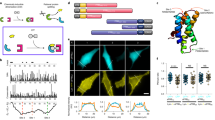
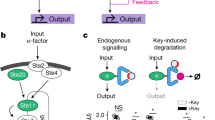
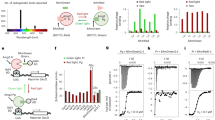
Abstract
Using a newly synthesized gibberellin analog containing an acetoxymethyl group (GA3-AM) and its binding proteins, we developed an efficient chemically inducible dimerization (CID) system that is completely orthogonal to existing rapamycin-mediated protein dimerization. Combining the two systems should allow applications that have been difficult or impossible with only one CID system. By using both chemical inputs (rapamycin and GA3-AM), we designed and synthesized Boolean logic gates in living mammalian cells. These gates produced output signals such as fluorescence and membrane ruffling on a timescale of seconds, substantially faster than earlier intracellular logic gates. The use of two orthogonal dimerization systems in the same cell also allows for finer modulation of protein perturbations than is possible with a single dimerizer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Fegan, A., White, B., Carlson, J.C. & Wagner, C.R. Chemically controlled protein assembly: techniques and applications. Chem. Rev. 110, 3315–3336 (2010).
Schreiber, S., Kapoor, T.M. & Wess, G. Chemical Biology: from Small Molecules to Systems Biology and Drug Design. (Wiley-VCH, 2007).
Ho, S.N., Biggar, S.R., Spencer, D.M., Schreiber, S.L. & Crabtree, G.R. Dimeric ligands define a role for transcriptional activation domains in reinitiation. Nature 382, 822–826 (1996).
Rivera, V.M. et al. A humanized system for pharmacologic control of gene expression. Nat. Med. 2, 1028–1032 (1996).
Spencer, D.M., Wandless, T.J., Schreiber, S.L. & Crabtree, G.R. Controlling signal transduction with synthetic ligands. Science 262, 1019–1024 (1993).
Komatsu, T. et al. Organelle-specific, rapid induction of molecular activities and membrane tethering. Nat. Methods 7, 206–208 (2010).
Korzeniowski, M.K., Manjarres, I.M., Varnai, P. & Balla, T. Activation of STIM1-Orai1 involves an intramolecular switching mechanism. Sci. Signal. 3, ra82 (2010).
Suh, B.C., Inoue, T., Meyer, T. & Hille, B. Rapid chemically induced changes of PtdIns(4,5)P2 gate KCNQ ion channels. Science 314, 1454–1457 (2006).
Ueno, T., Falkenburger, B.H., Pohlmeyer, C. & Inoue, T. Triggering actin comets versus membrane ruffles: distinctive effects of phosphoinositides on actin reorganization. Sci. Signal. 4, ra87 (2011).
Bayle, J.H. et al. Rapamycin analogs with differential binding specificity permit orthogonal control of protein activity. Chem. Biol. 13, 99–107 (2006).
Czlapinski, J.L. et al. Conditional glycosylation in eukaryotic cells using a biocompatible chemical inducer of dimerization. J. Am. Chem. Soc. 130, 13186–13187 (2008).
Liang, F.S., Ho, W.Q. & Crabtree, G.R. Engineering the ABA plant stress pathway for regulation of induced proximity. Sci. Signal. 4, rs2 (2011).
Liberles, S.D., Diver, S.T., Austin, D.J. & Schreiber, S.L. Inducible gene expression and protein translocation using nontoxic ligands identified by a mammalian three-hybrid screen. Proc. Natl. Acad. Sci. USA 94, 7825–7830 (1997).
Seelig, G., Soloveichik, D., Zhang, D.Y. & Winfree, E. Enzyme-free nucleic acid logic circuits. Science 314, 1585–1588 (2006).
Stojanovic, M.N. Molecular computing with deoxyribozymes. Prog. Nucleic Acid Res. Mol. Biol. 82, 199–217 (2008).
Yoshida, W. & Yokobayashi, Y. Photonic Boolean logic gates based on DNA aptamers. Chem. Commun. (Camb.) 195–197 (2007).
Katz, E. & Privman, V. Enzyme-based logic systems for information processing. Chem. Soc. Rev. 39, 1835–1857 (2010).
Qian, L. & Winfree, E. A simple DNA gate motif for synthesizing large-scale circuits. J. R. Soc. Interface 8, 1281–1297 (2011).
Rackham, O. & Chin, J.W. Cellular logic with orthogonal ribosomes. J. Am. Chem. Soc. 127, 17584–17585 (2005).
Rinaudo, K. et al. A universal RNAi-based logic evaluator that operates in mammalian cells. Nat. Biotechnol. 25, 795–801 (2007).
Anderson, J.C., Voigt, C.A. & Arkin, A.P. Environmental signal integration by a modular AND gate. Mol. Syst. Biol. 3, 133 (2007).
Guet, C.C., Elowitz, M.B., Hsing, W. & Leibler, S. Combinatorial synthesis of genetic networks. Science 296, 1466–1470 (2002).
Mayo, A.E., Setty, Y., Shavit, S., Zaslaver, A. & Alon, U. Plasticity of the cis-regulatory input function of a gene. PLoS Biol. 4, e45 (2006).
Bronson, J.E., Mazur, W.W. & Cornish, V.W. Transcription factor logic using chemical complementation. Mol. Biosyst. 4, 56–58 (2008).
Santner, A. & Estelle, M. Recent advances and emerging trends in plant hormone signalling. Nature 459, 1071–1078 (2009).
Ueguchi-Tanaka, M. et al. Gibberellin insenstitive DWARF1 encodes a soluble receptor for gibberellin. Nature 437, 693–698 (2005).
Hirano, K., Ueguchi-Tanaka, M. & Matsuoka, M. GID1-mediated gibberellin signaling in plants. Trends Plant Sci. 13, 192–199 (2008).
Ueguchi-Tanaka, M. et al. Molecular interactions of a soluble gibberellin receptor, GID1, with a rice DELLA protein, SLR1, and gibberellin. Plant Cell 19, 2140–2155 (2007).
Tsien, R.Y. A non-disruptive technique for loading calcium buffers and indicators into cells. Nature 290, 527–528 (1981).
Nishimura, K., Fukagawa, T., Takisawa, H., Kakimoto, T. & Kanemaki, M. An auxin-based degron system for the rapid depletion of proteins in nonplant cells. Nat. Methods 6, 917–922 (2009).
Griffiths, J. et al. Genetic characterization and functional analysis of the GID1 gibberellin receptors in Arabidopsis. Plant Cell 18, 3399–3414 (2006).
Hirano, K. et al. Characterization of the molecular mechanism underlying gibberellin perception complex formation in rice. Plant Cell 22, 2680–2696 (2010).
Inoue, T., Heo, W.D., Grimley, J.S., Wandless, T.J. & Meyer, T. An inducible translocation strategy to rapidly activate and inhibit small GTPase signaling pathways. Nat. Methods 2, 415–418 (2005).
Murase, K., Hirano, Y., Sun, T.P. & Hakoshima, T. Gibberellin-induced DELLA recognition by the gibberellin receptor GID1. Nature 456, 459–463 (2008).
Shimada, A. et al. Structural basis for gibberellin recognition by its receptor GID1. Nature 456, 520–523 (2008).
Malgaroli, A., Milani, D., Meldolesi, J. & Pozzan, T. Fura-2 measurement of cytosolic free Ca2+ in monolayers and suspensions of various types of animal cells. J. Cell Biol. 105, 2145–2155 (1987).
Swanson, S.J. & Jones, R.L. Gibberellic acid induces vacuolar acidification in barley aleurone. Plant Cell 8, 2211–2221 (1996).
Tojima, T. et al. Attractive axon guidance involves asymmetric membrane transport and exocytosis in the growth cone. Nat. Neurosci. 10, 58–66 (2007).
Regot, S. et al. Distributed biological computation with multicellular engineered networks. Nature 469, 207–211 (2011).
Tamsir, A., Tabor, J.J. & Voigt, C.A. Robust multicellular computing using genetically encoded NOR gates and chemical 'wires'. Nature 469, 212–215 (2011).
Ihara, S., Oka, T. & Fukui, Y. Direct binding of SWAP-70 to non-muscle actin is required for membrane ruffling. J. Cell Sci. 119, 500–507 (2006).
Acknowledgements
This study was supported in part by the US National Institutes of Health (NIH; GM092930 and DK090868 to T.I. and NS072241 to M.J.W), the US National Science Foundation (IOS-0641548 and MCB-0923723 to T.S.) and the National Center for Research Resources of the NIH and NIH Roadmap for Medical Research (UL1 RR 025005 to C.M. and D.J.M). T.U. is a recipient of a fellowship from the Japanese Society for the Promotion of Science. M.C. is a recipient of the Provost's Undergraduate Research Award.
Author information
Authors and Affiliations
Contributions
T.M., A.S., M.C. and T.I. generated DNA constructs and T.M., R.D., T.U., A.S. and T.I. carried out cell biology experiments. T.S. advised on design of the gibberellin system. T.M. conducted biochemical experiments under supervision of M.J.W. C.M. and D.J.M. synthesized GA3-AM and GA3-H. T.I. conceived ideas. R.D. and T.I. wrote the paper.
Corresponding author
Ethics declarations
Competing interests
There is a pending patent associated with the gibberellin-induced dimerization system.
Supplementary information
Supplementary Text and Figures
Supplementary Methods and Supplementary Results (PDF 1650 kb)
Supplementary Movie 1
Supplementary Movie 1 (Miyamoto and DeRose et al.) (AVI 11905 kb)
Rights and permissions
About this article
Cite this article
Miyamoto, T., DeRose, R., Suarez, A. et al. Rapid and orthogonal logic gating with a gibberellin-induced dimerization system. Nat Chem Biol 8, 465–470 (2012). https://doi.org/10.1038/nchembio.922
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchembio.922
This article is cited by
-
Engineered poly(A)-surrogates for translational regulation and therapeutic biocomputation in mammalian cells
Cell Research (2024)
-
Small molecule-nanobody conjugate induced proximity controls intracellular processes and modulates endogenous unligandable targets
Nature Communications (2023)
-
A simeprevir-inducible molecular switch for the control of cell and gene therapies
Nature Communications (2023)
-
Engineering receptors in the secretory pathway for orthogonal signalling control
Nature Communications (2022)
-
Mandipropamid as a chemical inducer of proximity for in vivo applications
Nature Chemical Biology (2022)