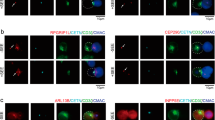
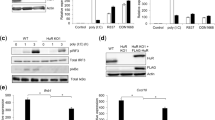
Abstract
DC-SIGN, a C-type lectin expressed on dendritic cells (DCs), can sequester human immunodeficiency virus (HIV) virions in multivesicular bodies. Here, using large-scale gene expression profiling and tyrosine-phosphorylated proteome analyses, we characterized signaling mediated by DC-SIGN after activation by either HIV or a DC-SIGN-specific antibody. Activation of DC-SIGN resulted in downregulation of genes encoding major histocompatibility complex class II, Jagged 1 and interferon-response molecules and upregulation of the gene encoding transcription factor ATF3. Phosphorylated proteome analysis showed that HIV- or antibody-stimulated DC-SIGN signaling was mediated by the Rho guanine nucleotide–exchange factor LARG and led to increased Rho-GTPase activity. Activation of LARG in DCs exposed to HIV was required for the formation of virus–T cell synapses. Thus, HIV sequestration by and stimulation of DC-SIGN helps HIV evade immune responses and spread to cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Accession codes
References
Iwasaki, A. & Medzhitov, R. Toll-like receptor control of the adaptive immune responses. Nat. Immunol. 5, 987–995 (2004).
Oda, K. & Kitano, H. A comprehensive map of the Toll-like receptor signaling network. Mol. Syst. Biol. 2, 2006.0015 (2006).
Cambi, A. & Figdor, C.G. Levels of complexity in pathogen recognition by C-type lectins. Curr. Opin. Immunol. 17, 345–351 (2005).
McGreal, E.P., Miller, J.L. & Gordon, S. Ligand recognition by antigen-presenting cell C-type lectin receptors. Curr. Opin. Immunol. 17, 18–24 (2005).
Gross, O. et al. Card9 controls a non-TLR signalling pathway for innate anti-fungal immunity. Nature 442, 651–656 (2006).
Rogers, N.C. et al. Syk-dependent cytokine induction by Dectin-1 reveals a novel pattern recognition pathway for C type lectins. Immunity 22, 507–517 (2005).
Geijtenbeek, T.B. et al. Identification of DC-SIGN, a novel dendritic cell-specific ICAM-3 receptor that supports primary immune responses. Cell 100, 575–585 (2000).
van Kooyk, Y. & Geijtenbeek, T.B. DC-SIGN: escape mechanism for pathogens. Nat. Rev. Immunol. 3, 697–709 (2003).
Geijtenbeek, T.B. et al. DC-SIGN, a dendritic cell-specific HIV-1-binding protein that enhances trans-infection of T cells. Cell 100, 587–597 (2000).
Turville, S.G. et al. Diversity of receptors binding HIV on dendritic cell subsets. Nat. Immunol. 3, 975–983 (2002).
Garcia, E. et al. HIV-1 trafficking to the dendritic cell-T-cell infectious synapse uses a pathway of tetraspanin sorting to the immunological synapse. Traffic 6, 488–501 (2005).
Turville, S.G. et al. Immunodeficiency virus uptake, turnover, and 2-phase transfer in human dendritic cells. Blood 103, 2170–2179 (2004).
Moris, A. et al. DC-SIGN promotes exogenous MHC-I-restricted HIV-1 antigen presentation. Blood 103, 2648–2654 (2004).
Arrighi, J.F. et al. Lentivirus-mediated RNA interference of DC-SIGN expression inhibits human immunodeficiency virus transmission from dendritic cells to T cells. J. Virol. 78, 10848–10855 (2004).
Arrighi, J.F. et al. DC-SIGN-mediated infectious synapse formation enhances X4 HIV-1 transmission from dendritic cells to T cells. J. Exp. Med. 200, 1279–1288 (2004).
Caparros, E. et al. DC-SIGN ligation on dendritic cells results in ERK and PI3K activation and modulates cytokine production. Blood 107, 3950–3958 (2006).
Dillon, S. et al. Toll-like receptor 2 ligand stimulates Th2 responses in vivo, via induction of extracellular signal-regulated kinase mitogen-activated protein kinase and c-Fos in dendritic cells. J. Immunol. 172, 4733–4743 (2004).
Napolitani, G., Rinaldi, A., Bertoni, F., Sallusto, F. & Lanzavecchia, A. Selected Toll-like receptor agonist combinations synergistically trigger a T helper type 1–polarizing program in dendritic cells. Nat. Immunol. 6, 769–776 (2005).
Gilchrist, M. et al. Systems biology approaches identify ATF3 as a negative regulator of Toll-like receptor 4. Nature 441, 173–178 (2006).
Rush, J. et al. Immunoaffinity profiling of tyrosine phosphorylation in cancer cells. Nat. Biotechnol. 23, 94–101 (2005).
Kratchmarova, I., Blagoev, B., Haack-Sorensen, M., Kassem, M. & Mann, M. Mechanism of divergent growth factor effects in mesenchymal stem cell differentiation. Science 308, 1472–1477 (2005).
Benvenuti, F. et al. Requirement of Rac1 and Rac2 expression by mature dendritic cells for T cell priming. Science 305, 1150–1153 (2004).
Eun, S.Y. Cutting edge: rho activation and actin polarization are dependent on plexin-A1 in dendritic cells. J. Immunol. 177, 4271–4275 (2006).
Deckert, M., Moon, C. & Le Bras, S. The immunological synapse and Rho GTPases. Curr. Top. Microbiol. Immunol. 291, 61–90 (2005).
Lore, K. et al. Accumulation of DC-SIGN+CD40+ dendritic cells with reduced CD80 and CD86 expression in lymphoid tissue during acute HIV-1 infection. AIDS 16, 683–692 (2002).
Granelli-Piperno, A., Golebiowska, A., Trumpfheller, C., Siegal, F.P. & Steinman, R.M. HIV-1-infected monocyte-derived dendritic cells do not undergo maturation but can elicit IL-10 production and T cell regulation. Proc. Natl. Acad. Sci. USA 101, 7669–7674 (2004).
Harman, A.N. et al. HIV induces maturation of monocyte-derived dendritic cells and Langerhans cells. J. Immunol. 177, 7103–7113 (2006).
Moris, A. et al. Dendritic cells and HIV-specific CD4+ T cells: HIV antigen presentation, T-cell activation, and viral transfer. Blood 108, 1643–1651 (2006).
Miller, M.J., Wei, S.H., Parker, I. & Cahalan, M.D. Two-photon imaging of lymphocyte motility and antigen response in intact lymph node. Science 296, 1869–1873 (2002).
Tao, L. et al. Opposing roles of serine/threonine kinases MEKK1 and LOK in regulating the CD28 responsive element in T-cells. Biochem. J. 2363, 175–182 (2002).
Swetman, C.A. et al. Extension, retraction and contraction in the formation of a dendritic cell dendrite: distinct roles for Rho GTPases. Eur. J. Immunol. 32, 2074–2083 (2002).
Loomis, R.J. et al. Citron kinase, a RhoA effector, enhances HIV-1 virion production by modulating exocytosis. Traffic 7, 1643–1653 (2006).
Mitsushima, M., Suwa, A., Amachi, T., Ueda, K. & Kioka, N. Extracellular signal-regulated kinase activated by epidermal growth factor and cell adhesion interacts with and phosphorylates vinexin. J. Biol. Chem. 279, 34570–34577 (2004).
Suwa, A. et al. Vinexin β regulates the anchorage dependence of ERK2 activation stimulated by epidermal growth factor. J. Biol. Chem. 277, 13053–13058 (2002).
Turville, S.G. et al. HIV gp120 receptors on human dendritic cells. Blood 98, 2482–2488 (2001).
Jefford, M. et al. Functional comparison of DCs generated in vivo with Flt3 ligand or in vitro from blood monocytes: differential regulation of function by specific classes of physiologic stimuli. Blood 102, 1753–1763 (2003).
Granelli-Piperno, A. et al. Dendritic cell-specific intercellular adhesion molecule 3-grabbing nonintegrin/CD209 is abundant on macrophages in the normal human lymph node and is not required for dendritic cell stimulation of the mixed leukocyte reaction. J. Immunol. 175, 4265–4273 (2005).
Brenchley, J.M. et al. CD4+ T cell depletion during all stages of HIV disease occurs predominantly in the gastrointestinal tract. J. Exp. Med. 200, 749–759 (2004).
Kinther, M. & Sherman, N.E. Protein Sequencing and Identification Using Tandem Mass Spectrometry (John Wiley, New York, 2000).
Batycka, M. et al. Ultra-fast tandem mass spectrometry scanning combined with monolithic column liquid chromatography increases throughput in proteomic analysis. Rapid Commun. Mass Spectrom. 20, 2074–2080 (2006).
Brazma, A. et al. Minimum information about a microarray experiment (MIAME)-toward standards for microarray data. Nat. Genet. 29, 365–371 (2001).
Bolstad, B.M., Irizarry, R.A., Astrand, M. & Speed, T.P. A comparison of normalization methods for high density oligonucleotide array data based on variance and bias. Bioinformatics 19, 185–193 (2003).
Irizarry, R.A. et al. Exploration, normalization, and summaries of high density oligonucleotide array probe level data. Biostatistics 4, 249–264 (2003).
Wu, Z. & Irizarry, R.A. Preprocessing of oligonucleotide array data. Nat. Biotechnol. 22, 656–658 (2004).
Acknowledgements
We thank K. Luhn for technical assistance and T. Brode for earlier work on the project. Anti-p24 was from M.H. Malim (through the AIDS Research and Reference Reagent Program, Division of AIDS, National Institute of Allergy and Infectious Diseases, National Institutes of Health). Supported by the UK Medical Research Council (A.S., K.S. and B.K.).
Author information
Authors and Affiliations
Contributions
A.H., K.S. and A.S., designed the study, did experiments, analyzed the data and wrote the paper; H.D. made HIV-1 viral stocks; M.E. and B.K. did and analyzed LC-tandem mass spectometry and proteomic experiments; D.B. and K.D. did and analyzed microarray experiments; A. Moris and O.S. provided DC-SIGN THP-1 stable transfectants; and A. McMichael, B.K., A. Moris and O.S. provided additional intellectual input.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Panel of DC-SIGN antibodies tested for signaling but showing undetectable changes following western blot with anti-phosphotyrosine or anti-phosphoserine. (PDF 64 kb)
Supplementary Fig. 2
Thp-1 DC-SIGN transfectants THP-1 + LL/AA and THP-1 + WT tested for signaling induced by H-200 prior to blocking with IB10 non-signaling anti-DC-SIGN. (PDF 48 kb)
Supplementary Table 1
DC-SIGN transcriptional program: selected genes differentially regulated. (PDF 20 kb)
Supplementary Table 2
Summary of differentially phosphorylated proteins. (PDF 23 kb)
Supplementary Table 3
Proteins exclusively phosphorylated following DC-SIGN activation. (PDF 35 kb)
Rights and permissions
About this article
Cite this article
Hodges, A., Sharrocks, K., Edelmann, M. et al. Activation of the lectin DC-SIGN induces an immature dendritic cell phenotype triggering Rho-GTPase activity required for HIV-1 replication. Nat Immunol 8, 569–577 (2007). https://doi.org/10.1038/ni1470
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni1470
This article is cited by
-
Carbohydrates from Pseudomonas aeruginosa biofilms interact with immune C-type lectins and interfere with their receptor function
npj Biofilms and Microbiomes (2021)
-
HIV-1 subtype C transmitted founders modulate dendritic cell inflammatory responses
Retrovirology (2020)
-
DC-SIGN–LEF1/TCF1–miR-185 feedback loop promotes colorectal cancer invasion and metastasis
Cell Death & Differentiation (2020)
-
HIV-1 envelope glycoprotein stimulates viral transcription and increases the infectivity of the progeny virus through the manipulation of cellular machinery
Scientific Reports (2017)
-
Diet-induced reconstruction of mucosal microbiota associated with alterations of epithelium lectin expression and regulation in the maintenance of rumen homeostasis
Scientific Reports (2017)