Abstract
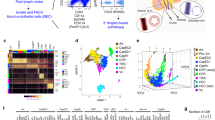
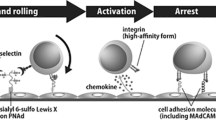
Lymphocytes are recruited from blood by high-endothelial venules (HEVs). We performed transcriptomic analyses and identified molecular signatures that distinguish HEVs from capillary endothelium and that define tissue-specific HEV specialization. Capillaries expressed gene programs for vascular development. HEV-expressed genes showed enrichment for genes encoding molecules involved in immunological defense and lymphocyte migration. We identify capillary and HEV markers and candidate mechanisms for regulated recruitment of lymphocytes, including a lymph node HEV–selective transmembrane mucin; transcriptional control of functionally specialized carbohydrate ligands for lymphocyte L-selectin; HEV expression of molecules for transendothelial migration; and metabolic programs for lipid mediators of lymphocyte motility and chemotaxis. We also elucidate a carbohydrate-recognition pathway that targets B cells to intestinal lymphoid tissues, defining CD22 as a lectin-homing receptor for mucosal HEVs.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Change history
27 October 2014
In the version of this article initially published, Hiroto Kawashima was omitted as an author. The correct author list is as follows: Mike Lee1, Helena Kiefel1, Melissa D LaJevic1, Matthew S Macauley2, Hiroto Kawashima3, Edward O'Hara4, Junliang Pan4, James C Paulson2 & Eugene C Butcher1,4,5. The affiliation for this author is as follows: Department of Biochemistry, School of Pharmacy and Pharmaceutical Sciences, Hoshi University, Tokyo, Japan. The Author Contributions section should include "H.K. provided advice and the S2 hybridoma" (and the corresponding first thanks in Acknowledgments should be removed). The error has been corrected in the HTML and PDF versions of the article.
References
Gauguet, J.M., Bonasio, R. & von Andrian, U.H. in Endothelial Biomedicine (ed. Aird, W.C.) Ch. 170 (Cambridge University Press, New York, 2007).
Anderson, N.D., Anderson, A.O. & Wyllie, R.G. Specialized structure and metabolic activities of high endothelial venules in rat lymphatic tissues. Immunology 31, 455–473 (1976).
Girard, J.P., Moussion, C. & Forster, R. HEVs, lymphatics and homeostatic immune cell trafficking in lymph nodes. Nat. Rev. Immunol. 12, 762–773 (2012).
Butcher, E.C. & Picker, L.J. Lymphocyte homing and homeostasis. Science 272, 60–66 (1996).
Malhotra, D. et al. Transcriptional profiling of stroma from inflamed and resting lymph nodes defines immunological hallmarks. Nat. Immunol. 13, 499–510 (2012).
Kruse, A., Hallmann, R. & Butcher, E.C. Specialized patterns of vascular differentiation antigens in the pregnant mouse uterus and the placenta. Biol. Reprod. 61, 1393–1401 (1999).
Doyle, E.L. et al. CD63 is an essential cofactor to leukocyte recruitment by endothelial P-selectin. Blood 118, 4265–4273 (2011).
Jutila, M.A. et al. Ly-6C is a monocyte/macrophage and endothelial cell differentiation antigen regulated by interferon-γ. Eur. J. Immunol. 18, 1819–1826 (1988).
Zhang, G. et al. Arterial-venous endothelial cell fate is related to vascular endothelial growth factor and Notch status during human bone mesenchymal stem cell differentiation. FEBS Lett. 582, 2957–2964 (2008).
Kitagawa, M. et al. Hes1 and Hes5 regulate vascular remodeling and arterial specification of endothelial cells in brain vascular development. Mech. Dev. 130, 458–466 (2013).
Brütsch, R. et al. Integrin cytoplasmic domain-associated protein-1 attenuates sprouting angiogenesis. Circ. Res. 107, 592–601 (2010).
Verkman, A.S. Aquaporin water channels and endothelial cell function. J. Anat. 200, 617–627 (2002).
Miyasaka, M. & Tanaka, T. Lymphocyte trafficking across high endothelial venules: dogmas and enigmas. Nat. Rev. Immunol. 4, 360–370 (2004).
Demyanets, S., Huber, K. & Wojta, J. Vascular effects of glycoprotein130 ligands–—part I: pathophysiological role. Vascul. Pharmacol. 56, 34–46 (2012).
Sarrazin, S., Lamanna, W.C. & Esko, J.D. Heparan sulfate proteoglycans. Cold Spring Harb. Perspect. Biol. 3, a004952 (2011).
Campbell, J.J. et al. Chemokines and the arrest of lymphocytes rolling under flow conditions. Science 279, 381–384 (1998).
Griffith, J.W., Sokol, C.L. & Luster, A.D. Chemokines and chemokine receptors: positioning cells for host defense and immunity. Annu. Rev. Immunol. 32, 659–702 (2014).
Zlotnik, A. & Yoshie, O. The chemokine superfamily revisited. Immunity 36, 705–716 (2012).
Kiefer, F. & Siekmann, A.F. The role of chemokines and their receptors in angiogenesis. Cell. Mol. Life Sci. 68, 2811–2830 (2011).
Ahn, S.Y. et al. Tumor necrosis factor-α induces fractalkine expression preferentially in arterial endothelial cells and mithramycin A suppresses TNF-α-induced fractalkine expression. Am. J. Pathol. 164, 1663–1672 (2004).
Ryu, J. et al. Activation of fractalkine/CX3CR1 by vascular endothelial cells induces angiogenesis through VEGF-A/KDR and reverses hindlimb ischaemia. Cardiovasc. Res. 78, 333–340 (2008).
Patnode, M.L. et al. KSGal6ST generates galactose-6-O-sulfate in high endothelial venules but does not contribute to L-selectin-dependent lymphocyte homing. Glycobiology 23, 381–394 (2013).
Strilic´, B. et al. The molecular basis of vascular lumen formation in the developing mouse aorta. Dev. Cell 17, 505–515 (2009).
Umemoto, E. et al. Novel regulators of lymphocyte trafficking across high endothelial venules. Crit. Rev. Immunol. 31, 147–169 (2011).
Charfi, C., Levros, L.C. Jr., Edouard, E. & Rassart, E. Characterization and identification of PARM-1 as a new potential oncogene. Mol. Cancer 12, 84–99 (2013).
Karikoski, M. et al. Clever-1/Stabilin-1 regulates lymphocyte migration within lymphatics and leukocyte entrance to sites of inflammation. Eur. J. Immunol. 39, 3477–3487 (2009).
Jalkanen, S. & Salmi, M. VAP-1 and CD73, endothelial cell surface enzymes in leukocyte extravasation. Arterioscler. Thromb. Vasc. Biol. 28, 18–26 (2008).
Bai, Z. et al. Constitutive lymphocyte transmigration across the basal lamina of high endothelial venules is regulated by the autotaxin/lysophosphatidic acid axis. J. Immunol. 190, 2036–2048 (2013).
Halin, C. et al. The S1P-analog FTY720 differentially modulates T-cell homing via HEV: T-cell-expressed S1P1 amplifies integrin activation in peripheral lymph nodes but not in Peyer patches. Blood 106, 1314–1322 (2005).
Preuss, I. et al. Transcriptional regulation and functional characterization of the oxysterol/EBI2 system in primary human macrophages. Biochem. Biophys. Res. Commun. 446, 663–668 (2014).
Spann, N.J. & Glass, C.K. Sterols and oxysterols in immune cell function. Nat. Immunol. 14, 893–900 (2013).
Wang, C.C. et al. Homeodomain factor Nkx2–3 controls regional expression of leukocyte homing coreceptor MAdCAM-1 in specialized endothelial cells of the viscera. Dev. Biol. 224, 152–167 (2000).
Ortolan, E. et al. CD157 plays a pivotal role in neutrophil transendothelial migration. Blood 108, 4214–4222 (2006).
Liu, X., Strable, M.S. & Ntambi, J.M. Stearoyl CoA desaturase 1: role in cellular inflammation and stress. Adv. Nutrition 2, 15–22 (2011).
Waller-Evans, H. et al. The orphan adhesion-GPCR GPR126 is required for embryonic development in the mouse. PLoS ONE 5, e14047 (2010).
Sperandio, M., Gleissner, C.A. & Ley, K. Glycosylation in immune cell trafficking. Immunol. Rev. 230, 97–113 (2009).
Uchimura, K. et al. A major class of L-selectin ligands is eliminated in mice deficient in two sulfotransferases expressed in high endothelial venules. Nat. Immunol. 6, 1105–1113 (2005).
Crocker, P.R., Paulson, J.C. & Varki, A. Siglecs and their roles in the immune system. Nat. Rev. Immunol. 7, 255–266 (2007).
Vincourt, J.B., Jullien, D., Amalric, F. & Girard, J.P. Molecular and functional characterization of SLC26A11, a sodium-independent sulfate transporter from high endothelial venules. FASEB J. 17, 890–892 (2003).
Andrews, P., Milsom, D.W. & Ford, W.L. Migration of lymphocytes across specialized vascular endothelium. V. Production of a sulphated macromolecule by high endothelial cells in lymph nodes. J. Cell Sci. 57, 277–292 (1982).
Angata, K. & Fukuda, M. Polysialyltransferases: major players in polysialic acid synthesis on the neural cell adhesion molecule. Biochimie 85, 195–206 (2003).
Hirakawa, J. et al. Novel anti-carbohydrate antibodies reveal the cooperative function of sulfated N- and O-glycans in lymphocyte homing. J. Biol. Chem. 285, 40864–40878 (2010).
Kannagi, R. et al. in The Molecular Immunology of Complex Carbohydrates (ed. Wu, A.M.) Ch. 29 (Springer Science+Business Media, New York, 2011).
Harduin-Lepers, A. et al. The human sialyltransferase family. Biochimie 83, 727–737 (2001).
Paulson, J.C., Prieels, J.P., Glasgow, L.R. & Hill, R.L. Sialyl- and fucosyltransferases in the biosynthesis of asparaginyl-linked oligosaccharides in glycoproteins. Mutually exclusive glycosylation by β-galactoside alpha2 goes to 6 sialyltransferase and N-acetylglucosaminide α1 goes to 3 fucosyltransferase. J. Biol. Chem. 253, 5617–5624 (1978).
Marth, J.D. & Grewal, P.K. Mammalian glycosylation in immunity. Nat. Rev. Immunol. 8, 874–887 (2008).
Naito, Y. et al. Germinal center marker GL7 probes activation-dependent repression of N-glycolylneuraminic acid, a sialic acid species involved in the negative modulation of B-cell activation. Mol. Cell. Biol. 27, 3008–3022 (2007).
Stevens, S.K., Weissman, I.L. & Butcher, E.C. Differences in the migration of B and T lymphocytes: organ-selective localization in vivo and the role of lymphocyte-endothelial cell recognition. J. Immunol. 128, 844–851 (1982).
Bargatze, R.F., Jutila, M.A. & Butcher, E.C. Distinct roles of L-selectin and integrins alpha 4 beta 7 and LFA-1 in lymphocyte homing to Peyer's patch-HEV in situ: the multistep model confirmed and refined. Immunity 3, 99–108 (1995).
Nitschke, L., Floyd, H., Ferguson, D.J. & Crocker, P.R. Identification of CD22 ligands on bone marrow sinusoidal endothelium implicated in CD22-dependent homing of recirculating B cells. J. Exp. Med. 189, 1513–1518 (1999).
Hanasaki, K., Varki, A. & Powell, L.D. CD22-mediated cell adhesion to cytokine-activated human endothelial cells. Positive and negative regulation by α2–6-sialylation of cellular glycoproteins. J. Biol. Chem. 270, 7533–7542 (1995).
Limaye, V., Vadas, M.A., Pitson, S.M. & Gamble, J.R. The effects of markedly raised intracellular sphingosine kinase-1 activity in endothelial cells. Cell. Mol. Biol. Lett. 14, 411–423 (2009).
McDonald, J.G. & Russell, D.W. 25-Hydroxycholesterol: a new life in immunology. J. Leukoc. Biol. 88, 1071–1072 (2010).
Yi, T. et al. Oxysterol gradient generation by lymphoid stromal cells guides activated B cell movement during humoral responses. Immunity 37, 535–548 (2012).
Fre, S. et al. Notch lineages and activity in intestinal stem cells determined by a new set of knock-in mice. PLoS ONE 6, e25785 (2011).
Nitschke, L., Carsetti, R., Ocker, B., Kohler, G. & Lamers, M.C. CD22 is a negative regulator of B-cell receptor signalling. Curr. Biol. 7, 133–143 (1997).
Hennet, T., Chui, D., Paulson, J.C. & Marth, J.D. Immune regulation by the ST6Gal sialyltransferase. Proc. Natl. Acad. Sci. USA 95, 4504–4509 (1998).
Shen, Z., Lu, Z., Chhatbar, P.Y., O'Herron, P. & Kara, P. An artery-specific fluorescent dye for studying neurovascular coupling. Nat. Methods 9, 273–276 (2012).
Suzuki, R. & Shimodaira, H. Pvclust: an R package for assessing the uncertainty in hierarchical clustering. Bioinformatics 22, 1540–1542 (2006).
Acknowledgements
We thank J. Sweere, A. Scholz, C. Czupalla and B. Arlian for help with experiments; J. Jang for antibody production; L. Rott for assistance with cell sorting; all members of the Butcher laboratory for discussions; B. Yoo (Stanford University) and T.A. Rando (Stanford University) for tissues from Hes1-EmGFPSAT mice; M. Salmi and M. Miyasaka for critical review of the manuscript; and the UniProt Consortium, the Kyoto Encyclopedia of Genes and Genomes, Enrichr software (E.Y. Chen, C.M. Tan, Y. Kou, Q. Duan, Z. Wang, G.V. Meirelles, N.R. Clark and Ma'ayan A. of the Icahn School of Medicine at Mount Sinai), iHOP (Information Hyperlinked over Proteins) online gene-guided access to PubMed abstracts, and the Immunological Genome Project for their informatics tools and compendia of data. Supported by the US National Institutes of Health (R37 GM37734, R37 AI047822, R01 AI093981 and R01 DK084647 to E.C.B.; 5T32AI007290 to M.L.; 5T32CA009151, 5T32AI007290 and F32CA180415 to M.D.L.; and R01 AI50143 to J.C.P.), the US Department of Veterans Affairs (E.C.B.), the Klaus Bensch Professorship (E.C.B.), Deutsche Forschungsgemeinschaft (KI1646/1-2 to H.Ki.), the Stanford Institute for Immunity, Transplantation and Infection (H.Ki.), the American Heart Association (H.Ki.) and the Crohn's and Colitis Foundation of America (Ref. 3782 to M.L.).
Author information
Authors and Affiliations
Contributions
M.L. developed HEC-isolation protocols and designed and performed experiments, including flow cytometry of ECs, whole-genome expression analyses, immunohistology and homing studies; H.Ki. performed flow cytometry and immunofluorescence staining; M.D.L. performed immunoprecipitation and immunoblot analysis studies; M.S.M. did homing studies; H.Ka. provided advice and the S2 hybridoma; M.S.M. and J.C.P. contributed to writing of the section on HEV glycans; E.O. established the parameters for the isolation of RNA from HECs; J.P. provided intellectual input; and E.C.B. designed and guided the study, analyzed data and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
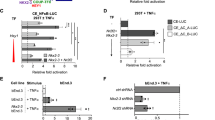
Supplementary Figure 2 PARM-1 is decorated with PNAd in mouse lymph nodes.
Representative immunoblot (WB) of Parm1 immunoprecipitate (IP) from mouse lymph node membrane enriched protein probed for Parm1 (green) and MECA-79 (red). * indicates two isoforms of Parm1 proteins, of which the higher molecular weight band is also detected by MECA-79, but in parallel experiments did not bind control rat IgM (not shown). Control IP is IP using anti-Hsp90 antibody, a monoclonal rabbit IgG. Numbers on the left indicate the position of molecular weight standard proteins of the indicated kDa. Data are representative of 3 independent experiments with axillary, brachial and in some instances mesenteric lymph nodes pooled from 2-3 mice in each case.
Supplementary Figure 3 Gating strategy for the isolation of PLN, MLN and PP HECs and CAP ECs by flow cytometry.
FACS plots (both pre-sort and post-sort) showing the gating strategy (successive gating starts from the lineage gating on the left to the CD31 versus gp38 gating in the middle to the HEC vs CAP gating on the right) for BEC subset isolation after standard forward/side scatter and live/dead gating. Scatter gating was designed to include only live cells. A strict forward angle gate was used to exclude doublets. Numbers in blue font represent the percent of cells within the gate.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–3 (PDF 349 kb)
Supplementary Table 1
Supplementary Table 1 (XLSX 776 kb)
Supplementary Table 2
Supplementary Table 2 (XLSX 145 kb)
Rights and permissions
About this article
Cite this article
Lee, M., Kiefel, H., LaJevic, M. et al. Transcriptional programs of lymphoid tissue capillary and high endothelium reveal control mechanisms for lymphocyte homing. Nat Immunol 15, 982–995 (2014). https://doi.org/10.1038/ni.2983
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.2983
This article is cited by
-
Enhancement of solvent tolerance and economical bioethanol production by modulated sigma 54 expression of Pseudomonas putida
Biomass Conversion and Biorefinery (2024)
-
Pathological angiogenesis: mechanisms and therapeutic strategies
Angiogenesis (2023)
-
Reprogramming of sentinel lymph node microenvironment during tumor metastasis
Journal of Biomedical Science (2022)
-
The crosstalk between intestinal bacterial microbiota and immune cells in colorectal cancer progression
Clinical and Translational Oncology (2022)
-
An NKX-COUP-TFII morphogenetic code directs mucosal endothelial addressin expression
Nature Communications (2022)