Abstract
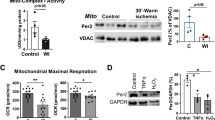
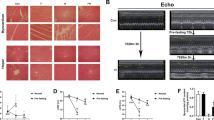
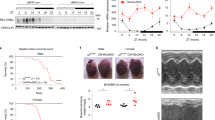
Adenosine signaling has been implicated in cardiac adaptation to limited oxygen availability. In a wide search for adenosine receptor A2b (Adora2b)-elicited cardioadaptive responses, we identified the circadian rhythm protein period 2 (Per2) as an Adora2b target. Adora2b signaling led to Per2 stabilization during myocardial ischemia, and in this setting, Per2−/− mice had larger infarct sizes compared to wild-type mice and loss of the cardioprotection conferred by ischemic preconditioning. Metabolic studies uncovered a limited ability of ischemic hearts in Per2−/− mice to use carbohydrates for oxygen-efficient glycolysis. This impairment was caused by a failure to stabilize hypoxia-inducible factor-1α (Hif-1α). Moreover, stabilization of Per2 in the heart by exposing mice to intense light resulted in the transcriptional induction of glycolytic enzymes and Per2-dependent cardioprotection from ischemia. Together, these studies identify adenosine-elicited stabilization of Per2 in the control of HIF-dependent cardiac metabolism and ischemia tolerance and implicate Per2 stabilization as a potential new strategy for treating myocardial ischemia.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Eltzschig, H.K. & Carmeliet, P. Hypoxia and inflammation. N. Engl. J. Med. 364, 656–665 (2011).
Sitkovsky, M.V. et al. Physiological control of immune response and inflammatory tissue damage by hypoxia-inducible factors and adenosine A2A receptors. Annu. Rev. Immunol. 22, 657–682 (2004).
Eltzschig, H.K. & Eckle, T. Ischemia and reperfusion—from mechanism to translation. Nat. Med. 17, 1391–1401 (2011).
Neubauer, S. The failing heart—an engine out of fuel. N. Engl. J. Med. 356, 1140–1151 (2007).
Aragonés, J., Fraisl, P., Baes, M. & Carmeliet, P. Oxygen sensors at the crossroad of metabolism. Cell Metab. 9, 11–22 (2009).
Thompson, L.F. et al. Crucial role for ecto-5′-nucleotidase (CD73) in vascular leakage during hypoxia. J. Exp. Med. 200, 1395–1405 (2004).
Fredholm, B.B. Adenosine, an endogenous distress signal, modulates tissue damage and repair. Cell Death Differ. 14, 1315–1323 (2007).
Eckle, T. et al. A2B adenosine receptor dampens hypoxia-induced vascular leak. Blood 111, 2024–2035 (2008).
Eckle, T., Kohler, D., Lehmann, R., El Kasmi, K. & Eltzschig, H.K. Hypoxia-inducible factor-1 is central to cardioprotection: a new paradigm for ischemic preconditioning. Circulation 118, 166–175 (2008).
Eckle, T. et al. Cardioprotection by ecto-5′-nucleotidase (CD73) and A2B adenosine receptors. Circulation 115, 1581–1590 (2007).
Zheng, B. et al. The mPer2 gene encodes a functional component of the mammalian circadian clock. Nature 400, 169–173 (1999).
Lee, C., Etchegaray, J.P., Cagampang, F.R., Loudon, A.S. & Reppert, S.M. Posttranslational mechanisms regulate the mammalian circadian clock. Cell 107, 855–867 (2001).
Gallego, M. & Virshup, D.M. Post-translational modifications regulate the ticking of the circadian clock. Nat. Rev. Mol. Cell Biol. 8, 139–148 (2007).
Wu, J.T., Lin, H.C., Hu, Y.C. & Chien, C.T. Neddylation and deneddylation regulate Cul1 and Cul3 protein accumulation. Nat. Cell Biol. 7, 1014–1020 (2005).
Khoury, J., Ibla, J.C., Neish, A.S. & Colgan, S.P. Antiinflammatory adaptation to hypoxia through adenosine-mediated cullin-1 deneddylation. J. Clin. Invest. 117, 703–711 (2007).
Mikus, P. & Zundel, W. COPing with hypoxia. Semin. Cell Dev. Biol. 16, 462–473 (2005).
Grimaldi, B. et al. PER2 controls lipid metabolism by direct regulation of PPARγ. Cell Metab. 12, 509–520 (2010).
Jennings, R.B. & Reimer, K.A. The cell biology of acute myocardial ischemia. Annu. Rev. Med. 42, 225–246 (1991).
Jaswal, J.S., Keung, W., Wang, W., Ussher, J.R. & Lopaschuk, G.D. Targeting fatty acid and carbohydrate oxidation—a novel therapeutic intervention in the ischemic and failing heart. Biochim. Biophys. Acta 1813, 1333–1350 (2011).
Finegan, B.A., Lopaschuk, G.D., Coulson, C.S. & Clanachan, A.S. Adenosine alters glucose use during ischemia and reperfusion in isolated rat hearts. Circulation 87, 900–908 (1993).
Lopaschuk, G.D. & Stanley, W.C. Glucose metabolism in the ischemic heart. Circulation 95, 313–315 (1997).
Semenza, G.L., Roth, P.H., Fang, H.M. & Wang, G.L. Transcriptional regulation of genes encoding glycolytic enzymes by hypoxia-inducible factor 1. J. Biol. Chem. 269, 23757–23763 (1994).
Safran, M. et al. Mouse model for noninvasive imaging of HIF prolyl hydroxylase activity: assessment of an oral agent that stimulates erythropoietin production. Proc. Natl. Acad. Sci. USA 103, 105–110 (2006).
Kong, T., Westerman, K.A., Faigle, M., Eltzschig, H.K. & Colgan, S.P. HIF-dependent induction of adenosine A2B receptor in hypoxia. FASEB J. 20, 2242–2250 (2006).
Reppert, S.M. & Weaver, D.R. Coordination of circadian timing in mammals. Nature 418, 935–941 (2002).
Bendová, Z. & Sumova, S. Photoperiodic regulation of PER1 and PER2 protein expression in rat peripheral tissues. Physiol. Res. 55, 623–632 (2006).
Wolff, A.A., Rotmensch, H.H., Stanley, W.C. & Ferrari, R. Metabolic approaches to the treatment of ischemic heart disease: the clinicians' perspective. Heart Fail. Rev. 7, 187–203 (2002).
Schulte, G. & Fredholm, B.B. The G(s)-coupled adenosine A(2B) receptor recruits divergent pathways to regulate ERK1/2 and p38. Exp. Cell Res. 290, 168–176 (2003).
Petrzilka, S., Taraborrelli, C., Cavadini, G., Fontana, A. & Birchler, T. Clock gene modulation by TNF-α depends on calcium and p38 MAP kinase signaling. J. Biol. Rhythms 24, 283–294 (2009).
O'Neill, J.S., Maywood, E.S., Chesham, J.E., Takahashi, J.S. & Hastings, M.H. cAMP-dependent signaling as a core component of the mammalian circadian pacemaker. Science 320, 949–953 (2008).
Ginty, D.D. et al. Regulation of CREB phosphorylation in the suprachiasmatic nucleus by light and a circadian clock. Science 260, 238–241 (1993).
Dibner, C., Schibler, U. & Albrecht, U. The mammalian circadian timing system: organization and coordination of central and peripheral clocks. Annu. Rev. Physiol. 72, 517–549 (2010).
Peliciari-Garcia, R.A. et al. Expression of circadian clock and melatonin receptors within cultured rat cardiomyocytes. Chronobiol. Int. 28, 21–30 (2011).
Zhang, J., Kaasik, K., Blackburn, M.R. & Lee, C.C. Constant darkness is a circadian metabolic signal in mammals. Nature 439, 340–343 (2006).
Daniels, I.S. et al. A role of erythrocytes in adenosine monophosphate initiation of hypometabolism in mammals. J. Biol. Chem. 285, 20716–20723 (2010).
Muller, J.E. et al. Circadian variation in the frequency of onset of acute myocardial infarction. N. Engl. J. Med. 313, 1315–1322 (1985).
Suárez-Barrientos, A. et al. Circadian variations of infarct size in acute myocardial infarction. Heart 97, 970–976 (2011).
Reiter, R., Swingen, C., Moore, L., Henry, T.D. & Traverse, J.H. Circadian dependence of infarct size and left ventricular function after ST elevation myocardial infarction. Circ. Res. 110, 105–110 (2012).
Leibetseder, V. et al. Clock genes display rhythmic expression in human hearts. Chronobiol. Int. 26, 621–636 (2009).
Roenneberg, T., Kumar, C.J. & Merrow, M. The human circadian clock entrains to sun time. Curr. Biol. 17, R44–R45 (2007).
Tian, R. & Abel, E.D. Responses of GLUT4-deficient hearts to ischemia underscore the importance of glycolysis. Circulation 103, 2961–2966 (2001).
Russell, R.R. III et al. AMP-activated protein kinase mediates ischemic glucose uptake and prevents postischemic cardiac dysfunction, apoptosis, and injury. J. Clin. Invest. 114, 495–503 (2004).
Hyvärinen, J. et al. Hearts of hypoxia-inducible factor prolyl 4-hydroxylase-2 hypomorphic mice show protection against acute ischemia-reperfusion injury. J. Biol. Chem. 285, 13646–13657 (2010).
Denko, N.C. Hypoxia, HIF1 and glucose metabolism in the solid tumour. Nat. Rev. Cancer 8, 705–713 (2008).
Durgan, D.J. & Young, M.E. The cardiomyocyte circadian clock: emerging roles in health and disease. Circ. Res. 106, 647–658 (2010).
Rutter, J., Reick, M. & McKnight, S.L. Metabolism and the control of circadian rhythms. Annu. Rev. Biochem. 71, 307–331 (2002).
Albrecht, U. Invited review: regulation of mammalian circadian clock genes. J. Appl. Physiol. 92, 1348–1355 (2002).
Durgan, D.J. et al. Short communication: ischemia/reperfusion tolerance is time-of-day-dependent: mediation by the cardiomyocyte circadian clock. Circ. Res. 106, 546–550 (2010).
Nossuli, T.O. et al. A chronic mouse model of myocardial ischemia-reperfusion: essential in cytokine studies. Am. J. Physiol. Heart Circ. Physiol. 278, H1049–H1055 (2000).
Virag, J.A. et al. Attenuation of myocardial injury in mice with functional deletion of the circadian rhythm gene mPer2. Am. J. Physiol. Heart Circ. Physiol. 298, H1088–H1095 (2010).
Wang, C.Y. et al. Increased vascular senescence and impaired endothelial progenitor cell function mediated by mutation of circadian gene Per2. Circulation 118, 2166–2173 (2008).
Viswambharan, H. et al. Mutation of the circadian clock gene Per2 alters vascular endothelial function. Circulation 115, 2188–2195 (2007).
Zheng, B. et al. Nonredundant roles of the mPer1 and mPer2 genes in the mammalian circadian clock. Cell 105, 683–694 (2001).
Ryan, H.E. et al. Hypoxia-inducible factor-1α is a positive factor in solid tumor growth. Cancer Res. 60, 4010–4015 (2000).
Sohal, D.S. et al. Temporally regulated and tissue-specific gene manipulations in the adult and embryonic heart using a tamoxifen-inducible Cre protein. Circ. Res. 89, 20–25 (2001).
Yoo, S.H. et al. PERIOD2:LUCIFERASE real-time reporting of circadian dynamics reveals persistent circadian oscillations in mouse peripheral tissues. Proc. Natl. Acad. Sci. USA 101, 5339–5346 (2004).
Eckle, T. et al. Systematic evaluation of a novel model for cardiac ischemic preconditioning in mice. Am. J. Physiol. Heart Circ. Physiol. 291, H2533–H2540 (2006).
Eltzschig, H.K. et al. Coordinated adenine nucleotide phosphohydrolysis and nucleoside signaling in posthypoxic endothelium: role of ectonucleotidases and adenosine A2B receptors. J. Exp. Med. 198, 783–796 (2003).
Wolska, B.M. & Solaro, R.J. Method for isolation of adult mouse cardiac myocytes for studies of contraction and microfluorimetry. Am. J. Physiol. 271, H1250–H1255 (1996).
Walker, L.A., Walker, J.S., Ambler, S.K. & Buttrick, P.M. Stage-specific changes in myofilament protein phosphorylation following myocardial infarction in mice. J. Mol. Cell. Cardiol. 48, 1180–1186 (2010).
Acknowledgements
The authors acknowledge S.A. Eltzschig (Mucosal Inflammation Program, Department of Anesthesiology, University of Colorado Denver, Aurora, Colorado, USA) for artwork during manuscript preparation, A. Medway (Division of Cardiology, Department of Medicine, University of Colorado Denver, Aurora, Colorado, USA), K. Hoffmann, J. Macas and C. Zachskorn (Institute of Neurology (Edinger Institute), University of Frankfurt, Frankfurt, Germany) and M. Bonney (Mucosal Inflammation Program, Department of Anesthesiology, University of Colorado Denver, Aurora, Colorado, USA) for technical assistance and C.-C. Lee (Department of Biochemistry and Molecular Biology, University of Texas Health Science Center, Houston, Texas, USA) for providing the Per1−/− mice. The present research work is supported by National Heart, Lung and Blood Institute grants R01-HL092188, R01-DK083385 and R01-HL098294 to H.K.E., R01-HL060569 to S.P.C. and 1K08HL102267 to T.E., Foundation for Anesthesia Education and Research grants to T.E. and H.K.E., an American Heart Association grant to T.E. and a Crohn's and Colitis Foundation of America grant to H.K.E.
Author information
Authors and Affiliations
Contributions
T.E. designed and supervised the study, wrote the manuscript and did mouse surgery. K.H. did western blots, RT-PCRs and siRNA knockdown studies. S.B. did western blots, coimmunoprecipitation, promoter studies, ELISAs and animal experiments. S.R. did western blots, RT-PCRs, ELISAs and mouse experiments. M.M. did immunohistochemistry and electron microscopy. L.A.W. isolated mouse myocytes and supervised the study. B.D.L. provided human heart samples. J.H., C.H.B. and D.J.K. did metabolic analysis. P.M.B., S.P.C. and H.K.E. supervised the study and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–19, Supplementary Tables 1–7 and Supplementary Methods (PDF 3456 kb)
Rights and permissions
About this article
Cite this article
Eckle, T., Hartmann, K., Bonney, S. et al. Adora2b-elicited Per2 stabilization promotes a HIF-dependent metabolic switch crucial for myocardial adaptation to ischemia. Nat Med 18, 774–782 (2012). https://doi.org/10.1038/nm.2728
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.2728
This article is cited by
-
Hypoxia-induced signaling in the cardiovascular system: pathogenesis and therapeutic targets
Signal Transduction and Targeted Therapy (2023)
-
Expression of HO1 and PER2 can predict the incidence of delirium in trauma patients with concomitant brain injury
Scientific Reports (2021)
-
Circadian rhythm as a therapeutic target
Nature Reviews Drug Discovery (2021)
-
Characterization of a Murine Model System to Study MicroRNA-147 During Inflammatory Organ Injury
Inflammation (2021)
-
Circadian rhythms and the molecular clock in cardiovascular biology and disease
Nature Reviews Cardiology (2019)