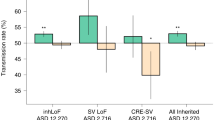
Abstract
Autism spectrum disorder (ASD) risk is influenced by common polygenic and de novo variation. We aimed to clarify the influence of polygenic risk for ASD and to identify subgroups of ASD cases, including those with strongly acting de novo variants, in which polygenic risk is relevant. Using a novel approach called the polygenic transmission disequilibrium test and data from 6,454 families with a child with ASD, we show that polygenic risk for ASD, schizophrenia, and greater educational attainment is over-transmitted to children with ASD. These findings hold independent of proband IQ. We find that polygenic variation contributes additively to risk in ASD cases who carry a strongly acting de novo variant. Lastly, we show that elements of polygenic risk are independent and differ in their relationship with phenotype. These results confirm that the genetic influences on ASD are additive and suggest that they create risk through at least partially distinct etiologic pathways.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Sanders, S.J. et al. Insights into autism spectrum disorder genomic architecture and biology from 71 risk loci. Neuron 87, 1215–1233 (2015).
Gaugler, T. et al. Most genetic risk for autism resides with common variation. Nat. Genet. 46, 881–885 (2014).
De Rubeis, S. et al. Synaptic, transcriptional and chromatin genes disrupted in autism. Nature 515, 209–215 (2014).
Iossifov, I. et al. The contribution of de novo coding mutations to autism spectrum disorder. Nature 515, 216–221 (2014).
Bulik-Sullivan, B.K. et al. LD Score regression distinguishes confounding from polygenicity in genome-wide association studies. Nat. Genet. 47, 291–295 (2015).
Bulik-Sullivan, B. et al. An atlas of genetic correlations across human diseases and traits. Nat. Genet. 47, 1236–1241 (2015).
Krumm, N. et al. Excess of rare, inherited truncating mutations in autism. Nat. Genet. 47, 582–588 (2015).
Anney, R. et al. Individual common variants exert weak effects on the risk for autism spectrum disorders. Hum. Mol. Genet. 21, 4781–4792 (2012).
Klei, L. et al. Common genetic variants, acting additively, are a major source of risk for autism. Mol. Autism 3, 9 (2012).
World Health Organization. WHO Motor Development Study: windows of achievement for six gross motor development milestones. Acta Paediatr. 450, 86–95 (2006).
Deciphering Developmental Disorders Study. Large-scale discovery of novel genetic causes of developmental disorders. Nature 519, 223–228 (2015).
Okbay, A. et al. Genome-wide association study identifies 74 loci associated with educational attainment. Nature 533, 539–542 (2016).
Clarke, T.K. et al. Common polygenic risk for autism spectrum disorder (ASD) is associated with cognitive ability in the general population. Mol. Psychiatry 21, 419–425 (2016).
Hagenaars, S.P. et al. Shared genetic aetiology between cognitive functions and physical and mental health in UK Biobank (N=112 151) and 24 GWAS consortia. Mol. Psychiatry 21, 1624–1632 (2016).
Robinson, E.B. et al. Autism spectrum disorder severity reflects the average contribution of de novo and familial influences. Proc. Natl. Acad. Sci. USA 111, 15161–15165 (2014).
Munafo, M.R., Tilling, K., Taylor, A.E., Evans, D.M. & Davey Smith, G. Collider Scope: how selection bias can induce spurious associations. Preprint at bioRxiv http://dx.doi.org/10.1101/079707 (2016).
Spielman, R.S., McGinnis, R.E. & Ewens, W.J. Transmission test for linkage disequilibrium: the insulin gene region and insulin-dependent diabetes mellitus (IDDM). Am. J. Hum. Genet. 52, 506–516 (1993).
Fischbach, G.D. & Lord, C. The Simons Simplex Collection: a resource for identification of autism genetic risk factors. Neuron 68, 192–195 (2010).
Schizophrenia Working Group of the Psychiatric Genomics Consortium. Biological insights from 108 schizophrenia-associated genetic loci. Nature 511, 421–427 (2014).
Cross-Disorder Group of the Psychiatric Genomic Consortium. Genetic relationship between five psychiatric disorders estimated from genome-wide SNPs. Nat. Genet. 45, 984–994 (2013).
Kosmicki, J.A. et al. Refining the role of de novo protein-truncating variants in neurodevelopmental disorders by using population reference samples. Nat. Genet. 49, 504–510 (2017).
Lek, M. et al. Analysis of protein-coding genetic variation in 60,706 humans. Nature 536, 285–291 (2016).
Samocha, K.E. et al. A framework for the interpretation of de novo mutation in human disease. Nat. Genet. 46, 944–950 (2014).
Robinson, E.B. et al. Genetic risk for autism spectrum disorders and neuropsychiatric variation in the general population. Nat. Genet. 48, 552–555 (2016).
1000 Genomes Project Consortium. An integrated map of genetic variation from 1,092 human genomes. Nature 491, 56–65 (2012).
International HapMap 3 Consortium. Integrating common and rare genetic variation in diverse human populations. Nature 467, 52–58 (2010).
Yang, J., Lee, S.H., Goddard, M.E. & Visscher, P.M. GCTA: a tool for genome-wide complex trait analysis. Am. J. Hum. Genet. 88, 76–82 (2011).
Wray, N.R., Goddard, M.E. & Visscher, P.M. Prediction of individual genetic risk to disease from genome-wide association studies. Genome Res. 17, 1520–1528 (2007).
Locke, A.E. et al. Genetic studies of body mass index yield new insights for obesity biology. Nature 518, 197–206 (2015).
Chang, C.C. et al. Second-generation PLINK: rising to the challenge of larger and richer datasets. Gigascience 4, 7 (2015).
Elliott, C. Differential Ability Scales (The Psychological Corporation, 2007).
Mullen, E. Mullen Scales of Early Learning (American Guidance Service, 1995).
Wechsler, D. Wechsler Abbreviated Scale of Intelligence (Psychological Corporation, 1999).
Chaste, P. et al. A genome-wide association study of autism using the Simons Simplex Collection: does reducing phenotypic heterogeneity in autism increase genetic homogeneity? Biol. Psychiatry 77, 775–784 (2015).
Provost, B., Lopez, B.R. & Heimerl, S. A comparison of motor delays in young children: autism spectrum disorder, developmental delay, and developmental concerns. J. Autism Dev. Disord. 37, 321–328 (2007).
Acknowledgements
We thank S. Hyman and R. Hosking for their thoughtful comments. We also thank A. Pai for his help in the development of the pTDT analytic software. E.B.R. and D.J.W. were funded by National Institute of Mental Health grant 1K01MH099286-01A1 and Brain Behavior Research Foundation (NARSAD) Young Investigator grant 22379. E.M.W. was funded by the Stanley Center for Psychiatric Research at the Broad Institute. A.O. was funded by an ERC Consolidator Grant (647648 EdGe). We thank the families who took part in the Simons Simplex Collection study and the clinicians who collected data at each of the study sites. The iPSYCH project is funded by the Lundbeck Foundation and the universities and university hospitals of Aarhus and Copenhagen. Genotyping of iPSYCH and PGC samples was supported by grants from the Stanley Foundation, the Simons Foundation (SFARI 311789 to M.J.D.), and the National Institute of Mental Health (5U01MH094432-02 to M.J.D.). This work was also supported by a grant from the Simons Foundation (SFARI 402281 to S.J.S.). The authors would like to thank the Exome Aggregation Consortium and the groups that provided exome variant data for comparison. A full list of contributing groups can be found on the ExAC website (see URLs).
Author information
Authors and Affiliations
Consortia
Contributions
D.J.W., E.M.W., S.R., R.K.W., J.A.K., J.G., K.E.S., J.I.G., A.O., J.B.-G., T.W., D.M.H., R.A., and S.J.S. generated data and/or conducted analyses. E.B.R., M.J.D., D.J.W., S.B., and G.D.S. designed the experiment and tools. D.S., B.D., and J.T. aided in interpretation of the data. E.B.R., M.J.D., P.B.M., and A.D.B. supervised the research. E.B.R., D.J.W., and E.M.W. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
A list of members and affiliations appears at the end of the paper.
A list of members and affiliations appears at the end of the paper.
Integrated supplementary information
Supplementary Figure 1 Illustrative example of pTDT using height
The expected value of a child’s polygenic risk score (PRS) for a trait is the average of maternal and paternal PRS values. For example, if a mother’s PRS is A, the expected PRS of an egg, which contains half of the maternal genetic material, is A/2. If the father’s PRS is B, the expected PRS of a sperm is B/2. The expected value of the child’s PRS is then (A + B)/2. (a) In a randomly selected cohort of parent–child trios, the average of the children’s PRSs for height, in light blue, is expected to equal the average of the mid-parent PRS for height, in dark blue; for each pair of parents, the mid-parent PRS is calculated by averaging the maternal and paternal PRSs; the variance of the mid-parent PRS is reduced because it is the average of the maternal and paternal values. (b) In a cohort of trios phenotypically selected for very high height in the offspring (offspring who are taller than expected based on the height of the parents), we expect offspring PRS to exceed mid-parent PRS. The difference between the mean of the offspring PRS distribution and mid-parent PRS distribution, n, we refer to as polygenic transmission disequilibrium.
Supplementary Figure 2 ASD probands of European ancestry over-inherit ASD-associated polygenic risk.
We performed pTDT after restricting the cohort to European ancestry (Supplementary Note; n = 1,851 SSC probands, n = 3,209 PGC ASD probands, n = 5,060 SSC and PGC ASD probands combined, n = 1,509 SSC unaffected siblings). Transmission disequilibrium is shown in terms of standard deviations on mid-parent distribution ± 1.96 standard error (95% confidence interval). P values denote the probability that the mean of the pTDT deviation distribution is 0 (two-sided, one-sample t test).
Supplementary Figure 3 Polygenic risk for schizophrenia stratifies by ancestry.
See the Supplementary Note for discussion of ancestral stratification of schizophrenia polygenic risk score.
Supplementary Figure 4 Large de novo deletions and de novo deletions in constrained genes were associated with ASD case status.
Constrained genes are intolerant of heterozygous loss-of-function variations (probability of being loss-of-function intolerant (pLI) ≥ 0.9); P values are from Fisher’s exact test and estimate the probability with which the variant type is equally likely to be seen in cases (n = 2,587 subjects) and controls (n = 2,100 subjects); error bars are ±1 standard error.
Supplementary Figure 5 Unconstrained de novo deletions were not associated with ASD case status.
Contributing deletions are deletions in either category in Supplementary Figure 4 (constrained or ≥500 kb and unconstrained); error bars are ±1 standard error; P values are from Fisher’s exact tests and estimate the probability that the variant type is equally likely to be seen in cases (n = 2,587 subjects) and controls (n = 2,100 subjects).
Supplementary Figure 6 Ancestry of Simons Simplex Collection probands
Included/excluded denotes whether the first two proband principal components of ancestry were within the study-defined bounds of European ancestry; HapMap population CEU, individuals of Northern and Western European ancestry residing in Utah, USA; HapMap population TSI, Tuscans in Italy; non-European, all HapMap cohorts excluding CEU and TSI; see the Online Methods for more information.
Supplementary Figure 7 Ancestry of parents of ASD probands in the Psychiatric Genomics Consortium Autism Group.
Included/excluded denotes whether the first two parent principal components of ancestry were within the study-defined bounds of European ancestry; families were marked as European ancestry if both parents were marked as included; HapMap population CEU, individuals of Northern and Western European ancestry residing in Utah, USA; HapMap population TSI, Tuscans in Italy; non-European, all HapMap cohorts excluding CEU and TSI; see the Online Methods for more information.
Supplementary Figure 8 Association between constrained PTV rate and proband IQ in SSC.
The red line denotes the linear relationship between contributing PTVs (Online Methods) and full-scale IQ in SSC probands (n = 2,492 subjects); the blue line denotes the linear relationship between all other PTVs and full-scale IQ in SSC probands (n = 2,492 subjects). Shaded regions denote 95% confidence intervals. The red line P value is associated with a Poisson regression predicting count of contributing de novo PTVs from proband IQ and proband sex and estimates the probability of no association between proband IQ and the rate of contributing de novo PTVs; the blue line P value is associated with a Poisson regression predicting count of non-contributing de novo PTVs from proband IQ and proband sex and estimates the probability of no association between proband IQ and the rate of non-contributing de novo PTVs. Control rate dots were calculated from n = 1,902 unaffected sibling controls.
Supplementary Figure 9 Association between contributing deletions and proband IQ in SSC.
The red line denotes the linear relationship between rate of contributing deletions (Online Methods) and full-scale IQ in SSC probands (n = 2,581 subjects); the blue line denotes the linear relationship between all other de novo deletions and full-scale IQ in SSC probands (n = 2,581 subjects). Shaded regions denote 95% confidence interval. The red line P value is associated with a Poisson regression predicting count of contributing de novo deletions from proband IQ and proband sex and estimates the probability of no association between proband IQ and the rate of contributing de novo deletions; the blue line P value is associated with a Poisson regression predicting count of non-contributing de novo deletions from proband IQ and proband sex and estimates the probability of no association between proband IQ and the rate of non-contributing de novo deletions. Control rate dots were calculated from n = 2,100 unaffected sibling controls.
Supplementary Figure 10 De novo deletions, but not duplications, in constrained genes were associated with ASD.
Rates are the fraction of CNVs that include a constrained gene. P values are from Fisher’s exact tests and estimate the probability with which case (n = 82 with duplication, n = 116 with deletion) and control (n = 27 with duplication, n = 45 with deletion) carriers are equally likely to have a deletion that includes a constrained gene.
Supplementary Figure 11 Association between CDNV rate and proband IQ in SSC.
The red line denotes the linear relationship between the rate of CDNVs (contributing de novo variants; Online Methods) and full-scale IQ in SSC probands (n = 2,340 subjects); the blue line denotes the linear relationship between all other de novo deletions and PTVs and full-scale IQ in SSC probands (n = 2,340 subjects). Shaded regions denote 95% confidence intervals. The red line P value is associated with a Poisson regression predicting count of CDNVs from proband IQ and proband sex and estimates the probability of no association between proband IQ and the rate of CDNVs; the blue line P value is associated with a Poisson regression predicting count of non-CDNV de novo deletions and PTVs from proband IQ and proband sex and estimates the probability of no association between proband IQ and the rate of non-CDNV de novo deletions and PTVs. Control rate dots were calculated from n = 1,736 unaffected sibling controls.
Supplementary Figure 12 Association between the male:female carrier ratio and de novo variant category.
P values were generated using Fisher’s exact tests and estimate the probability that there is no difference between male proband (n = 2,029) and female proband (n = 317) variant rates; see the Online Methods for variant description.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–12, Supplementary Tables 1–22 and Supplementary Note (PDF 2163 kb)
Rights and permissions
About this article
Cite this article
Weiner, D., Wigdor, E., Ripke, S. et al. Polygenic transmission disequilibrium confirms that common and rare variation act additively to create risk for autism spectrum disorders. Nat Genet 49, 978–985 (2017). https://doi.org/10.1038/ng.3863
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng.3863
This article is cited by
-
Statistical methods for assessing the effects of de novo variants on birth defects
Human Genomics (2024)
-
Pervasive alterations of intra-axonal volume and network organization in young children with a 16p11.2 deletion
Translational Psychiatry (2024)
-
Structural models of genome-wide covariance identify multiple common dimensions in autism
Nature Communications (2024)
-
A genome-wide association study of Chinese and English language phenotypes in Hong Kong Chinese children
npj Science of Learning (2024)
-
Genomic analysis of 116 autism families strengthens known risk genes and highlights promising candidates
npj Genomic Medicine (2024)