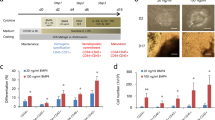
Abstract
Hematopoietic stem cell (HSC) self-renewal is regulated by both intrinsic and extrinsic signals. Although some of the pathways that regulate HSC self-renewal have been uncovered, it remains largely unknown whether these pathways can be triggered by deliverable growth factors to induce HSC growth or regeneration. Here we show that pleiotrophin, a neurite outgrowth factor with no known function in hematopoiesis, efficiently promotes HSC expansion in vitro and HSC regeneration in vivo. Treatment of mouse bone marrow HSCs with pleiotrophin caused a marked increase in long-term repopulating HSC numbers in culture, as measured in competitive repopulating assays. Treatment of human cord blood CD34+CDCD38−Lin− cells with pleiotrophin also substantially increased severe combined immunodeficient (SCID)-repopulating cell counts in culture, compared to input and cytokine-treated cultures. Systemic administration of pleiotrophin to irradiated mice caused a pronounced expansion of bone marrow stem and progenitor cells in vivo, indicating that pleiotrophin is a regenerative growth factor for HSCs. Mechanistically, pleiotrophin activated phosphoinositide 3-kinase (PI3K) signaling in HSCs; antagonism of PI3K or Notch signaling inhibited pleiotrophin-mediated expansion of HSCs in culture. We identify the secreted growth factor pleiotrophin as a new regulator of both HSC expansion and regeneration.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Accession codes
References
Zon, L.I. Intrinsic and extrinsic control of haematopoietic stem-cell self-renewal. Nature 453, 306–313 (2008).
Orkin, S.H. & Zon, L.I. SnapShot: hematopoiesis. Cell 132, 712 (2008).
Kiel, M.J. & Morrison, S.J. Uncertainty in the niches that maintain haematopoietic stem cells. Nat. Rev. Immunol. 8, 290–301 (2008).
Varnum-Finney, B. et al. The Notch ligand, Jagged-1, influences the development of primitive hematopoietic precursor cells. Blood 91, 4084–4091 (1998).
Stier, S., Cheng, T., Dombkowski, D., Carlesso, N. & Scadden, D.T. Notch1 activation increases hematopoietic stem cell self-renewal in vivo and favors lymphoid over myeloid lineage outcome. Blood 99, 2369–2378 (2002).
Reya, T. et al. A role for Wnt signalling in self-renewal of haematopoietic stem cells. Nature 423, 409–414 (2003).
Karlsson, G. et al. Smad4 is critical for self-renewal of hematopoietic stem cells. J. Exp. Med. 204, 467–474 (2007).
Zhang, C.C. et al. Angiopoietin-like proteins stimulate ex vivo expansion of hematopoietic stem cells. Nat. Med. 12, 240–245 (2006).
North, T.E. et al. Prostaglandin E2 regulates vertebrate haematopoietic stem cell homeostasis. Nature 447, 1007–1011 (2007).
Hackney, J.A. et al. A molecular profile of a hematopoietic stem cell niche. Proc. Natl. Acad. Sci. USA 99, 13061–13066 (2002).
Chute, J.P. et al. Ex vivo culture with human brain endothelial cells increases the SCID-repopulating capacity of adult human bone marrow. Blood 100, 4433–4439 (2002).
Antonchuk, J., Sauvageau, G. & Humphries, R.K. HOXB4-induced expansion of adult hematopoietic stem cells ex vivo. Cell 109, 39–45 (2002).
Blank, U., Karlsson, G. & Karlsson, S. Signaling pathways governing stem-cell fate. Blood 111, 492–503 (2008).
Meng, K. et al. Pleiotrophin signals increased tyrosine phosphorylation of β-catenin through inactivation of the intrinsic catalytic activity of the receptor-type protein tyrosine phosphatase β/ζ. Proc. Natl. Acad. Sci. USA 97, 2603–2608 (2000).
Stoica, G.E. et al. Identification of anaplastic lymphoma kinase as a receptor for the growth factor pleiotrophin. J. Biol. Chem. 276, 16772–16779 (2001).
Landgraf, P., Wahle, P., Pape, H.C., Gundelfinger, E.D. & Kreutz, M.R. The survival-promoting peptide Y-P30 enhances binding of pleiotrophin to syndecan-2 and -3 and supports its neuritogenic activity. J. Biol. Chem. 283, 25036–25045 (2008).
Perez-Pinera, P., Berenson, J.R. & Deuel, T.F. Pleiotrophin, a multifunctional angiogenic factor: mechanisms and pathways in normal and pathological angiogenesis. Curr. Opin. Hematol. 15, 210–214 (2008).
Yeh, H.J., He, Y.Y., Xu, J., Hsu, C.Y. & Deuel, T.F. Upregulation of pleiotrophin gene expression in developing microvasculature, macrophages and astrocytes after acute ischemic brain injury. J. Neurosci. 18, 3699–3707 (1998).
Chang, Y. et al. Secretion of pleiotrophin stimulates breast cancer progression through remodeling of the tumor microenvironment. Proc. Natl. Acad. Sci. USA 104, 10888–10893 (2007).
Chute, J.P., Muramoto, G.G., Fung, J. & Oxford, C. Soluble factors elaborated by human brain endothelial cells induce the concomitant expansion of purified human bone marrow CD34+CDCD38− cells and SCID-repopulating cells. Blood 105, 576–583 (2005).
Chute, J.P. et al. Transplantation of vascular endothelial cells mediates the hematopoietic recovery and survival of lethally irradiated mice. Blood 109, 2365–2372 (2007).
Gottschling, S. et al. Human mesenchymal stromal cells regulate initial self-renewing divisions of hematopoietic progenitor cells by a β1-integrin–dependent mechanism. Stem Cells 25, 798–806 (2007).
Salter, A.B. et al. Endothelial progenitor cell infusion induces hematopoietic stem cell reconstitution in vivo. Blood 113, 2104–2107 (2009).
Goodell, M.A., Brose, K., Paradis, G., Conner, A. & Mulligan, R. Isolation and functional properties of murine hematopoietic stem cells that are replicating in vivo. J. Exp. Med. 183, 1797–1806 (1996).
Christopherson, K.W., Hangoc, G., Mantel, C. & Broxmeyer, H.E. Modulation of hematopoietic stem cell homing and engraftment by CD26. Science 305, 1000–1003 (2004).
Deuel, T.F., Zhang, N., Yeh, H.J., Silos-Santiago, I. & Wang, Z.Y. Pleiotrophin: a cytokine with diverse functions and a novel signaling pathway. Arch. Biochem. Biophys. 397, 162–171 (2002).
Souttou, B., Ahmad, S., Riegel, A.T. & Wellstein, A. Signal transduction pathways involved in the mitogenic activity of pleiotrophin. Implication of mitogen-activated protein kinase and phosphoinositide 3-kinase pathways. J. Biol. Chem. 272, 19588–19593 (1997).
Gu, D. et al. The effect of pleiotrophin signaling on adipogenesis. FEBS Lett. 581, 382–388 (2007).
Kunisato, A. et al. HES-1 preserves purified hematopoietic stem cells ex vivo and accumulates side population cells in vivo. Blood 101, 1777–1783 (2003).
Calvi, L.M. et al. Osteoblastic cells regulate the haematopoietic stem cell niche. Nature 425, 841–846 (2003).
Palomero, T., Dominguez, M. & Ferrando, A.A. The role of the PTEN/AKT pathway in NOTCH1-induced leukemia. Cell Cycle 7, 965–970 (2008).
Carracedo, A. & Pandolfi, P.P. The PTEN-PI3K pathway: of feedbacks and cross-talks. Oncogene 27, 5527–5541 (2008).
Hooper, A.T. et al. Engraftment and reconstitution of hematopoiesis is dependent on VEGFR2-mediated regeneration of sinusoidal endothelial cells. Cell Stem Cell 4, 263–274 (2009).
Congdon, K.L. et al. Activation of Wnt signaling in hematopoietic regeneration. Stem Cells 26, 1202–1210 (2008).
Zhang, J. et al. PTEN maintains haematopoietic stem cells and acts in lineage choice and leukaemia prevention. Nature 441, 518–522 (2006).
Miyamoto, K. et al. Foxo3a is essential for maintenance of the hematopoietic stem cell pool. Cell Stem Cell 1, 101–112 (2007).
Adams, G.B. et al. Therapeutic targeting of a stem cell niche. Nat. Biotechnol. 25, 238–243 (2007).
Dressman, H. et al. Gene expression signatures that predict radiation exposure in mice and humans. PLoS Med. 4, e106 (2007).
Chute, J.P. et al. Inhibition of aldehyde dehydrogenase and retinoid signaling induces the expansion of human hematopoietic stem cells. Proc. Natl. Acad. Sci. USA 103, 11707–11712 (2006).
Dorrell, C., Gan, O., Hawley, R. & Dick, J. Expansion of human CB CD34+CD38− in ex vivo culture during retroviral transduction without a corresponding increase in SCID-repopulating cell frequency: dissocation of SRC function and phenotype. Blood 95, 102–110 (2000).
Acknowledgements
We acknowledge J. Whitesides for assistance with cell sorting procedures. This work was supported in part by US National Institutes of Health grant AI067798 to J.P.C., H.A.H. is supported by a post-doctoral training grant from the Center for Biomolecular and Tissue Engineering, US National Institute of Biomedical Imaging and Bioengineering.
Author information
Authors and Affiliations
Contributions
H.A.H. designed and performed experiments, analyzed data and wrote the paper; G.G.M., P.D., S.K.M., J.L.R., P.D., A.B.S. and W.E.L. performed experiments; J.-T.C. guided the microarray analysis; T.R. and N.J.C. analyzed data and wrote the paper; J.P.C. designed the experiments, analyzed the data and wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figs. 1–5, Supplementary Tables 1 and 2 and Supplementary Methods (PDF 799 kb)
Rights and permissions
About this article
Cite this article
Himburg, H., Muramoto, G., Daher, P. et al. Pleiotrophin regulates the expansion and regeneration of hematopoietic stem cells. Nat Med 16, 475–482 (2010). https://doi.org/10.1038/nm.2119
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.2119
This article is cited by
-
Rational engineering of lung alveolar epithelium
npj Regenerative Medicine (2023)
-
Cyclical dermal micro-niche switching governs the morphological infradian rhythm of mouse zigzag hair
Nature Communications (2023)
-
Engineering a niche supporting hematopoietic stem cell development using integrated single-cell transcriptomics
Nature Communications (2022)
-
Pleiotrophin Potentiates Sevoflurane Anesthesia-induced Learning Deficits in Mice
Journal of Molecular Neuroscience (2022)
-
Effect of expansion of human umbilical cord blood CD34 + cells on neurotrophic and angiogenic factor expression and function
Cell and Tissue Research (2022)