Abstract
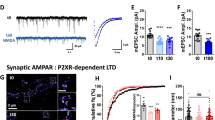
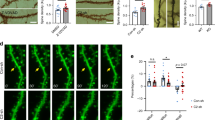
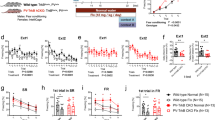
Phosphatidylinositol 3-kinase (PI3K) has been implicated in synaptic plasticity and other neural functions in the brain. However, the role of individual PI3K isoforms in the brain is unclear. We investigated the role of PI3Kγ in hippocampal-dependent synaptic plasticity and cognitive functions. We found that PI3Kγ has a crucial and specific role in NMDA receptor (NMDAR)-mediated synaptic plasticity at mouse Schaffer collateral–commissural synapses. Both genetic deletion and pharmacological inhibition of PI3Kγ disrupted NMDAR long-term depression (LTD) while leaving other forms of synaptic plasticity intact. Accompanying this physiological deficit, the impairment of NMDAR LTD by PI3Kγ blockade was specifically correlated with deficits in behavioral flexibility. These findings suggest that a specific PI3K isoform, PI3Kγ, is critical for NMDAR LTD and some forms of cognitive function. Thus, individual isoforms of PI3Ks may have distinct roles in different types of synaptic plasticity and may therefore influence various kinds of behavior.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Dhand, R. et al. PI 3-kinase is a dual specificity enzyme: autoregulation by an intrinsic protein-serine kinase activity. EMBO J. 13, 522–533 (1994).
Bondev, A., Rubio, I. & Wetzker, R. Differential regulation of lipid and protein kinase activities of phosphoinositide 3-kinase gamma in vitro. Biol. Chem. 380, 1337–1340 (1999).
Toker, A. & Cantley, L.C. Signaling through the lipid products of phosphoinositide-3-OH kinase. Nature 387, 673–676 (1997).
Carpenter, C.L. & Cantley, L.C. Phosphoinositide kinases. Curr. Opin. Cell Biol. 8, 153–158 (1996).
Oudit, G.Y. et al. The role of phosphoinositide-3 kinase and PTEN in cardiovascular physiology and disease. J. Mol. Cell Cardiol. 37, 449–471 (2004).
Wymann, M.P. & Pirola, L. Structure and function of phosphoinositide 3-kinases. Biochim. Biophys. Acta 1436, 127–150 (1998).
Hawkins, P.T., Anderson, K.E., Davidson, K. & Stephens, L.R. Signaling through Class I PI3Ks in mammalian cells. Biochem. Soc. Trans. 34, 647–662 (2006).
Rückle, T., Schwarz, M.K. & Rommel, C. PI3Kγ inhibition: towards an 'aspirin of the 21st century'? Nat. Rev. Drug Discov. 5, 903–918 (2006).
Kerfant, B.G., Rose, R.A., Sun, H. & Backx, P.H. Phosphoinositide 3-kinase gamma regulates cardiac contractility by locally controlling cyclic adenosine monophosphate levels. Trends Cardiovasc. Med. 16, 250–256 (2006).
Kang, H., Chang, W., Hurley, M., Vignery, A. & Wu, D. Important roles of PI3Kγ in osteoclastogenesis and bone homeostasis. Proc. Natl. Acad. Sci. USA 107, 12901–12906 (2010).
Hayer, S. et al. PI3Kγ regulates cartilage damage in chronic inflammatory arthritis. FASEB J. 23, 4288–4298 (2009).
Kelly, A. & Lynch, M.A. Long-term potentiation in dentate gyrus of the rat is inhibited by the phosphoinositide 3-kinase inhibitor, wortmannin. Neuropharmacology 39, 643–651 (2000).
Sanna, P.P. et al. Phosphatidylinositol 3-kinase is required for the expression but not for the induction or the maintenance of long-term potentiation in the hippocampal CA1 region. J. Neurosci. 22, 3359–3365 (2002).
Opazo, P., Watabe, A.M., Grant, S.G.N. & O'Dell, T.J. Phosphatidylinositol 3-kinase regulates the induction of long-term potentiation through extracellular signal–related kinase-independent mechanisms. J. Neurosci. 23, 3679–3688 (2003).
Horwood, J.M., Dufour, F., Laroche, S. & Davis, S. Signaling mechanisms mediated by the phosphoinositide 3-kinase/Akt cascade in synaptic plasticity and memory in the rat. Eur. J. Neurosci. 23, 3375–3384 (2006).
Ran, I. et al. Persistent transcription- and translation-dependent long-term potentiation induced by mGluR1 in hippocampal interneurons. J. Neurosci. 29, 5605–5615 (2009).
Lin, C.H. et al. A role for the PI-3 kinase signaling pathway in fear conditioning and synaptic plasticity in the amygdala. Neuron 31, 841–851 (2001).
Hou, L. & Klann, E. Activation of the phosphoinositide 3-kinase-akt-mammalian target of rapamycin signaling pathway is required for metabotropic glutamate receptor-dependent long-term depression. J. Neurosci. 24, 6352–6361 (2004).
Daw, M.I. et al. Phosphatidylinositol 3 kinase regulates synapse specificity of hippocampal long-term depression. Nat. Neurosci. 5, 835–836 (2002).
van der Heide, L.P., Kamal, A., Artola, A., Gispen, W.H. & Ramakers, G.M.J. Insulin modulates hippocampal activity–dependent synaptic plasticity in a N-methyl-D-aspartate receptor and phosphatidyl-inositol-3-kinase–dependent manner. J. Neurochem. 94, 1158–1166 (2005).
Selbach, O. et al. Orexins/hypocretins control bistability of hippocampal long-term synaptic plasticity through co-activation of multiple kinases. Acta Physiol. (Oxf) 198, 277–285 (2009).
Chen, X. et al. PI3 kinase signaling is required for retrieval and extinction of contextual memory. Nat. Neurosci. 8, 925–931 (2005).
Mizuno, M. et al. Phosphatidylinositol 3-kinase: a molecule mediating BDNF-dependent spatial memory formation. Mol. Psychiatry 8, 217–224 (2003).
Barros, D.M. et al. LY294002, an inhibitor of phosphoinositide 3-kinase given into rat hippocampus impairs acquisition, consolidation and retrieval of memory for one-trial step-down inhibitory avoidance. Behav. Pharmacol. 12, 629–634 (2001).
Bartlett, S.E., Reynolds, A.J., Tan, T., Heydon, K. & Hendry, I.A. Differential mRNA expression and subcellular locations of PI3-kinase isoforms in sympathetic and sensory neurons. J. Neurosci. Res. 56, 44–53 (1999).
Bernstein, H.G., Keilhoff, C., Reiser, M., Freese, S. & Wetzker, R. Tissue distribution and subcellular localization of a G protein–activated phosphoinositide 3-kinase. An immunohistochemical study. Cell Mol Biol. 44, 973–983 (1998).
Sasaki, T. et al. Function of PI3Kγ in thymocyte development, T cell activation, and neutrophil migration. Science 287, 1040–1046 (2000).
Liu, L. et al. Role of NMDA receptor subtypes in governing the direction of hippocampal synaptic plasticity. Science 304, 1021–1024 (2004).
Zhuo, M. Plasticity of NMDA receptor NR2B subunit in memory and chronic pain. Mol Brain 2, 4 (2009).
Camps, M. et al. Blockade of PI3Kγ suppresses joint inflammation and damage in mouse models of rheumatoid arthritis. Nat. Med. 11, 936–943 (2005).
Hansel, C., Linden, D.J. & D'Angelo, E. Beyond parallel fiber LTD: the diversity of synaptic and nonsynaptic plasticity in the cerebellum. Nat. Neurosci. 4, 467–475 (2001).
Peineau, S. et al. LTP inhibits LTD in the hippocampus via regulation of GSK3 beta. Neuron 53, 703–717 (2007).
Peineau, S. et al. A systematic investigation of the protein kinases involved in NMDA receptor–dependent LTD: evidence for a role of GSK-3 but not other serine/threonine kinases. Mol Brain 2, 22 (2009).
Hayakawa, M. et al. Synthesis and biological evaluation of 4-morpholino-2-phenylquinazolines and related derivatives as novel PI3 kinase p110α inhibitors. Bioorg. Med. Chem. 14, 6847–6858 (2006).
Morishita, W. et al. N-ethylmaleimide blocks depolarization-induced suppression of inhibition and enhances GABA release in the rat hippocampal slice in vitro. J. Neurosci. 17, 941–950 (1997).
Collingridge, G.L., Peineau, S., Howland, J.G. & Wang, Y.T. Long-term depression in the CNS. Nat. Rev. Neurosci. 11, 459–473 (2010).
Zhu, J.J., Qin, Y., Zhao, M., Van Aelst, L. & Malinow, R. Ras and Rap control AMPA receptor trafficking during synaptic plasticity. Cell 110, 443–455 (2002).
Zhu, Y. et al. Rap2-JNK removes synaptic AMPA receptors during depotentiation. Neuron 46, 905–916 (2005).
Bos, J.L., de Rooij, J. & Reedquist, K.A. Rap1 signaling: adhering to new models. Nat. Rev. Mol. Cell Biol. 2, 369–377 (2001).
Yamboliev, I.A., Wiesmann, K.M., Singer, C.A., Hedges, J.C. & Gerthoffer, W.T. Phosphatidylinositol 3-kinases regulate ERK and p38 MAP kinases in canine colonic smooth muscle. Am. J. Physiol. Cell Physiol. 279, C352–C360 (2000).
Nicholls, R.E. et al. Transgenic mice lacking NMDAR-dependent LTD exhibit deficits in behavioral flexibility. Neuron 58, 104–117 (2008).
Vanhaesebroeck, B., Guillermet-Guibert, J., Graupera, M. & Bilanges, B. The emerging mechanisms of isoform-specific PI3K signaling. Nat. Rev. Mol. Cell Biol. 11, 329–341 (2010).
Hisatsune, C., Umemori, H., Mishina, M. & Yamamoto, T. Phosphorylation-dependent interaction of the N-methyl-D-aspartate receptor epsilon 2 subunit with phosphatidylinositol 3-kinase. Genes Cells 4, 657–666 (1999).
Husi, H., Ward, M.A., Choudhary, J.S., Blackstock, W.P. & Grant, S.G. Proteomic analysis of NMDA receptor-adhesion protein signaling complexes. Nat. Neurosci. 3, 661–669 (2000).
Etkin, A. et al. A role in learning for SRF: Deletion in the adult forebrain disrupts LTD and the formation of an immediate memory of a novel context. Neuron 50, 127–143 (2006).
Kemp, A. & Manahan-Vaughan, D. Hippocampal long-term depression and long-term potentiation encode different aspects of novelty acquisition. Proc. Natl. Acad. Sci. USA 101, 8192–8197 (2004).
Manahan-Vaughan, D. & Braunewell, K.H. Novelty acquisition is associated with induction of hippocampal long-term depression. Proc. Natl. Acad. Sci. USA 96, 8739–8744 (1999).
Wong, T.P. et al. Hippocampal long-term depression mediates acute stress-induced spatial memory retrieval impairment. Proc. Natl. Acad. Sci. USA 104, 11471–11476 (2007).
Zeng, H. et al. Forebrain-specific calcineurin knockout selectively impairs bidirectional synaptic plasticity and working/episodic-like memory. Cell 107, 617–629 (2001).
Jin, Y., Kim, S.J., Kim, J., Worley, P.F. & Linden, D.J. Long-term depression of mGluR1 signaling. Neuron 55, 277–287 (2007).
Acknowledgements
This work was supported by the Creative Research Initiatives Program of the Korean Ministry of Science and Technology and World Class University project. J.-I.K. and H.-R.L. are supported by a BK21 fellowship and Seoul Science fellowship. S.-E.S. and J.B. are supported by a BK21 fellowship. P.H.B. is a Career Investigator of the Heart and Stroke Foundation of Ontario and has support from a Canadian Institutes of Health Research grant (62954). B.-K.K. is a Yonam Foundation Scholar. G.L.C. and M.Z. are WCU International Scholars.
Author information
Authors and Affiliations
Contributions
J.-I.K. and H.-R.L. designed, performed and analyzed most of the electrophysiology and behavioral experiments, and wrote the manuscript. S.S., J.B., N.-K.Y., J.-H.C., H.-G.K., Y.-S.L., S.-W.P., C.K., S.-J.A., S.Y.C., H.K., K.-H.K., D.-J.J., K.L. and S.J.K. conducted the biochemical, electrophysiological and behavioral studies. Y.-S.L., P.H.B., C.A.B., D.-J.J., K.L., E.K., M.Z. and G.L.C. aided in the interpretation of data and contributed to editing the manuscript. B.-K.K. supervised the project, designed the experiments and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–7 (PDF 9005 kb)
Rights and permissions
About this article
Cite this article
Kim, JI., Lee, HR., Sim, Se. et al. PI3Kγ is required for NMDA receptor–dependent long-term depression and behavioral flexibility. Nat Neurosci 14, 1447–1454 (2011). https://doi.org/10.1038/nn.2937
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.2937
This article is cited by
-
Intranasal delivery of siRNA targeting NR2B attenuates cancer-associated neuropathic pain
Journal of Pharmaceutical Investigation (2024)
-
Long-term depression in neurons involves temporal and ultra-structural dynamics of phosphatidylinositol-4,5-bisphosphate relying on PIP5K, PTEN and PLC
Communications Biology (2023)
-
Cyclin Y regulates spatial learning and memory flexibility through distinct control of the actin pathway
Molecular Psychiatry (2023)
-
Neddylation-dependent protein degradation is a nexus between synaptic insulin resistance, neuroinflammation and Alzheimer’s disease
Translational Neurodegeneration (2022)
-
Reset of hippocampal–prefrontal circuitry facilitates learning
Nature (2021)