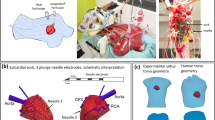
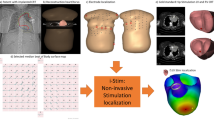
Abstract
Over 7 million people worldwide die annually from erratic heart rhythms (cardiac arrhythmias), and many more are disabled. Yet there is no imaging modality to identify patients at risk, provide accurate diagnosis and guide therapy. Standard diagnostic techniques such as the electrocardiogram (ECG) provide only low-resolution projections of cardiac electrical activity on the body surface. Here we demonstrate the successful application in humans of a new imaging modality called electrocardiographic imaging (ECGI), which noninvasively images cardiac electrical activity in the heart. In ECGI, a multielectrode vest records 224 body-surface electrocardiograms; electrical potentials, electrograms and isochrones are then reconstructed on the heart's surface using geometrical information from computed tomography (CT) and a mathematical algorithm. We provide examples of ECGI application during atrial and ventricular activation and ventricular repolarization in (i) normal heart (ii) heart with a conduction disorder (right bundle branch block) (iii) focal activation initiated by right or left ventricular pacing, and (iv) atrial flutter.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Rudy, Y. & Burnes, J.E. Noninvasive electrocardiographic imaging. Ann. Noninvasive Electrocardiol. 4, 340–358 (1999).
Oster, H.S., Taccardi, B., Lux, R.L., Ershler, P.R. & Rudy, Y. Noninvasive electrocardiographic imaging: reconstruction of epicardial potentials, electrograms, and isochrones and localization of single and multiple electrocardiac events. Circulation 96, 1012–1024 (1997).
Oster, H.S., Taccardi, B., Lux, R.L., Ershler, P.R. & Rudy, Y. Electrocardiographic imaging: Noninvasive characterization of intramural myocardial activation from inverse-reconstructed epicardial potentials and electrograms. Circulation 97, 1496–1507 (1998).
Burnes, J.E., Taccardi, B., MacLeod, R.S. & Rudy, Y. Noninvasive ECG imaging of electrophysiologically abnormal substrates in infarcted hearts: a model study. Circulation 101, 533–540 (2000).
Burnes, J.E., Taccardi, B. & Rudy, Y. A noninvasive imaging modality for cardiac arrhythmias. Circulation 102, 2152–2158 (2000).
Burnes, J.E., Taccardi, B., Ershler, P.R. & Rudy, Y. Noninvasive electrocardiographic imaging of substrate and intramural ventricular tachycardia in infarcted hearts. J. Am. Coll. Cardiol. 38, 2071–2078 (2001).
Ghanem, R.N., Burnes, J.E., Waldo, A.L. & Rudy, Y. Imaging dispersion of myocardial repolarization, II: noninvasive reconstruction of epicardial measures. Circulation 104, 1306–1312 (2001).
Durrer, D. et al. Total excitation of the isolated human heart. Circulation 41, 899–912 (1970).
Arisi, G., Macchi, E., Baruffi, S., Spaggiari, S. & Taccardi, B. Potential fields on the ventricular surface of the exposed dog heart during normal excitation. Circ. Res. 52, 706–715 (1983).
Wyndham, C.R. et al. Epicardial activation of the intact human heart without conduction defect. Circulation 59, 161–168 (1979).
Kupersmith, J. Electrophysiologic mapping during open heart surgery. Prog. Cardiovasc. Dis. XIX, 167–202 (1976).
Spach, M.S., Barr, R.C., Lanning, C.F. & Tucek, P.C. Origin of body surface QRS and T wave potentials from epicardial potential distributions in the intact chimpanzee. Circulation 55, 268–268 (1977).
Jouve, A. et al. Epicardial leads in man. Am. Heart J. 59, 856–868 (1959).
Roos, J.P., van Dam, R.T. & Durrer, D. Epicardial and intramural excitation of normal heart in six patients 50 years of age and older. Br. Heart J. 30, 630–637 (1968).
Lux, R.L., Green, L.S., MacLeod, R.S. & Taccardi, B. Assessment of spatial and temporal characteristics of ventricular repolarization. J. Electrocardiol. 27, 100–105 (1994).
Gima, K. & Rudy, Y. Ionic current basis of electrocardiographic waveforms: a model study. Circ. Res. 90, 889–896 (2002).
Liebman, J., Rudy, Y., Diaz, P., Thomas, C.W. & Plonsey, R. The spectrum of right bundle branch block as manifested in electrocardiographic body surface potential maps. J. Electrocardiol. 17, 329–346 (1984).
Matsuo, K., Uno, K., Khrestian, C.M. & Waldo, A.L. Conduction left-to-right and right-to-left across the crista terminalis. Am. J. Physiol. Heart Circ. Physiol. 280, H1683–H1691 (2001).
Yamabe, H. et al. Conduction properties of the crista terminalis and its influence on the right atrial activation sequence in patients with typical atrial flutter. J. Pacing Clin. Electrophysiol. 25, 132–141 (2002).
Daoud, E.G. & Morady, F. Pathophysiology of atrial flutter. Ann. Rev. Med. 49, 77–83 (1998).
Rodriguez, L.-M., Timmermans, C., Nabar, A., Hofstra, L. & Wellens, H.J.J. Biatrial activation in isthmus-dependent atrial flutter. Circulation 104, 2545–2550 (2001).
Janse, M.J. & Wit, A.L. Electrophysiological mechanisms of ventricular arrhythmias resulting from myocardial ischemia and infarction. Physiol. Rev. 69, 1049–1169 (1989).
Sosa, E., Scanavacca, M. & D'Avila, A. Endocardial and epicardial ablation guided by nonsurgical transthoracic epicardial mapping to treat recurrent ventricular tachycardia. J. Cardiovasc. Electrophysiol. 9, 229–239 (1998).
Ghanem, R.N., Ramanathan, C., Jia, P. & Rudy, Y. Heart-surface reconstruction and ECG electrodes localization using fluoroscopy, epipolar geometry and stereovision: application to noninvasive imaging of cardiac electrical activity. IEEE Trans. Med. Imaging 22, 1307–1318 (2003).
Tikhonov, A.N. & Arsenin, V.Y. The regularization method. in Solutions of Ill-Posed Problems 45–94 (V.H. Winston & Sons, Washington DC, 1977).
Rudy, Y. & Oster, H.S. The electrocardiographic inverse problem. Crit. Rev. Biomed. Eng. 20, 25–45 (1992).
Ramanathan, C., Jia, P., Ghanem, R.N., Calvetti, D. & Rudy, Y. Noninvasive electrocardiographic imaging (ECGI): application of the generalized minimal residual method (GMRes). Ann. Biomed. Eng. 31, 981–994 (2003).
Biermann, M. et al. The interpretation of cardiac electrograms. in Cardiac Mapping (eds. Shenasa, M., Borggrefe, M. & Breithardt, G.) 15–39 (Futura Publishing, Mount Kisco, New York, 2003).
Haws, C.W. & Lux, R.L. Correlation between in vivo transmembrane action potential durations and activation-recovery intervals from electrograms. Effects of interventions that alter repolarization time. Circulation 81, 281–288 (1990).
Acknowledgements
This study was supported by National Institutes of Health/National Heart, Lung and Blood Institute grants R37-HL-33343 and R01-HL-49054 to Y.R. We thank L. Ciancibello for technical assistance in CT imaging; J. Haaga for access to CT; J. Sahadevan, A. Markowitz, A. Waldo, N. Varma and P. Dorostkar for their time, expert advice and access to electrophysiology facilities and patients; and E.T. DuPont for assistance in transporting images from the hospital to our computer facility.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
C.R., R.N.G., P.J. and Y.R. are inventors on pending patent applications related to this technology. C.R. and P.J. have the prospect of future employment by a company for application of this technology.
Rights and permissions
About this article
Cite this article
Ramanathan, C., Ghanem, R., Jia, P. et al. Noninvasive electrocardiographic imaging for cardiac electrophysiology and arrhythmia. Nat Med 10, 422–428 (2004). https://doi.org/10.1038/nm1011
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm1011
This article is cited by
-
Electrocardiographic imaging in the atria
Medical & Biological Engineering & Computing (2023)
-
Technical development and feasibility of a reusable vest to integrate cardiovascular magnetic resonance with electrocardiographic imaging
Journal of Cardiovascular Magnetic Resonance (2023)
-
Study protocol: MyoFit46—the cardiac sub-study of the MRC National Survey of Health and Development
BMC Cardiovascular Disorders (2022)
-
3-Dimensional ventricular electrical activation pattern assessed from a novel high-frequency electrocardiographic imaging technique: principles and clinical importance
Scientific Reports (2021)
-
Negative-ResNet: noisy ambulatory electrocardiogram signal classification scheme
Neural Computing and Applications (2021)