Abstract
Regulation of AMPA receptor (AMPAR) membrane trafficking is critical for synaptic plasticity, as well as for learning and memory. However, the mechanisms of AMPAR trafficking in vivo remain elusive. Using in vivo two-photon microscopy in the mouse somatosensory barrel cortex, we found that acute whisker stimulation led to a significant increase in the intensity of surface AMPAR GluA1 subunit (sGluA1) in both spines and dendritic shafts and a small increase in spine size relative to prestimulation values. Interestingly, the initial spine properties biased spine changes following whisker stimulation. Changes in spine sGluA1 intensity were positively correlated with changes in spine size and dendritic shaft sGluA1 intensity following whisker stimulation. The increase in spine sGluA1 intensity evoked by whisker stimulation was NMDA receptor dependent and long lasting, similar to major forms of synaptic plasticity in the brain. In this study we were able to observe experience-dependent AMPAR trafficking in real time and characterize, in vivo, a major form of synaptic plasticity in the brain.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Huganir, R.L. & Nicoll, R.A. AMPARs and synaptic plasticity: the last 25 years. Neuron 80, 704–717 (2013).
Lee, H.K. & Kirkwood, A. AMPA receptor regulation during synaptic plasticity in hippocampus and neocortex. Semin. Cell Dev. Biol. 22, 514–520 (2011).
Lüscher, C. & Malenka, R.C. NMDA receptor-dependent long-term potentiation and long-term depression (LTP/LTD). Cold Spring Harb. Perspect. Biol. 4, a005710 (2012).
Zuo, Y., Yang, G., Kwon, E. & Gan, W.B. Long-term sensory deprivation prevents dendritic spine loss in primary somatosensory cortex. Nature 436, 261–265 (2005).
Holtmaat, A., Wilbrecht, L., Knott, G.W., Welker, E. & Svoboda, K. Experience-dependent and cell-type-specific spine growth in the neocortex. Nature 441, 979–983 (2006).
Clem, R.L., Celikel, T. & Barth, A.L. Ongoing in vivo experience triggers synaptic metaplasticity in the neocortex. Science 319, 101–104 (2008).
Makino, H. & Malinow, R. Compartmentalized versus global synaptic plasticity on dendrites controlled by experience. Neuron 72, 1001–1011 (2011).
Saito, T. & Nakatsuji, N. Efficient gene transfer into the embryonic mouse brain using in vivo electroporation. Dev. Biol. 240, 237–246 (2001).
Petersen, C.C. The functional organization of the barrel cortex. Neuron 56, 339–355 (2007).
Holtmaat, A. et al. Long-term, high-resolution imaging in the mouse neocortex through a chronic cranial window. Nat. Protoc. 4, 1128–1144 (2009).
Xu, H.T., Pan, F., Yang, G. & Gan, W.B. Choice of cranial window type for in vivo imaging affects dendritic spine turnover in the cortex. Nat. Neurosci. 10, 549–551 (2007).
Harrison, T.C., Sigler, A. & Murphy, T.H. Simple and cost-effective hardware and software for functional brain mapping using intrinsic optical signal imaging. J. Neurosci. Methods 182, 211–218 (2009).
Kopec, C.D., Real, E., Kessels, H.W. & Malinow, R. GluR1 links structural and functional plasticity at excitatory synapses. J. Neurosci. 27, 13706–13718 (2007).
Noguchi, J. et al. In vivo two-photon uncaging of glutamate revealing the structure-function relationships of dendritic spines in the neocortex of adult mice. J. Physiol. 589, 2447–2457 (2011).
O'Brien, R.J. et al. Activity-dependent modulation of synaptic AMPA receptor accumulation. Neuron 21, 1067–1078 (1998).
Mammen, A.L., Huganir, R.L. & O'Brien, R.J. Redistribution and stabilization of cell surface glutamate receptors during synapse formation. J. Neurosci. 17, 7351–7358 (1997).
Clem, R.L. & Barth, A. Pathway-specific trafficking of native AMPARs by in vivo experience. Neuron 49, 663–670 (2006).
Takahashi, T., Svoboda, K. & Malinow, R. Experience strengthening transmission by driving AMPA receptors into synapses. Science 299, 1585–1588 (2003).
Petersen, C.C. & Crochet, S. Synaptic computation and sensory processing in neocortical layer 2/3. Neuron 78, 28–48 (2013).
Crochet, S., Poulet, J.F., Kremer, Y. & Petersen, C.C. Synaptic mechanisms underlying sparse coding of active touch. Neuron 69, 1160–1175 (2011).
O'Connor, D.H., Peron, S.P., Huber, D. & Svoboda, K. Neural activity in barrel cortex underlying vibrissa-based object localization in mice. Neuron 67, 1048–1061 (2010).
Matsuzaki, M., Honkura, N., Ellis-Davies, G.C. & Kasai, H. Structural basis of long-term potentiation in single dendritic spines. Nature 429, 761–766 (2004).
Nägerl, U.V., Eberhorn, N., Cambridge, S.B. & Bonhoeffer, T. Bidirectional activity-dependent morphological plasticity in hippocampal neurons. Neuron 44, 759–767 (2004).
Sdrulla, A.D. & Linden, D.J. Double dissociation between long-term depression and dendritic spine morphology in cerebellar Purkinje cells. Nat. Neurosci. 10, 546–548 (2007).
Knott, G.W., Quairiaux, C., Genoud, C. & Welker, E. Formation of dendritic spines with GABAergic synapses induced by whisker stimulation in adult mice. Neuron 34, 265–273 (2002).
Bhatt, D.H., Zhang, S. & Gan, W.B. Dendritic spine dynamics. Annu. Rev. Physiol. 71, 261–282 (2009).
Fu, M., Yu, X., Lu, J. & Zuo, Y. Repetitive motor learning induces coordinated formation of clustered dendritic spines in vivo. Nature 483, 92–95 (2012).
Trachtenberg, J.T. et al. Long-term in vivo imaging of experience-dependent synaptic plasticity in adult cortex. Nature 420, 788–794 (2002).
Yang, G., Chang, P.C., Bekker, A., Blanck, T.J. & Gan, W.B. Transient effects of anesthetics on dendritic spines and filopodia in the living mouse cortex. Anesthesiology 115, 718–726 (2011).
Pérez-Otaño, I. & Ehlers, M.D. Homeostatic plasticity and NMDA receptor trafficking. Trends Neurosci. 28, 229–238 (2005).
Gambino, F. et al. Sensory-evoked LTP driven by dendritic plateau potentials in vivo. Nature 515, 116–119 (2014).
Pologruto, T.A., Sabatini, B.L. & Svoboda, K. ScanImage: flexible software for operating laser scanning microscopes. Biomed. Eng. Online 2, 13 (2003).
Longair, M.H., Baker, D.A. & Armstrong, J.D. Simple Neurite Tracer: open source software for reconstruction, visualization and analysis of neuronal processes. Bioinformatics 27, 2453–2454 (2011).
Acknowledgements
We thank L.J. Volk and N.K. Hussain for manuscript comments, T. Shelley for technical support and L. Hamm for management support. This work was supported by NIH grants P50MH100024, R01NS036715 (both to R.L.H.) and R01MH051106 (to D.J.L.) and by the Howard Hughes Medical Institute (to R.L.H.).
Author information
Authors and Affiliations
Contributions
Y.Z., D.-T.L. and R.L.H. designed experiments. Y.Z. performed experiments and analyzed data. R.H.C. developed analytic tools and designed the software. Y.Z., R.H.C., D.J.L. and R.L.H. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
Under a licensing agreement between Millipore Corporation and The Johns Hopkins University, R.L.H. is entitled to a share of royalties received by the University on sales of products described in this article. R.L.H. is a paid consultant to Millipore Corporation. The terms of this arrangement are being managed by The Johns Hopkins University in accordance with its conflict-of-interest policies.
Integrated supplementary information
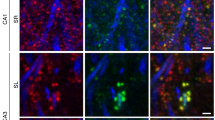
Supplementary Figure 1 SEP-GluA1 is moderately overexpressed in electroporated animals.
a, Immunostaining of GluA1 in brain slices of GluA1 knockout (left) and wild type animals (middle). High-magnification view of punctate GluA1 staining in wild type animals (right). b, Immunostaining of GluA1 (left) and GFP (right) to reveal SEP-GluA1 in brain slices of electroporated pups. Note that the level of total GluA1 is similar between transfected and untransfected neurons. Arrows mark neurons transfected with SEP-GluA1. c, Quantification of relative GluA1 expression level in transfected (red) and untransfected (black) neurons from b, GluA1 expression level in untransfected neurons are normalized to 100%. p > 0.08, student’s t test.
Supplementary Figure 2 Chronic cranial window surgery does not cause astrocyte proliferation.
Immunostaining of astrocyte marker GFAP under a chronic cranial window after animals are finished with two-photon imaging. Surgery hemisphere is displayed in the upper panel, control hemisphere in the middle panel, and hemisphere from intentionally injured animals in the lower panel.
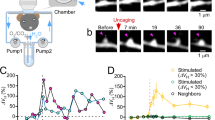
Supplementary Figure 3 Acute whisker stimulation evokes a significant increase in average spine sGluA1 intensity and average shaft sGluA1 intensity, with little effect on average spine size.
a, Absolute changes in spine structure intensity for control and stimulated animals (0.01 ± 0.005 vs. 0.004 ± 0.005 at hour 1, 0.02 ± 0.006 vs. 0.02 ± 0.006 at hour 2, 0.025 ± 0.007 vs. 0.016 ± 0.007 at hour 3). b, Percent change in spine structure intensity for control and stimulated animals with raw data as in Fig. 1c (107.05% ± 1.36% vs. 105.92% ± 1.3% at hour 1, 108.96 ± 1.54% vs. 111.59% ± 1.5% at hour 2, 112.11% ± 1.68% vs. 110.61% ± 1.81% at hour 3). c, Absolute changes in spine sGluA1 for control and stimulated animals ((–0.10 ± 0.04 vs. 0.35 ± 0.05 at hour 1, 0.03 ± 0.05 vs. 0.41 ± 0.05 at hour 2, 0.17 ± 0.06 vs. 0.41 ± 0.05 at hour 3). d, Percent change in spine sGluA1 intensity for control and stimulated animals with raw data as in Fig. 1e (96.19% ± 1.34% vs. 128.55% ± 2.91% at hour 1, 100.74% ± 1.55% vs. 133.75% ± 3.15% at hour 2, 106.47% ± 1.75% vs. 135.75% ± 3.29% at hour 3). e, Absolute changes in shaft sGluA1 for control and stimulated animals ((–0.19 ± 0.03 vs. 0.58 ± 0.05 at hour 1, 0.005 ± 0.03 vs. 0.65 ± 0.05 at hour 2, 0.18 ± 0.04 vs. 0.65 ± 0.05 at hour 3). f, Percent change in shaft sGluA1 intensity for control and stimulated animals with raw data as in Fig. 1g (94.19% ± 0.68% vs. 127.52% ± 2.42% at hour 1, 97.09% ± 0.73% vs. 130.07% ± 2.59% at hour 2, 99.41% ± 0.95% vs. 129.35% ± 2.34% at hour 3). ***p < 0.001, **p < 0.01, two-way ANOVA with Bonferroni posttests. a.u., artificial unit. Error bars = s.e.m.
Supplementary Figure 4 Determining regions of interest for intensity analysis.
a, Each spine was assigned three different non-overlapping regions of interest (ROIs): spineROI, shaftROI and backgroundROI. The spineROI enclosed the spine head and did not include pixels within the radius of the dendritic backbone. The shaftROI was constructed from the backbone line and radius of the dendrite, centered on each spine and symmetrically expanded along the dendrite to match the number of pixels in the spineROI. The backgroundROI (same shape and number of pixels as the spineROI) was translated in x/y to a nearby region of the image that was representative of the background fluorescence. b, To obtain intensity measurements for the GluA1 channel, the three ROIs created using the backbone and spine markings in the structural channel (panel a), were placed in the same position on the GluA1 channel and the backgroundROI was translated again to minimize intensity. Note, all spine markings and intensity analysis were performed on the raw 3D image stacks.
Supplementary Figure 5 Whisker stimulation does not alter spine turnover.
a, Example time series with 4 persistent spines. Dendritic backbone (cyan), manually selected spines (yellow numbers and arrows) and the line connecting the spine head to the dendritic backbone (blue). b, Pooled data of spine turnover for dendrites imaged in stimulated and control animals (stimulated: n = 38 dendrites, 5 animals; control n = 38 dendrites, 4 animals). In stimulated animals, whisker stimulation is given between hour 0 and 1. All analysis is performed on individual dendrites and then pooled across each condition. Spine density (spines/μm) is the number of spines on a given dendrite divided by the total dendritic length (μm) that was scored for that dendrite. Percent added is the number of spines categorized as added (on a given dendrite) divided by the total number of spines for the same dendrite in the previous time-point. Likewise, percent subtracted is the number of spines categorized as subtracted on a given dendrite divided by the total number of spines for the same dendrite in the previous time-point. Turn over ratio (TOR) is the sum of added and subtracted spines within a dendrite divided by two times the total number of spines for the same dendrite in the previous time-point. There is no significant difference between dendrites from stimulated and control animals in any of the spine turnover measurements at any of the time-points (Two-way ANOVA).
Supplementary Figure 6 Initial spine and shaft properties bias spine changes following whisker stimulation.
a, Scatter plots of absolute change in spine structure intensity at 1 hour versus initial spine structure intensity at 0 hour with linear fits. (r = –0.2546, p < 0.001). b, Scatter plots of absolute change in spine sGluA1 intensity at 1 hour versus initial spine structure intensity at 0 hour with linear fits. (r = –0.1057, p = 0.0124). c, Scatter plots of absolute change in shaft sGluA1 intensity at 1 hour versus initial spine structure intensity at 0 hour with linear fits. (r = 0.0686, p = 0.1052). d, Scatter plots of absolute change in spine sGluA1 intensity at 1 hour versus initial spine sGluA1 intensity at 0 hour with linear fits. (r = 0.0446, p = 0.2927). e, Scatter plots of absolute change in spine sGluA1 intensity at 1 hour versus initial shaft sGluA1 intensity at 0 hour with linear fits. (r = 0.0647, p = 0.1262). f, Scatter plots of absolute change in shaft sGluA1 intensity at 1 hour versus initial shaft sGluA1 intensity at 0 hour with linear fits. (r = 0.1608, p < 0.001). In each panel, center solid black line is linear fit, enclosing solid black lines are 95% confidence intervals, horizontal dashed line is 0. a.u., artificial unit. r, Pearson’s linear correlation coefficient. p, Pearson’s correlation t test.
Supplementary Figure 7 Correlation between changes in intensity in spine sGluA1 and shaft sGluA1.
No significant correlation was detected between spine sGluA1 and spine size at 1 hour in the subset of spines that showed a > 30% increase in spine sGluA1 after whisker stimulation. 206 spines, r, Pearson’s linear correlation coefficient.
Supplementary Figure 8 Correlation between changes in intensity in spine sGluA1 and shaft sGluA1 over time in stimulated animals.
One hour after stimulation, the changes in spine sGluA1 are correlated with shaft sGluA1 (upper left). This correlation diminishes by hour 48 (lower right). 105 spines, 13 dendrites in 3 stimulated animals. r, Pearson’s linear correlation coefficient, p value is from Monte-Carlo shuffling.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–8 (PDF 4812 kb)
41593_2015_BFnn3936_MOESM36_ESM.mov
Z-stack two-photon images of layer 2/3 pyramidal neurons in the barrel cortex of electroporated animals (SEP-GluA1 in green, dsRed2 in magenta). (MOV 5689 kb)
Rights and permissions
About this article
Cite this article
Zhang, Y., Cudmore, R., Lin, DT. et al. Visualization of NMDA receptor–dependent AMPA receptor synaptic plasticity in vivo. Nat Neurosci 18, 402–407 (2015). https://doi.org/10.1038/nn.3936
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.3936
This article is cited by
-
Cross-modality supervised image restoration enables nanoscale tracking of synaptic plasticity in living mice
Nature Methods (2023)
-
Rabphilin-3A undergoes phase separation to regulate GluN2A mobility and surface clustering
Nature Communications (2023)
-
Nanoscale rules governing the organization of glutamate receptors in spine synapses are subunit specific
Nature Communications (2022)
-
Weight dependence in BCM leads to adjustable synaptic competition
Journal of Computational Neuroscience (2022)
-
Net decrease in spine-surface GluA1-containing AMPA receptors after post-learning sleep in the adult mouse cortex
Nature Communications (2021)