis of a first episode of chronic pulmonary tuberculosis in man: recrudescence of residuals of the primary infection or exogenous reinfection? Am. Rev. Respir. Dis. 95, 729–745 (1967).
Nardell, E. et al. Exogenous reinfection with tuberculosis in a shelter for the homeless. N. Engl. J. Med. 315, 1570–1575 (1996).
Small, P.M. et al. Exogenous reinfection with multidrug-resistant M. tuberculosis in patients with advanced HIV infection. N. Engl. J. Med. 328, 1137–1144 (1993).
van Rie, A. et al. Exogenous reinfection as a cause of recurrent tuberculosis after curative treatment. N. Engl. J. Med. 341, 1174–9 (1999).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Manabe, Y., Bishai, W. Latent Mycobacterium tuberculosis–persistence, patience, and winning by waiting. Nat Med 6, 1327–1329 (2000). https://doi.org/10.1038/82139
Issue Date:
DOI: https://doi.org/10.1038/82139
This article is cited by
-
Immunopathogenesis in HIV-associated pediatric tuberculosis
Pediatric Research (2022)
-
Design of 2-Nitroimidazooxazine Derivatives as Deazaflavin-Dependent Nitroreductase (Ddn) Activators as Anti-Mycobacterial Agents Based on 3D QSAR, HQSAR, and Docking Study with In Silico Prediction of Activity and Toxicity
Interdisciplinary Sciences: Computational Life Sciences (2019)
-
IL-17 and IL-22 production in HIV+ individuals with latent and active tuberculosis
BMC Infectious Diseases (2018)
-
High-density lipoprotein suppresses tumor necrosis factor alpha production by mycobacteria-infected human macrophages
Scientific Reports (2018)
-
Mycobacterium tuberculosis
Bundesgesundheitsblatt - Gesundheitsforschung - Gesundheitsschutz (2018)