Abstract
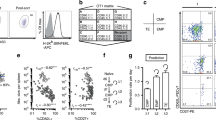
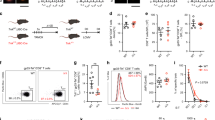
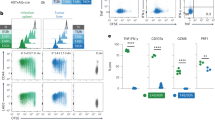
Antigen-driven CD8 memory T cell proliferation is more rapid than that of naive T cells, ensuring efficient control of the pathogen after reinfection. We show here that naive and memory cells are in different states of G0/G1 arrest. Naive cells are in a classical state of G0/G1 arrest, with high expression of p27Kip1 and low CDK6 and CDK2 kinase activity. In contrast, memory cells have low expression of p27Kip1 and high CDK6 kinase activity. This preactivated kinase is associated with cyclin D3 in the cytoplasm, and neutralization of these complexes with antibody to cyclin D3 blocks the rapid division of memory cells. Therefore G0/G1 memory cells are at a unique preactivated state that favors rapid division after antigen stimulation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Freitas, A.A. & Rocha, B. Population biology of lymphocytes: the flight for survival. Annu. Rev. Immunol. 18, 83–111 (2000).
Doherty, P.C., Topham, D.J. & Tripp, R.A. Establishment and persistence of virus-specific CD4+ and CD8+ T cell memory. Immunol. Rev. 150, 23–44 (1996).
Ahmed, R. & Gray, D. Immunological memory and protective immunity: understanding their relation. Science 272, 54–60 (1996).
Sprent, J., Tough, D.F. & Sun, S. Factors controlling the turnover of T memory cells. Immunol. Rev. 156, 79–85 (1997).
Tripp, R.A., Lahti, J.M. & Doherty, P.C. Laser light suicide of proliferating virus-specific CD8+ T cells in an in vivo response. J. Immunol. 155, 3719–3721 (1995).
Veiga-Fernandes, H., Walter, U., Bourgeois, C., McLean, A. & Rocha, B. Response of naive and memory CD8+ T cells to antigen stimulation in vivo. Nat. Immunol. 1, 47–53 (2000).
Sherr, C.J. Mammalian G1 cyclins. Cell 73, 1059–1065 (1993).
Sherr, C.J. & Roberts, J.M. CDK inhibitors: positive and negative regulators of G1-phase progression. Genes Dev. 13, 1501–1512 (1999).
Sherr, C.J. The Pezcoller lecture: cancer cell cycles revisited. Cancer Res. 60, 3689–3695 (2000).
Blain, S.W., Montalvo, E. & Massague, J. Differential interaction of the cyclin-dependent kinase (Cdk) inhibitor p27Kip1 with cyclin A-Cdk2 and cyclin D2-Cdk4. J. Biol. Chem. 272, 25863–25872 (1997).
Cheng, M. et al. The p21Cip1 and p27Kip1 CDK 'inhibitors' are essential activators of cyclin D-dependent kinases in murine fibroblasts. Embo J. 18, 1571–1583 (1999).
LaBaer, J. et al. New functional activities for the p21 family of CDK inhibitors. Genes Dev. 11, 847–862 (1997).
Parry, D., Mahony, D., Wills, K. & Lees, E. Cyclin D-CDK subunit arrangement is dependent on the availability of competing INK4 and p21 class inhibitors. Mol. Cell. Biol. 19, 1775–1783 (1999).
Soos, T.J. et al. Formation of p27-CDK complexes during the human mitotic cell cycle. Cell Growth Differ. 7, 135–146 (1996).
Tanchot, C., Lemonnier, F.A., Perarnau, B., Freitas, A.A. & Rocha, B. Differential requirements for survival and proliferation of CD8 naive or memory T cells. Science 276, 2057–2062 (1997).
Olashaw, N. & Pledger, W.J. Paradigms of growth control: relation to cdk activation. Sci. STKE 2002, Re7 (2002).
Boussiotis, V.A. et al. p27kip1 functions as an anergy factor inhibiting interleukin 2 transcription and clonal expansion of alloreactive human and mouse helper T lymphocytes. Nat. Med. 6, 290–297 (2000).
Mohapatra, S., Agrawal, D. & Pledger, W.J. p27Kip1 regulates T cell proliferation. J. Biol. Chem. 276, 21976–21983 (2001).
Kitagawa, M. et al. The consensus motif for phosphorylation by cyclin D1-Cdk4 is different from that for phosphorylation by cyclin A/E-Cdk2. Embo J. 15, 7060–7069 (1996).
Tourigny, M.R. et al. CDK inhibitor p18INK4c is required for the generation of functional plasma cells. Immunity 17, 179–189 (2002).
Morse, L., Chen, D., Franklin, D., Xiong, Y. & Chen-Kiang, S. Induction of cell cycle arrest and B cell terminal differentiation by CDK inhibitor p18INK4c and IL-6. Immunity 6, 47–56 (1997).
Matsuoka, S. et al. p57KIP2, a structurally distinct member of the p21CIP1 Cdk inhibitor family, is a candidate tumor suppressor gene. Genes Dev. 9, 650–662 (1995).
Balomenos, D. et al. The cell cycle inhibitor p21 controls T-cell proliferation and sex-linked lupus development. Nat. Med. 6, 171–176 (2000).
Parker, S.B. et al. p53-independent expression of p21Cip1 in muscle and other terminally differentiating cells. Science 267, 1024–1027 (1995).
Pines, J. & Hunter, T. The differential localization of human cyclins A and B is due to a cytoplasmic retention signal in cyclin B. Embo J. 13, 3772–3781 (1994).
Dickens, M. et al. A cytoplasmic inhibitor of the JNK signal transduction pathway. Science 277, 693–696 (1997).
Kiyokawa, H. et al. Enhanced growth of mice lacking the cyclin-dependent kinase inhibitor function of p27Kip1. Cell 85, 721–732 (1996).
Nakayama, K. et al. Mice lacking p27Kip1 display increased body size, multiple organ hyperplasia, retinal dysplasia, and pituitary tumors. Cell 85, 707–720 (1996).
Fero, M.L. et al. A syndrome of multiorgan hyperplasia with features of gigantism, tumorigenesis, and female sterility in p27Kip1-deficient mice. Cell 85, 733–744 (1996).
Acknowledgements
We thank C. Garcia for cell sorting; J. Lipecka for confocal microscopy; M. Serrano, T. Toda, F. Uhlmann, C. Reis e Sousa, V. Dulic, V. Boussiotis, F. Sigaux, A.A. Freitas, A. Sarukhan, A. Peixoto, S. Guillaume and C. Saint-Ruf for discussions; and P. Sicinski for allowing us to cite his unpublished data. Supported by Association pour la Recherche sur le Cancer and Ligue pour la Recherche sur le Cancer, and the Technology and Science Foundation (Portugal) and Fondation Recherche Médicale (both H.V.-F.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Rights and permissions
About this article
Cite this article
Veiga-Fernandes, H., Rocha, B. High expression of active CDK6 in the cytoplasm of CD8 memory cells favors rapid division. Nat Immunol 5, 31–37 (2004). https://doi.org/10.1038/ni1015
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni1015
This article is cited by
-
The cyclin D1 carboxyl regulatory domain controls the division and differentiation of hematopoietic cells
Biology Direct (2016)
-
Diabetes mellitus—advances and challenges in human β-cell proliferation
Nature Reviews Endocrinology (2015)
-
Real-time tracking of cell cycle progression during CD8+ effector and memory T-cell differentiation
Nature Communications (2015)
-
(S)-Garner aldehyde derived Baylis-Hillman adduct: A potential substrate for the synthesis of D-lyxo phytosphinosine analogue
Journal of Chemical Sciences (2014)
-
Influence of time and number of antigen encounters on memory CD8 T cell development
Immunologic Research (2014)