Abstract
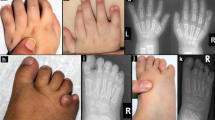
McKusick-Kaufman syndrome (MKKS, MIM 236700) is a human developmental anomaly syndrome comprising hydrometrocolpos (HMC), postaxial polydactyly (PAP) and congenital heart disease1,2 (CHD). MKKS has been mapped in the Old Order Amish population to 20p12, between D20S162 and D20S894 (ref. 3). Here we describe the identification of a gene mutated in MKKS. We analysed the approximately 450-kb candidate region by sample sequencing, which revealed the presence of several known genes and EST clusters. We evaluated candidate transcripts by northern-blot analysis of adult and fetal tissues. We selected one transcript with widespread expression, MKKS, for analysis in a patient from the Amish pedigree and a sporadic, non-Amish case. The Old Order Amish patient was found to be homozygous for an allele that had two missense substitutions and the non-Amish patient was a compound heterozygote for a frameshift mutation predicting premature protein truncation and a distinct missense mutation. The MKKS predicted protein shows amino acid similarity to the chaperonin family of proteins, suggesting a role for protein processing in limb, cardiac and reproductive system development. We believe that this is the first description of a human disorder caused by mutations affecting a putative chaperonin molecule.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
McKusick, V.A., Bauer, R.L., Koop, C.E. & Scott, R.B. Hydrometrocolpos as a simply inherited malformation. JAMA 189, 813–816 (1964).
Kaufman, R.L., Hartmann, A.F. & McAlister, W.H. Family studies in congenital heart disease, II: a syndrome of hydrometrocolpos, postaxial polydactyly and congenital heart disease. in Birth Defects Original Articles Series 85–87 (March of Dimes, White Plains, 1972).
Stone, D. et al. Genetic and physical mapping of the McKusick-Kaufman syndrome. Hum. Mol. Genet. 7, 475–481 (1998).
Oda, T. et al. Mutations in the human Jagged1 gene are responsible for Alagille syndrome. Nature Genet. 16, 235–242 (1997).
Li, L. et al. Alagille syndrome is caused by mutations in human Jagged1, which encodes a ligand for Notch1. Nature Genet. 16, 243–251 (1997).
Zhao, N., Hashida, H., Takahashi, N. & Sakaki, Y. Cloning and sequence analysis of the human SNAP25 cDNA. Gene 145, 313–314 (1994).
Agarwala, R., Biesecker, L.G., Hopkins, K.A., Francomano, C.A. & Schäffer, A.A. Software for constructing and verifying pedigrees within large genealogies and an application to the Old Order Amish of Lancaster County. Genome Res. 8, 211–221 (1998).
Wickner, S., Maurizi, M.R. & Gottesman, S. Posttranslational quality control: folding, refolding, and degrading proteins. Science 286, 1888–1893 (1999).
Braig, K. Chaperonins. Curr. Opin. Struct. Biol. 8, 159–165 (1998).
Ditzel, L. et al. Crystal structure of the thermosome, the archaeal chaperonin and homolog of CCT. Cell 93, 125–138 (1998).
Horwich, A.L., Weber-Ban, E.U. & Finley, D. Chaperone rings in protein folding and degradation. Proc. Natl Acad. Sci. USA 96, 11033–11040 (1999).
Llorca, O. et al. Eukaryotic type II chaperonin CCT interacts with actin through specific subunits. Nature 402, 693–696 (1999).
Wilson, R.K. & Mardis, E.R. Shotgun sequencing. in Analyzing DNA (Birren, B., Green, E.D., Klapholz, S., Myers, R.M. & Roskams, J. (Cold Spring Harbor Press, Cold Spring Harbor, 1997).
Gordon, D., Abajian, C. & Green, P. Consed: a graphical tool for sequence finishing. Genome Res. 8, 195–202 (1998).
Ewing, B. & Green, P. Base-calling of automated sequencer traces using phred. II. Error probabilities. Genome Res. 8, 186–194 (1998).
Ewing, B., Hillier, L., Wendl, M.C. & Green, P. Base-calling of automated sequencer traces using phred. I. Accuracy assessment. Genome Res. 8, 175–185 (1998).
Riley, J. et al. A novel, rapid method for the isolation of terminal sequences from yeast artificial chromosome (YAC) clones. Nucleic Acids Res. 18, 2887–2890 (1990).
Rosenberg, M.J. et al. The detection of chromosomal aberrations by a whole genome microsatellite screen. Am. J. Hum. Genet. (in press).
Slavotinek, A. et al. McKusick-Kaufman syndrome—phenotypic overlap with Bardet-Biedl syndrome necessitates modification of diagnostic criteria, management and genetic counseling. Am. J. Hum. Genet. 65, A186 (1999).
David, A. et al. Hydrometrocolpos and polydactyly: a common neonatal presentation of Bardet-Biedl and McKusick-Kaufman syndromes. J. Med. Genet. 36, 599–603 (1999).
Acknowledgements
We thank T. Ben-Yosef, L. Brody, S. Chandrasekharappa, P. Clouser, J. Davis, C. Francomano, E. Green, J. Johnston, D. Leja, R. Nussbaum, T. Oda, W. Pavan, M. Rosenberg, A. Schäffer, J. Touchman and the staff of NISC for production sequencing, critical review, helpful discussions, patient ascertainment, graphics and laboratory support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Stone, D., Slavotinek, A., Bouffard, G. et al. Mutation of a gene encoding a putative chaperonin causes McKusick-Kaufman syndrome. Nat Genet 25, 79–82 (2000). https://doi.org/10.1038/75637
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/75637
This article is cited by
-
Sequence variants in different genes underlying Bardet-Biedl syndrome in four consanguineous families
Molecular Biology Reports (2023)
-
A change of heart: new roles for cilia in cardiac development and disease
Nature Reviews Cardiology (2022)
-
Functional analysis of new human Bardet-Biedl syndrome loci specific variants in the zebrafish model
Scientific Reports (2019)
-
Combining targeted panel-based resequencing and copy-number variation analysis for the diagnosis of inherited syndromic retinopathies and associated ciliopathies
Scientific Reports (2018)