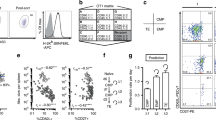
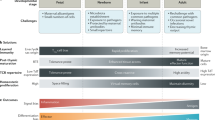
Abstract
Two potential outcomes confront proliferating antigen-stimulated naive T cells: differentiation to effector and memory cells, or deletion. How stimulation affects cell fate is unclear. Autonomous CD8+ T cell differentiation has been proposed, but this does not explain the abortive proliferation of T cells induced by immature dendritic cells. Here we show that human and mouse CD4+ and CD8+ T cells receiving short or weak stimulation of the T cell receptor proliferate in response to interleukin 2 (IL-2) but are not 'fit' because they die by neglect, fail to proliferate in response to IL-7 and IL-15 and disappear in vivo. Conversely, prolonged or strong stimulation promotes 'fitness' by enhancing survival and cytokine responsiveness. Our results are consistent with the concept that signal strength drives progressive T cell differentiation and the acquisition of fitness.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Kurts, C., Kosaka, H., Carbone, F.R., Miller, J.F. & Heath, W.R. Class I–restricted cross-presentation of exogenous self-antigens leads to deletion of autoreactive CD8+ T cells. J. Exp. Med. 186, 239–245 (1997).
Heath, W.R. & Carbone, F.R. Cross-presentation in viral immunity and self-tolerance. Nat. Rev. Immunol. 1, 126–134 (2001).
Hernandez, J., Aung, S., Redmond, W.L. & Sherman, L.A. Phenotypic and functional analysis of CD8+ T cells undergoing peripheral deletion in response to cross-presentation of self-antigen. J. Exp. Med. 194, 707–717 (2001).
Hawiger, D. et al. Dendritic cells induce peripheral T cell unresponsiveness under steady state conditions in vivo. J. Exp. Med. 194, 769–779 (2001).
Lauvau, G. et al. Priming of memory but not effector CD8 T cells by a killed bacterial vaccine. Science 294, 1735–1739 (2001).
Iezzi, G., Karjalainen, K. & Lanzavecchia, A. The duration of antigenic stimulation determines the fate of naive and effector T cells. Immunity 8, 89–95 (1998).
Iezzi, G., Scotet, E., Scheidegger, D. & Lanzavecchia, A. The interplay between the duration of TCR and cytokine signalling determines T cell polarization. Eur. J. Immunol. 29, 4092–4101 (1999).
Sallusto, F., Lenig, D., Forster, R., Lipp, M. & Lanzavecchia, A. Two subsets of memory T lymphocytes with distinct homing potentials and effector functions. Nature 401, 708–712 (1999).
Reinhardt, R.L., Khoruts, A., Merica, R., Zell, T. & Jenkins, M.K. Visualizing the generation of memory CD4 T cells in the whole body. Nature 410, 101–105 (2001).
Langenkamp, A. et al. T cell priming by dendritic cells: thresholds for proliferation, differentiation and death and intraclonal functional diversification. Eur. J. Immunol. 32, 2046–2054 (2002).
Lanzavecchia, A. & Sallusto, F. Dynamics of T lymphocyte responses: intermediates, effectors and memory cells. Science 290, 92–97 (2000).
Valitutti, S., Muller, S., Cella, M., Padovan, E. & Lanzavecchia, A. Serial triggering of many T-cell receptors by a few peptide–MHC complexes. Nature 375, 148–151 (1995).
Viola, A. & Lanzavecchia, A. T cell activation determined by T cell receptor number and tunable thresholds. Science 273, 104–106 (1996).
Wulfing, C. et al. Kinetics and extent of T cell activation as measured with the calcium signal. J. Exp. Med. 185, 1815–1825 (1997).
Viola, A., Schroeder, S., Sakakibara, Y. & Lanzavecchia, A. T lymphocyte costimulation mediated by reorganization of membrane microdomains. Science 283, 680–682 (1999).
Tuosto, L. & Acuto, O. CD28 affects the earliest signaling events generated by TCR engagement. Eur. J. Immunol. 28, 2131–2142 (1998).
Fraser, J.D., Irving, B.A., Crabtree, G.R. & Weiss, A. Regulation of interleukin-2 gene enhancer activity by the T cell accessory molecule CD28. Science 251, 313–316 (1991).
van Stipdonk, M.J., Lemmens, E.E. & Schoenberger, S.P. Naive CTLs require a single brief period of antigenic stimulation for clonal expansion and differentiation. Nat. Immunol. 2, 423–429 (2001).
Kaech, S.M. & Ahmed, R. Memory CD8+ T cell differentiation: initial antigen encounter triggers a developmental program in naive cells. Nat. Immunol. 2, 415–422 (2001).
Plas, D.R., Rathmell, J.C. & Thompson, C.B. Homeostatic control of lymphocyte survival: potential origins and implications. Nat. Immunol. 3, 515–521 (2002).
Goldrath, A.W. et al. Cytokine requirements for acute and basal homeostatic proliferation of naive and memory CD8+ T cells. J. Exp. Med. 195, 1515–1522 (2002).
Becker, T.C. et al. Interleukin 15 is required for proliferative renewal of virus-specific memory CD8 T cells. J. Exp. Med. 195, 1541–1548 (2002).
Schluns, K.S., Kieper, W.C., Jameson, S.C. & Lefrancois, L. Interleukin-7 mediates the homeostasis of naive and memory CD8 T cells in vivo. Nat. Immunol. 1, 426–432 (2000).
Zhang, X., Sun, S., Hwang, I., Tough, D.F. & Sprent, J. Potent and selective stimulation of memory-phenotype CD8+ T cells in vivo by IL-15. Immunity 8, 591–599 (1998).
Ku, C.C., Murakami, M., Sakamoto, A., Kappler, J. & Marrack, P. Control of homeostasis of CD8+ memory T cells by opposing cytokines. Science 288, 675–678 (2000).
Vander Heiden, M.G. & Thompson, C.B. Bcl-2 proteins: regulators of apoptosis or of mitochondrial homeostasis? Nat. Cell Biol. 1, E209–E216 (1999).
Newton, K. & Strasser, A. Cell death control in lymphocytes. Adv. Immunol. 76, 179–226 (2000).
Broome, H.E., Dargan, C.M., Krajewski, S. & Reed, J.C. Expression of Bcl-2, Bcl-x, and Bax after T cell activation and IL-2 withdrawal. J. Immunol. 155, 2311–2317 (1995).
Gett, A.V. & Hodgkin, P.D. Cell division regulates the T cell cytokine repertoire, revealing a mechanism underlying immune class regulation. Proc. Natl. Acad. Sci. USA 95, 9488–9493 (1998).
Bird, J.J. et al. Helper T cell differentiation is controlled by the cell cycle. Immunity 9, 229–237 (1998).
Tanchot, C. et al. Modifications of CD8+ T cell function during in vivo memory or tolerance induction. Immunity 8, 581–590 (1998).
Schwartz, R.H. T cell clonal anergy. Curr. Opin. Immunol. 9, 351–357 (1997).
Wells, A.D., Walsh, M.C., Sankaran, D. & Turka, L.A. T cell effector function and anergy avoidance are quantitatively linked to cell division. J. Immunol. 165, 2432–2443 (2000).
Valitutti, S., Dessing, M., Aktories, K., Gallati, H. & Lanzavecchia, A. Sustained signaling leading to T cell activation results from prolonged T cell receptor occupancy. Role of T cell actin cytoskeleton. J. Exp. Med. 181, 577–584 (1995).
Sugamura, K. et al. The interleukin-2 receptor γ chain: its role in the multiple cytokine receptor complexes and T cell development in XSCID. Annu. Rev. Immunol. 14, 179–205 (1996).
Vander Heiden, M.G., Chandel, N.S., Williamson, E.K., Schumacker, P.T. & Thompson, C.B. Bcl-xL regulates the membrane potential and volume homeostasis of mitochondria. Cell 91, 627–637 (1997).
Boise, L.H. et al. CD28 costimulation can promote T cell survival by enhancing the expression of Bcl-xL . Immunity 3, 87–98 (1995).
Hogquist, K.A. et al. T cell receptor antagonist peptides induce positive selection. Cell 76, 17–27 (1994).
Weber, S., Traunecker, A., Oliveri, F., Gerhard, W. & Karjalainen, K. Specific low-affinity recognition of major histocompatibility complex plus peptide by soluble T-cell receptor. Nature 356, 793–796 (1992).
Weninger, W., Crowley, M.A., Manjunath, N. & von Andrian, U.H. Migratory properties of naive, effector, and memory CD8+ T cells. J. Exp. Med. 194, 953–966 (2001).
Iezzi, G., Scheidegger, D. & Lanzavecchia, A. Migration and function of antigen primed nonpolarized T lymphocytes in vivo. J. Exp. Med. 193, 987–993 (2001).
Potsch, C., Vohringer, D. & Pircher, H. Distinct migration patterns of naive and effector CD8 T cells in the spleen: correlation with CCR7 receptor expression and chemokine reactivity. Eur. J. Immunol. 29, 3562–3570 (1999).
Masopust, D., Vezys, V., Marzo, A.L. & Lefrancois, L. Preferential localization of effector memory cells in nonlymphoid tissue. Science 291, 2413–2417 (2001).
Cantrell, D.A. & Smith, K.A. Transient expression of interleukin 2 receptors. Consequences for T cell growth. J. Exp. Med. 158, 1895–1911 (1983).
Geginat, J., Sallusto, F. & Lanzavecchia, A. Cytokine-driven proliferation and differentiation of human naive, central memory, and effector memory CD4+ T cells. J. Exp. Med. 194, 1711–1719 (2001).
Hernandez, J., Aung, S., Marquardt, K. & Sherman, L.A. Uncoupling of proliferative potential and gain of effector function by CD8+ T cells responding to self-antigens. J. Exp. Med. 196, 323–333 (2002).
Gorelik, L. & Flavell, R.A. Abrogation of TGFβ signaling in T cells leads to spontaneous T cell differentiation and autoimmune disease. Immunity 12, 171–181 (2000).
Lenardo, M. et al. Mature T lymphocyte apoptosis-immune regulation in a dynamic and unpredictable antigenic environment. Annu. Rev. Immunol. 17, 221–253 (1999).
Ferreira, C., Barthlott, T., Garcia, S., Zamoyska, R. & Stockinger, B. Differential survival of naive CD4 and CD8 T cells. J. Immunol. 165, 3689–3694 (2000).
Stoll, S., Delon, J., Brotz, T.M. & Germain, R.N. Dynamic imaging of T cell-dendritic cell interactions in lymph nodes. Science 296, 1873–1876 (2002).
Acknowledgements
We thank P. Dellabona for OT-I mice; K. Karjalainen for critical reading and discussion; D. Jarossay for cell sorting; and L. Perlini and G. Bosshard for technical assistance. Supported by the European Community (contract QLK-CT-201-0105), the Swiss National Science Foundation (grant 31-63885) and the Helmut Horten Foundation (to A.L.).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Gett, A., Sallusto, F., Lanzavecchia, A. et al. T cell fitness determined by signal strength. Nat Immunol 4, 355–360 (2003). https://doi.org/10.1038/ni908
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni908
This article is cited by
-
Enhancing CAR-T cell functionality in a patient-specific manner
Nature Communications (2023)
-
Generation of functionally distinct T-cell populations by altering the viscoelasticity of their extracellular matrix
Nature Biomedical Engineering (2023)
-
Malabaricone C, a constituent of spice Myristica malabarica, exhibits anti-inflammatory effects via modulation of cellular redox
Journal of Biosciences (2023)
-
The timing of differentiation and potency of CD8 effector function is set by RNA binding proteins
Nature Communications (2022)
-
Targeting FAK in anticancer combination therapies
Nature Reviews Cancer (2021)