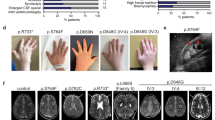
Abstract
Ciliary dysfunction leads to a broad range of overlapping phenotypes, collectively termed ciliopathies. This grouping is underscored by genetic overlap, where causal genes can also contribute modifier alleles to clinically distinct disorders. Here we show that mutations in TTC21B, which encodes the retrograde intraflagellar transport protein IFT139, cause both isolated nephronophthisis and syndromic Jeune asphyxiating thoracic dystrophy. Moreover, although resequencing of TTC21B in a large, clinically diverse ciliopathy cohort and matched controls showed a similar frequency of rare changes, in vivo and in vitro evaluations showed a significant enrichment of pathogenic alleles in cases (P < 0.003), suggesting that TTC21B contributes pathogenic alleles to ∼5% of ciliopathy cases. Our data illustrate how genetic lesions can be both causally associated with diverse ciliopathies and interact in trans with other disease-causing genes and highlight how saturated resequencing followed by functional analysis of all variants informs the genetic architecture of inherited disorders.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Accession codes
Change history
29 March 2011
In the version of this article initially published, the authors should have acknowledged that the work was also funded by a grant from the European Union (EU-SYSCILIA) to E.E.D., C.A.J., P.L.B. and N.K. The error has been corrected in the HTML and PDF versions of the article.
References
Badano, J.L., Mitsuma, N., Beales, P.L. & Katsanis, N. The ciliopathies: an emerging class of human genetic disorders. Annu. Rev. Genomics Hum. Genet. 7, 125–148 (2006).
Zaghloul, N.A. & Katsanis, N. Functional modules, mutational load and human genetic disease. Trends Genet. 26, 168–176 (2010).
Hoefele, J. et al. Evidence of oligogenic inheritance in nephronophthisis. J. Am. Soc. Nephrol. 18, 2789–2795 (2007).
Bergmann, C. et al. Loss of nephrocystin-3 function can cause embryonic lethality, Meckel-Gruber-like syndrome, situs inversus, and renal-hepatic-pancreatic dysplasia. Am. J. Hum. Genet. 82, 959–970 (2008).
Otto, E.A. et al. Hypomorphic mutations in Meckelin (MKS3/TMEM67) cause nephronophthisis with liver fibrosis (NPHP11). J. Med. Genet. 46, 663–670 (2009).
Baala, L. et al. Pleiotropic effects of CEP290 (NPHP6) mutations extend to Meckel syndrome. Am. J. Hum. Genet. 81, 170–179 (2007).
Baala, L. et al. The Meckel-Gruber syndrome gene, MKS3, is mutated in Joubert syndrome. Am. J. Hum. Genet. 80, 186–194 (2007).
Delous, M. et al. The ciliary gene RPGRIP1L is mutated in cerebello-oculo-renal syndrome (Joubert syndrome type B) and Meckel syndrome. Nat. Genet. 39, 875–881 (2007).
Gorden, N.T. et al. CC2D2A is mutated in Joubert syndrome and interacts with the ciliopathy-associated basal body protein CEP290. Am. J. Hum. Genet. 83, 559–571 (2008).
Sayer, J.A. et al. The centrosomal protein nephrocystin-6 is mutated in Joubert syndrome and activates transcription factor ATF4. Nat. Genet. 38, 674–681 (2006).
Valente, E.M. et al. Mutations in CEP290, which encodes a centrosomal protein, cause pleiotropic forms of Joubert syndrome. Nat. Genet. 38, 623–625 (2006).
Wolf, M.T. et al. Mutational analysis of the RPGRIP1L gene in patients with Joubert syndrome and nephronophthisis. Kidney Int. 72, 1520–1526 (2007).
Valente, E.M. et al. Mutations in TMEM216 perturb ciliogenesis and cause Joubert, Meckel and related syndromes. Nat. Genet. 42, 619–625 (2010).
Leitch, C.C. et al. Hypomorphic mutations in syndromic encephalocele genes are associated with Bardet-Biedl syndrome. Nat. Genet. 40, 443–448 (2008).
Khanna, H. et al. A common allele in RPGRIP1L is a modifier of retinal degeneration in ciliopathies. Nat. Genet. 41, 739–745 (2009).
Louie, C.M. et al. AHI1 is required for photoreceptor outer segment development and is a modifier for retinal degeneration in nephronophthisis. Nat. Genet. 42, 175–180 (2010).
Piperno, G. et al. Distinct mutants of retrograde intraflagellar transport (IFT) share similar morphological and molecular defects. J. Cell Biol. 143, 1591–1601 (1998).
Tran, P.V. et al. THM1 negatively modulates mouse sonic hedgehog signal transduction and affects retrograde intraflagellar transport in cilia. Nat. Genet. 40, 403–410 (2008).
Li, J.B. et al. Comparative genomics identifies a flagellar and basal body proteome that includes the BBS5 human disease gene. Cell 117, 541–552 (2004).
Liu, Q. et al. The proteome of the mouse photoreceptor sensory cilium complex. Mol. Cell. Proteomics 6, 1299–1317 (2007).
Mykytyn, K. et al. Identification of the gene that, when mutated, causes the human obesity syndrome BBS4. Nat. Genet. 28, 188–191 (2001).
Ansley, S.J. et al. Basal body dysfunction is a likely cause of pleiotropic Bardet-Biedl syndrome. Nature 425, 628–633 (2003).
Zaghloul, N.A. et al. Functional analyses of variants reveal a significant role for dominant negative and common alleles in oligogenic Bardet-Biedl syndrome. Proc. Natl. Acad. Sci. USA 107, 10602–10607 (2010).
Herron, B.J. et al. Efficient generation and mapping of recessive developmental mutations using ENU mutagenesis. Nat. Genet. 30, 185–189 (2002).
Liu, Q., Zuo, J. & Pierce, E.A. The retinitis pigmentosa 1 protein is a photoreceptor microtubule-associated protein. J. Neurosci. 24, 6427–6436 (2004).
Neuhaus, T.J., Stallmach, T., Leumann, E., Altorfer, J. & Braegger, C.P. Familial progressive tubulo-interstitial nephropathy and cholestatic liver disease—a newly recognized entity? Eur. J. Pediatr. 156, 723–726 (1997).
Beales, P.L., Elcioglu, N., Woolf, A.S., Parker, D. & Flinter, F.A. New criteria for improved diagnosis of Bardet-Biedl syndrome: results of a population survey. J. Med. Genet. 36, 437–446 (1999).
Gherman, A., Davis, E.E. & Katsanis, N. The ciliary proteome database: an integrated community resource for the genetic and functional dissection of cilia. Nat. Genet. 38, 961–962 (2006).
Badano, J.L. et al. Dissection of epistasis in oligogenic Bardet-Biedl syndrome. Nature 439, 326–330 (2006).
Matsuda, T. & Cepko, C.L. Electroporation and RNA interference in the rodent retina in vivo and in vitro. Proc. Natl. Acad. Sci. USA 101, 16–22 (2004).
Matsuda, T. & Cepko, C.L. Controlled expression of transgenes introduced by in vivo electroporation. Proc. Natl. Acad. Sci. USA 104, 1027–1032 (2007).
Acknowledgements
We are grateful to the individuals affected with ciliopathies and their families for their continued participation and enthusiasm. We thank N. Elkhartoufi for technical assistance, P. Parvex for subject recruitment, D. Parker for critical reading of the manuscript and Yorkshire Regional Genetics Service for help in obtaining control DNA samples. This research was supported by funds from the US National Institutes of Health grant R01HD04260 from the National Institute of Child Health and Development (N.K.), R01DK072301, R01DK075972 (N.K.), R01DK068306, R01DK064614, R01DK069274 (F.H.) and National Research Service Award (NRSA) fellowship F32 DK079541 (E.E.D.) from the National Institute of Diabetes, Digestive and Kidney disorders, RO1EY12910 from the National Eye Institute (E.A.P.), the Macular Vision Research Foundation (N.K.) the Foundation Fighting Blindness (N.K., R.A.L., E.A.P. and Q.L.), the F.M. Kirby Foundation (E.A.P.), the Rosanne Silbermann Foundation (E.A.P.), the Polycystic Kidney Disease (PKD) Foundation (C.B.), German Kidney Foundation (C.B.), German Research Foundation (DFG BE 3910/5-1 and SFB/TRR57; C.B.), UNADEV, Retina France, Programme Hospitalier de Recherche Clinique 2007, L'Agence nationale de la recherche 2009 (H.D.), a Medical Research Council (MRC) research training fellowship (J.H.) and the European Union (EU-SYSCILIA; E.E.D., C.A.J., P.L.B. and N.K.). This work was also supported in part by the Intramural Research Program of the National Human Genome Research Institute. R.A.L. is a Senior Scientific Investigator of Research to Prevent Blindness, New York, New York. P.L.B. is a Wellcome Trust Senior Research Fellow. F.H. is an Investigator of the Howard Hughes Medical Institute, a Doris Duke Distinguished Clinical Scientist and the Frederick G. L. Huetwell Professor. N.K. is a Distinguished George W. Brumley Professor.
Author information
Authors and Affiliations
Consortia
Contributions
Experiments were designed by E.E.D., E.A.P. and N.K. Mutational screening, analysis and confirmation was conducted by E.E.D., J.H., C.S., K.S., G.R., C.V.L., D.M.M., A.C.Y., D.A.W., P. Cruz., M.M., L.R.L., P. Cherukuri., B.M., N.F.H., J.C.M., R.W.B., G.G.B., the NISC Comparative Sequencing Program, G.G., E.A.O., J.G.G., T.A.-B., C.A.J., E.D.G. and R.A.G. Ciliopathy case samples were provided by J.H., S.R., B.T., I.K., N.A.S., T.J.N., K.J.S., H.K., T.E.G., R.A.L., C.B., S.S., P.J.S., P.L.B., J.G.G., E.R.M., T.A.-B., H.D., C.A.J., F.H. and N.K. In vivo and in vitro functional studies were carried out by E.E.D., Q.Z., Q.L., B.H.D. and L.M.D. The manuscript was written by E.E.D., Q.Z., E.A.P. and N.K. with helpful comments from C.B., J.G.G., E.R.M., T.A.-B., C.A.J. and F.H.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Tables 1–9 and Supplementary Figures 1–4 (PDF 6526 kb)
Rights and permissions
About this article
Cite this article
Davis, E., Zhang, Q., Liu, Q. et al. TTC21B contributes both causal and modifying alleles across the ciliopathy spectrum. Nat Genet 43, 189–196 (2011). https://doi.org/10.1038/ng.756
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng.756
This article is cited by
-
Transport and barrier mechanisms that regulate ciliary compartmentalization and ciliopathies
Nature Reviews Nephrology (2024)
-
Genetic and protein interaction studies between the ciliary dyslexia candidate genes DYX1C1 and DCDC2
BMC Molecular and Cell Biology (2023)
-
Skeletal ciliopathy: pathogenesis and related signaling pathways
Molecular and Cellular Biochemistry (2023)
-
Lethal neonatal respiratory failure due to biallelic variants in BBS1 and monoallelic variant in TTC21B
Pediatric Nephrology (2023)
-
Nephronophthisis: a pathological and genetic perspective
Pediatric Nephrology (2023)