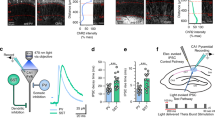
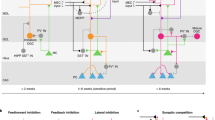
Abstract
Long-term consolidation of memories depends on processes occurring many hours after acquisition. Whether this involves plasticity that is specifically required for long-term consolidation remains unclear. We found that learning-induced plasticity of local parvalbumin (PV) basket cells was specifically required for long-term, but not short/intermediate-term, memory consolidation in mice. PV plasticity, which involves changes in PV and GAD67 expression and connectivity onto PV neurons, was regulated by cAMP signaling in PV neurons. Following induction, PV plasticity depended on local D1/5 dopamine receptor signaling at 0–5 h to regulate its magnitude, and at 12–14 h for its continuance, ensuring memory consolidation. D1/5 dopamine receptor activation selectively induced DARPP-32 and ERK phosphorylation in PV neurons. At 12–14 h, PV plasticity was required for enhanced sharp-wave ripple densities and c-Fos expression in pyramidal neurons. Our results reveal general network mechanisms of long-term memory consolidation that requires plasticity of PV basket cells induced after acquisition and sustained subsequently through D1/5 receptor signaling.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Change history
01 February 2016
In the version of this article initially published online, Charles Quairiaux and Christoph M Michel were listed as being affiliated with the Friedrich Miescher Institut, Basel, Switzerland. The correct affiliation is Neuroscience Department of the Medical Faculty and Center for Biomedical Imaging, University of Geneva, Geneva, Switzerland. The error has been corrected for the print, PDF and HTML versions of this article.
16 July 2018
In the version of this article initially published, the right panel in Fig. 2b was duplicated from the corresponding panel in Fig. 2c, and some data points in Fig. 3b were duplicated from Fig. 3a. None of the conclusions in the paper are affected. The errors have been corrected in the HTML and PDF versions of the article, and source data have been posted for the revised panels. The original and corrected figures are shown in the accompanying Author Correction.
References
Kandel, E.R., Dudai, Y. & Mayford, M.R. The molecular and systems biology of memory. Cell 157, 163–186 (2014).
Dudai, Y. The restless engram: consolidations never end. Annu. Rev. Neurosci. 35, 227–247 (2012).
Redondo, R.L. & Morris, R.G. Making memories last: the synaptic tagging and capture hypothesis. Nat. Rev. Neurosci. 12, 17–30 (2011).
Bekinschtein, P. et al. Persistence of long-term memory storage requires a late protein synthesis- and BDNF- dependent phase in the hippocampus. Neuron 53, 261–277 (2007).
Caroni, P., Chowdhury, A. & Lahr, M. Synapse rearrangements upon learning: from divergent-sparse connectivity to dedicated sub-circuits. Trends Neurosci. 37, 604–614 (2014).
Katche, C. et al. Delayed wave of c-Fos expression in the dorsal hippocampus involved specifically in persistence of long-term memory storage. Proc. Natl. Acad. Sci. USA 107, 349–354 (2010).
Sekeres, M.J. et al. Increasing CRTC1 function in the dentate gyrus during memory formation or reactivation increases memory strength without compromising memory quality. J. Neurosci. 32, 17857–17868 (2012).
Katche, C., Cammarota, M. & Medina, J.H. Molecular signatures and mechanisms of long-lasting memory consolidation and storage. Neurobiol. Learn. Mem. 106, 40–47 (2013).
O'Neill, J., Pleydell-Bouverie, B., Dupret, D. & Csicsvari, J. Play it again: reactivation of waking experience and memory. Trends Neurosci. 33, 220–229 (2010).
Carr, M.F., Jadhav, S.P. & Frank, L.M. Hippocampal replay in the awake state: a potential substrate for memory consolidation and retrieval. Nat. Neurosci. 14, 147–153 (2011).
Singer, A.C. & Frank, L.M. Rewarded outcomes enhance reactivation of experience in the hippocampus. Neuron 64, 910–921 (2009).
Girardeau, G., Cei, A. & Zugaro, M. Learning-induced plasticity regulates hippocampal sharp wave-ripple drive. J. Neurosci. 34, 5176–5183 (2014).
Girardeau, G., Benchenane, K., Wiener, S.I., Buzsáki, G. & Zugaro, M.B. Selective suppression of hippocampal ripples impairs spatial memory. Nat. Neurosci. 12, 1222–1223 (2009).
Ego-Stengel, V. & Wilson, M.A. Disruption of ripple-associated hippocampal activity during rest impairs spatial learning in the rat. Hippocampus 20, 1–10 (2010).
Girardeau, G. & Zugaro, M. Hippocampal ripples and memory consolidation. Curr. Opin. Neurobiol. 21, 452–459 (2011).
Hsiang, H.L. et al. Manipulating a “cocaine engram” in mice. J. Neurosci. 34, 14115–14127 (2014).
Nakashiba, T., Buhl, D.L., McHugh, T.J. & Tonegawa, S. Hippocampal CA3 output is crucial for ripple-associated reactivation and consolidation of memory. Neuron 62, 781–787 (2009).
Jadhav, S.P., Kemere, C., German, P.W. & Frank, L.M. Awake hippocampal sharp-wave ripples support spatial memory. Science 336, 1454–1458 (2012).
Singer, A.C., Carr, M.F., Karlsson, M.P. & Frank, L.M. Hippocampal SWR activity predicts correct decisions during the initial learning of an alternation task. Neuron 77, 1163–1173 (2013).
Hu, H., Gan, J. & Jonas, P. Interneurons. Fast-spiking, parvalbumin GABAergic interneurons: from cellular design to microcircuit function. Science 345, 1255263 (2014).
Sullivan, D. et al. Relationships between hippocampal sharp waves, ripples, and fast gamma oscillation: influence of dentate and entorhinal cortical activity. J. Neurosci. 31, 8605–8616 (2011).
Ellender, T.J., Nissen, W., Colgin, L.L., Mann, E.O. & Paulsen, O. Priming of hippocampal population bursts by individual perisomatic-targeting interneurons. J. Neurosci. 30, 5979–5991 (2010).
Carr, M.F., Karlsson, M.P. & Frank, L.M. Transient slow gamma synchrony underlies hippocampal memory replay. Neuron 75, 700–713 (2012).
Hájos, N. et al. Input-output features of anatomically identified CA3 neurons during hippocampal sharp wave/ripple oscillation in vitro. J. Neurosci. 33, 11677–11691 (2013).
Stark, E. et al. Pyramidal cell-interneuron interactions underlie hippocampal ripple oscillations. Neuron 83, 467–480 (2014).
Chiovini, B. et al. Dendritic spikes induce ripples in parvalbumin interneurons during hippocampal sharp waves. Neuron 82, 908–924 (2014).
Korotkova, T., Fuchs, E.C., Ponomarenko, A., von Engelhardt, J. & Monyer, H. NMDA receptor ablation on parvalbumin-positive interneurons impairs hippocampal synchrony, spatial representations, and working memory. Neuron 68, 557–569 (2010).
Cardin, J.A. et al. Driving fast-spiking cells induces gamma rhythm and controls sensory responses. Nature 459, 663–667 (2009).
Hensch, T.K. Critical period plasticity in local cortical circuits. Nat. Rev. Neurosci. 6, 877–888 (2005).
Donato, F., Rompani, S.B. & Caroni, P. Parvalbumin-expressing basket-cell network plasticity induced by experience regulates adult learning. Nature 504, 272–276 (2013).
Donato, F., Chowdhury, A., Lahr, M. & Caroni, P. Early- and late-born parvalbumin basket cell subpopulations exhibiting distinct regulation and roles in learning. Neuron 85, 770–786 (2015).
Magnus, C.J. et al. Chemical and genetic engineering of selective ion channel-ligand interactions. Science 333, 1292–1296 (2011).
Zelikowsky, M., Bissiere, S. & Fanselow, M.S. Contextual fear memories formed in the absence of the dorsal hippocampus decay across time. J. Neurosci. 32, 3393–3397 (2012).
Lisman, J.E. & Grace, A.A. The hippocampal-VTA loop: controlling the entry of information into long-term memory. Neuron 46, 703–713 (2005).
Rossato, J.I., Bevilaqua, L.R., Izquierdo, I., Medina, J.H. & Cammarota, M. Dopamine controls persistence of long-term memory storage. Science 325, 1017–1020 (2009).
Shohamy, D. & Adcock, R.A. Dopamine and adaptive memory. Trends Cogn. Sci. 14, 464–472 (2010).
Huang, Y.Y. & Kandel, E.R. D1/D5 receptor agonists induce a protein synthesis-dependent late potentiation in the CA1 region of the hippocampus. Proc. Natl. Acad. Sci. USA 92, 2446–2450 (1995).
Hansen, N. & Manahan-Vaughan, D. Dopamine D1/D5 receptors mediate informational saliency that promotes persistent hippocampal long-term plasticity. Cereb. Cortex 24, 845–858 (2014).
Ortiz, O. et al. Associative learning and CA3-CA1 synaptic plasticity are impaired in D1R null, Drd1a-/- mice and in hippocampal siRNA silenced Drd1a mice. J. Neurosci. 30, 12288–12300 (2010).
Beaulieu, J.M. & Gainetdinov, R.R. The physiology, signaling, and pharmacology of dopamine receptors. Pharmacol. Rev. 63, 182–217 (2011).
Valjent, E. et al. Regulation of a protein phosphatase cascade allows convergent dopamine and glutamate signals to activate ERK in the striatum. Proc. Natl. Acad. Sci. USA 102, 491–496 (2005).
Sternson, S.M. & Roth, B.L. Chemogenetic tools to interrogate brain functions. Annu. Rev. Neurosci. 37, 387–407 (2014).
Lemon, N. & Manahan-Vaughan, D. Dopamine D1/D5 receptors gate the acquisition of novel information through hippocampal long-term potentiation and long-term depression. J. Neurosci. 26, 7723–7729 (2006).
Baudonnat, M., Huber, A., David, V. & Walton, M.E. Heads for learning, tails for memory: reward, reinforcement and a role of dopamine in determining behavioral relevance across multiple timescales. Front. Neurosci. 7, 175 (2013).
Kilts, C.D., Dew, K.L., Ely, T.D. & Mailman, R.B. Quantification of R-(+)-7-chloro-8-hydroxy-1-phenyl-2,3,4,5-tetrahydro-1H-3-methyl-3- benzazepine in brain and blood by use of reversed-phase high-performance liquid chromatography with electrochemical detection. J. Chromatogr. 342, 452–457 (1985).
Ruediger, S., Spirig, D., Donato, F. & Caroni, P. Goal-oriented searching mediated by ventral hippocampus early in trial-and-error learning. Nat. Neurosci. 15, 1563–1571 (2012).
Hosp, J.A., Pekanovic, A., Rioult-Pedotti, M.S. & Luft, A.R. Dopaminergic projections from midbrain to primary motor cortex mediate motor skill learning. J. Neurosci. 31, 2481–2487 (2011).
McNamara, C.G., Tejero-Cantero, Á., Trouche, S., Campo-Urriza, N. & Dupret, D. Dopaminergic neurons promote hippocampal reactivation and spatial memory persistence. Nat. Neurosci. 17, 1658–1660 (2014).
Rasch, B. & Born, J. About sleep's role in memory. Physiol. Rev. 93, 681–766 (2013).
Abel, T., Havekes, R., Saletin, J.M. & Walker, M.P. Sleep, plasticity and memory from molecules to whole-brain networks. Curr. Biol. 23, R774–R788 (2013).
Acknowledgements
F. Carvalho (Friedrich Miescher Institut) found that PV plasticity was influenced by cAMP levels in PV neurons, and we acknowledge his contribution to this study. We thank S. Arber (FMI) for valuable comments on the manuscript. This work was supported in part by a Swiss National Fund grant to P.C. and by the NCCR Synapsy. The Friedrich Miescher Institut is part of the Novartis Research Foundation.
Author information
Authors and Affiliations
Contributions
S.K. devised and carried out the local DA and PV interference and behavior experiments. A.C. carried out detailed time course analyses of hippocampal PV network plasticity following fear conditioning and maze learning, as well as experiments addressing signaling in PV neurons. F.D. carried out the initial pharmacogenetic and ChABC interference, as well as PV analysis experiments. C.Q. and C.M.M. carried out the LFP experiments. P.C. helped to devise the experiments and wrote the manuscript. All of the authors discussed the results and commented the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 D1/5 receptor signaling at acquisition required for PV plasticity and long-term fear memory consolidation.
a. Analysis of vH D1/5 signaling interference during fear memory acquisition. Note low-PV instead of high-PV shift induction (left), unaffected intermediate-term memory (+6.5h) and absence of long-term fear memory (+24h). b. No rescue of PV plasticity or fear memory by D1 agonist delivery at +12h in mice treated with D1/5 antagonist just before acquisition. Note sustainment of low-PV plasticity by D1 agonist at +12h (left), and absence of fear memory (right). Average values from 4-6 mice and 60 PV neurons each; ANOVA, followed by Dunnet’s post-hoc; p <0.001 (***).
Supplementary Figure 2 Specific suppression of long-term memory consolidation by D1/5 receptor antagonist at +12h.
a. Analysis of average center of mass movement (translocation) velocity in mice subjected to cFC, treated with D1/5 receptor antagonist at different times after acquisition (x-axis), and analyzed at +24h in training context. Note how mice with suppressed fear memory (e.g. +13h) do not move faster than untreated control mice (0h), arguing against loss of freezing due to hyper-locomotion. b. Mice that underwent cFC and were treated with D1/5 antagonist at +12h exhibit robust fear memory at +2d when reconditioned at +24h (orange), indicating that D1/5 antagonist at +12h specifically suppressed long-term consolidation of the fear memory induced 12h before (red). c. NE receptor antagonist delivered at +12h to vH does not interfere with long-term consolidation of fear memory. Average values from 4-6 mice.
Supplementary Figure 3 Specific suppression of individual long-term memories by D1/5 receptor antagonist at +12h.
a. Delivery of D1/5 receptor antagonist to vH at +12h suppresses fear memory consolidation regardless of whether acquisition occurred at 09:00, 15:00 or 21:00. b, c. Memory specific requirement for vH D1/5 receptor signaling at +12h for long-term memory consolidation. b: Specific suppression of TR1 or TR2 fear memory by D1/5 receptor antagonist at +12h after corresponding fear conditioning. c: Enhanced high-PV plasticity upon second cFC protocol, and reversal to high-PV levels comparable to those induced by one cFC protocol upon delivery of D1/5 receptor antagonist at +12h. Average values from 4-6 mice and 60 PV neurons each; Student’s t-test; p<0.001 (***).
Supplementary Figure 4 Pharmacogenetic induction of high-PV plasticity in naïve mice not sufficient to induce freezing.
Mice expressing pharmacogenetic activator virus in vH CA3 PV neurons explored context without foot shocks (noUS), where treated with pharmacogenetic ligand at +3h, and tested for freezing in context at +12h or +24. Average values from 4-6 mice and 60 PV neurons each.
Supplementary Figure 5 No detectable expression of cFos in vH CA3b PV neurons 90min after cFC.
Representative example of c-Fos/PV double-labeling experiment in vH CA3b 90min after cFC. Yellow arrows: PV+ neurons. Bar: 50 μm. Average values from 3 mice and 80 PV neurons each.
Supplementary Figure 6 Analysis of dye spread and electrode paths.
a. Left: Mouse brain coronal section with location of vH (DG, CA3, CA1). Numbers: antero-posterior coordinates caudal to bregma. Right: Representative image of Bodipy dye targeted at vH CA3 and its spread from the target site 6h after injection. Bar, 300μm. b. 16 channel Neuronexus probes (LFP experiments) inserted in both vH with 16 contacts covering 0.8mm length at the electrode tip. Example of electrode track position revealed with Dye I (right). Bar, 1500μm. c. Representative images of Nissl stained vH section (50 μm) through the cannula track after dye injection (left). Bar: 200 μm. Infusion site (dotted line) and injector track (black line) (right). Bar 100 μm. d. Injection sites from 8 mice into CA3 region of vH.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–6 (PDF 1036 kb)
Source data
Rights and permissions
About this article
Cite this article
Karunakaran, S., Chowdhury, A., Donato, F. et al. PV plasticity sustained through D1/5 dopamine signaling required for long-term memory consolidation. Nat Neurosci 19, 454–464 (2016). https://doi.org/10.1038/nn.4231
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.4231
This article is cited by
-
Biphasic Npas4 expression promotes inhibitory plasticity and suppression of fear memory consolidation in mice
Molecular Psychiatry (2024)
-
Fast-spiking parvalbumin-positive interneurons in brain physiology and Alzheimer’s disease
Molecular Psychiatry (2023)
-
Knockdown of METTL16 disrupts learning and memory by reducing the stability of MAT2A mRNA
Cell Death Discovery (2022)
-
Forgetting as a form of adaptive engram cell plasticity
Nature Reviews Neuroscience (2022)
-
PV network plasticity mediated by neuregulin1-ErbB4 signalling controls fear extinction
Molecular Psychiatry (2022)