Abstract
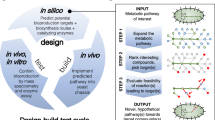
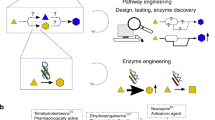
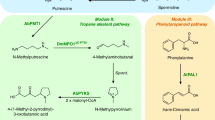
Numerous drugs and drug precursors in the current pharmacopoeia originate from plant sources. The limited yield of some bioactive compounds in plant tissues, however, presents a significant challenge for large-scale drug development. Metabolic engineering has facilitated the development of plant cell and tissue systems as alternative production platforms that can be scaled up in a controlled environment. Nevertheless, effective metabolic engineering approaches and the predictability of genetic transformations are often obscured due to the myriad cellular complexities. Progress in systems biology has aided the understanding of genome-wide interconnectivities in plant-based systems. In parallel, the bottom-up assembly of plant biosynthetic pathways in microorganisms demonstrated the possibilities of a new means of production. In this Perspective, we discuss the opportunities and challenges of implementing metabolic engineering in various platforms for the synthesis of natural and unnatural plant alkaloids.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Costantino, L. & Barlocco, D. Privileged structures as leads in medicinal chemistry. Curr. Med. Chem. 13, 65–85 (2006).
Evans, B.E. et al. Methods for drug discovery: development of potent, selective, orally effective cholecystokinin antagonists. J. Med. Chem. 31, 2235–2246 (1988).
Horton, D.A., Bourne, G.T. & Smythe, M.L. The combinatorial synthesis of bicyclic privileged structures or privileged substructures. Chem. Rev. 103, 893–930 (2003).
Maclean, D. et al. Glossary of terms used in combinatorial chemistry (technical report). J. Comb. Chem. 2, 562–578 (2000).
Koehn, F.E. & Carter, G.T. The evolving role of natural products in drug discovery. Nat. Rev. Drug Discov. 4, 206–220 (2005).
Maplestone, R.A., Stone, M.J. & Williams, D.H. The evolutionary role of secondary metabolites–a review. Gene 115, 151–157 (1992).
Williams, D.H., Stone, M.J., Hauck, P.R. & Rahman, S.K. Why are secondary metabolites (natural products) biosynthesized? J. Nat. Prod. 52, 1189–1208 (1989).
Newman, D.J. & Cragg, G.M. Natural products as sources of new drugs over the last 25 years. J. Nat. Prod. 70, 461–477 (2007).
Beghyn, T., Deprez-Poulain, R., Willand, N., Folleas, B. & Deprez, B. Natural compounds: leads or ideas? Bioinspired molecules for drug discovery. Chem. Biol. Drug Des. 72, 3–15 (2008).
Facchini, P.J. & De Luca, V. Opium poppy and Madagascar periwinkle: model non-model systems to investigate alkaloid biosynthesis in plants. Plant J. 54, 763–784 (2008).
Kutchan, T.M. Alkaloid biosynthesis—the basis for metabolic engineering of medicinal plants. Plant Cell 7, 1059–1070 (1995).
Verpoorte, R., van der Heijden, R. & Memelink, J. Engineering the plant cell factory for secondary metabolite production. Transgenic Res. 9, 323–343, discussion 321 (2000).
Hartmann, T. Plant-derived secondary metabolites as defensive chemicals in herbivorous insects: a case study in chemical ecology. Planta 219, 1–4 (2004).
Kuboyama, T., Yokoshima, S., Tokuyama, H. & Fukuyama, T. Stereocontrolled total synthesis of (+)-vincristine. Proc. Natl. Acad. Sci. USA 101, 11966–11970 (2004).
Miyazaki, T. et al. Synthesis of (+)-vinblastine and its analogues. Org. Lett. 9, 4737–4740 (2007).
Uchida, K., Yokoshima, S., Kan, T. & Fukuyama, T. Total synthesis of (+/−)-morphine. Org. Lett. 8, 5311–5313 (2006).
Yokoshima, S. et al. Stereocontrolled total synthesis of (+)-vinblastine. J. Am. Chem. Soc. 124, 2137–2139 (2002).
Liscombe, D.K. & Facchini, P.J. Evolutionary and cellular webs in benzylisoquinoline alkaloid biosynthesis. Curr. Opin. Biotechnol. 19, 173–180 (2008).
O'Connor, S.E. & Maresh, J.J. Chemistry and biology of monoterpene indole alkaloid biosynthesis. Nat. Prod. Rep. 23, 532–547 (2006).
Maresh, J.J. et al. Strictosidine synthase: mechanism of a Pictet-Spengler catalyzing enzyme. J. Am. Chem. Soc. 130, 710–723 (2008).
Sato, F. et al. Metabolic engineering of plant alkaloid biosynthesis. Proc. Natl. Acad. Sci. USA 98, 367–372 (2001).
Ashihara, H., Sano, H. & Crozier, A. Caffeine and related purine alkaloids: biosynthesis, catabolism, function and genetic engineering. Phytochemistry 69, 841–856 (2008).
Ogita, S., Uefuji, H., Yamaguchi, Y., Koizumi, N. & Sano, H. Producing decaffeinated coffee plants. Nature 423, 823 (2003).
Uefuji, H. et al. Caffeine production in tobacco plants by simultaneous expression of three coffee N-methyltrasferases and its potential as a pest repellant. Plant Mol. Biol. 59, 221–227 (2005).
Yun, D.J., Hashimoto, T. & Yamada, Y. Metabolic engineering of medicinal plants: transgenic Atropa belladonna with an improved alkaloid composition. Proc. Natl. Acad. Sci. USA 89, 11799–11803 (1992).
Larkin, P.J. et al. Increasing morphinan alkaloid production by over-expressing codeinone reductase in transgenic Papaver somniferum. Plant Biotechnol. J. 5, 26–37 (2007).
Allen, R.S. et al. RNAi-mediated replacement of morphine with the nonnarcotic alkaloid reticuline in opium poppy. Nat. Biotechnol. 22, 1559–1566 (2004).
Frick, S., Kramell, R. & Kutchan, T.M. Metabolic engineering with a morphine biosynthetic P450 in opium poppy surpasses breeding. Metab. Eng. 9, 169–176 (2007).
Ye, K. et al. Opium alkaloid noscapine is an antitumor agent that arrests metaphase and induces apoptosis in dividing cells. Proc. Natl. Acad. Sci. USA 95, 1601–1606 (1998).
Stafford, A.M., Pazoles, C.J., Siegel, S. & Yeh, L.-A. Plant Cell Culture: a Vehicle for Drug Delivery (John Wiley & Sons, New York, 1998).
Yukimune, Y., Tabata, H., Higashi, Y. & Hara, Y. Methyl jasmonate-induced overproduction of paclitaxel and baccatin III in Taxus cell suspension cultures. Nat. Biotechnol. 14, 1129–1132 (1996).
Witherup, K.M. et al. Taxus spp. needles contain amounts of taxol comparable to the bark of Taxus brevifolia: analysis and isolation. J. Nat. Prod. 53, 1249–1255 (1990).
Touno, K., Tamaoka, J., Ohashi, Y. & Shimomura, K. Ethylene induced shikonin biosynthesis in shoot culture of Lithospermum erythrorhizon. Plant Physiol. Biochem. 43, 101–105 (2005).
Gamborg, O.L. Plant tissue culture. Biotechnology. Milestones. In Vitro Cell. Dev. Biol. Plant 38, 84–92 (2002).
Filner, P., Varner, J.E. & Wray, J.L. Environmental or developmental changes cause many enzyme activities of higher plants to rise or fall. Science 165, 358–367 (1969).
Shanks, J.V. & Morgan, J. Plant 'hairy root' culture. Curr. Opin. Biotechnol. 10, 151–155 (1999).
De Luca, V. & St Pierre, B. The cell and developmental biology of alkaloid biosynthesis. Trends Plant Sci. 5, 168–173 (2000).
Vázquez-Flota, F.A., St-Pierre, B. & De Luca, V. Light activation of vindoline biosynthesis does not require cytomorphogenesis in Catharanthus roseus seedlings. Phytochemistry 55, 531–536 (2000).
Zhang, L. et al. Engineering tropane biosynthetic pathway in Hyoscyamus niger hairy root cultures. Proc. Natl. Acad. Sci. USA 101, 6786–6791 (2004).
Canel, C. et al. Effects of over-expression of strictosidine synthase and tryptophan decarboxylase on alkaloid production by cell cultures of Catharanthus roseus. Planta 205, 414–419 (1998).
Hughes, E.H., Hong, S.B., Gibson, S.I., Shanks, J.V. & San, K.Y. Expression of a feedback-resistant anthranilate synthase in Catharanthus roseus hairy roots provides evidence for tight regulation of terpenoid indole alkaloid levels. Biotechnol. Bioeng. 86, 718–727 (2004).
Hughes, E.H., Hong, S.B., Gibson, S.I., Shanks, J.V. & San, K.Y. Metabolic engineering of the indole pathway in Catharanthus roseus hairy roots and increased accumulation of tryptamine and serpentine. Metab. Eng. 6, 268–276 (2004).
Hong, S.B., Peebles, C.A., Shanks, J.V., San, K.Y. & Gibson, S.I. Expression of the Arabidopsis feedback-insensitive anthranilate synthase holoenzyme and tryptophan decarboxylase genes in Catharanthus roseus hairy roots. J. Biotechnol. 122, 28–38 (2006).
Whitmer, S., Canel, C., Hallard, D., Goncalves, C. & Verpoorte, R. Influence of precursor availability on alkaloid accumulation by transgenic cell line of Catharanthus roseus. Plant Physiol. 116, 853–857 (1998).
Whitmer, S., van der Heijden, R. & Verpoorte, R. Effect of precursor feeding on alkaloid accumulation by a tryptophan decarboxylase over-expressing transgenic cell line T22 of Catharanthus roseus. J. Biotechnol. 96, 193–203 (2002).
Morgan, J.A. & Shanks, J.V. Determination of metabolic rate-limitations by precursor feeding in Catharanthus roseus hairy root cultures. J. Biotechnol. 79, 137–145 (2000).
Magnotta, M., Murata, J., Chen, J. & De Luca, V. Expression of deacetylvindoline-4-O-acetyltransferase in Catharanthus roseus hairy roots. Phytochemistry 68, 1922–1931 (2007).
Schwender, J., Goffman, F., Ohlrogge, J.B. & Shachar-Hill, Y. Rubisco without the Calvin cycle improves the carbon efficiency of developing green seeds. Nature 432, 779–782 (2004).
Schwender, J., Ohlrogge, J. & Shachar-Hill, Y. Understanding flux in plant metabolic networks. Curr. Opin. Plant Biol. 7, 309–317 (2004).
Sriram, G., Fulton, D.B. & Shanks, J.V. Flux quantification in central carbon metabolism of Catharanthus roseus hairy roots by 13C labeling and comprehensive bondomer balancing. Phytochemistry 68, 2243–2257 (2007).
Sriram, G. & Shanks, J.V. Improvements in metabolic flux analysis using carbon bond labeling experiments: bondomer balancing and Boolean function mapping. Metab. Eng. 6, 116–132 (2004).
Rischer, H. et al. Gene-to-metabolite networks for terpenoid indole alkaloid biosynthesis in Catharanthus roseus cells. Proc. Natl. Acad. Sci. USA 103, 5614–5619 (2006).
van der Fits, L. & Memelink, J. ORCA3, a jasmonate-responsive transcriptional regulator of plant primary and secondary metabolism. Science 289, 295–297 (2000).
Menke, F.L., Champion, A., Kijne, J.W. & Memelink, J. A novel jasmonate- and elicitor-responsive element in the periwinkle secondary metabolite biosynthetic gene Str interacts with a jasmonate- and elicitor-inducible AP2-domain transcription factor, ORCA2. EMBO J. 18, 4455–4463 (1999).
Peebles, C.A., Hughes, E.H., Shanks, J.V. & San, K.Y. Transcriptional response of the terpenoid indole alkaloid pathway to the overexpression of ORCA3 along with jasmonic acid elicitation of Catharanthus roseus hairy roots over time. Metab. Eng. 11, 76–86 (2009).
Chen, S., Galan, M.C., Coltharp, C. & O'Connor, S.E. Redesign of a central enzyme in alkaloid biosynthesis. Chem. Biol. 13, 1137–1141 (2006).
O'Hagan, D. et al. Fluorinated tropane alkaloids generated by directed biosynthesis in transformed root cultures of Datura stramonium. J. Chem. Soc. [Perkin 1] 1, 2117–2120 (1999).
McCoy, E., Galan, M.C. & O'Connor, S.E. Substrate specificity of strictosidine synthase. Bioorg. Med. Chem. Lett. 16, 2475–2478 (2006).
McCoy, E. & O'Connor, S.E. Directed biosynthesis of alkaloid analogs in the medicinal plant Catharanthus roseus. J. Am. Chem. Soc. 128, 14276–14277 (2006).
Ma, X., Panjikar, S., Koepke, J., Loris, E. & Stockigt, J. The structure of Rauvolfia serpentina strictosidine synthase is a novel six-bladed beta-propeller fold in plant proteins. Plant Cell 18, 907–920 (2006).
Loris, E.A. et al. Structure-based engineering of strictosidine synthase: auxiliary for alkaloid libraries. Chem. Biol. 14, 979–985 (2007).
Bernhardt, P., McCoy, E. & O'Connor, S.E. Rapid identification of enzyme variants for reengineered alkaloid biosynthesis in periwinkle. Chem. Biol. 14, 888–897 (2007).
Runguphan, W. & O'Connor, S.E. Metabolic reprogramming of periwinkle plant culture. Nat. Chem. Biol. 5, 151–153 (2009).
Minami, H. et al. Microbial production of plant benzylisoquinoline alkaloids. Proc. Natl. Acad. Sci. USA 105, 7393–7398 (2008).
Hawkins, K.M. & Smolke, C.D. Production of benzylisoquinoline alkaloids in Saccharomyces cerevisiae. Nat. Chem. Biol. 4, 564–573 (2008).
Nevoigt, E. et al. Engineering of promoter replacement cassettes for fine-tuning of gene expression in Saccharomyces cerevisiae. Appl. Environ. Microbiol. 72, 5266–5273 (2006).
Geerlings, A. et al. Biotransformation of tryptamine and secologanin into plant terpenoid indole alkaloids by transgenic yeast. Appl. Microbiol. Biotechnol. 56, 420–424 (2001).
Chang, M.C., Eachus, R.A., Trieu, W., Ro, D.K. & Keasling, J.D. Engineering Escherichia coli for production of functionalized terpenoids using plant P450s. Nat. Chem. Biol. 3, 274–277 (2007).
Leonard, E. & Koffas, M.A. Engineering of artificial plant cytochrome P450 enzymes for synthesis of isoflavones by Escherichia coli. Appl. Environ. Microbiol. 73, 7246–7251 (2007).
Leonard, E., Lim, K.H., Saw, P.N. & Koffas, M.A. Engineering central metabolic pathways for high-level flavonoid production in Escherichia coli. Appl. Environ. Microbiol. 73, 3877–3886 (2007).
Leonard, E. et al. Strain improvement of recombinant Escherichia coli for efficient production of plant flavonoids. Mol. Pharm. 5, 257–265 (2008).
Ro, D.K. et al. Production of the antimalarial drug precursor artemisinic acid in engineered yeast. Nature 440, 940–943 (2006).
McCoy, E. & O'Connor, S.E. Natural products from plant cell cultures. Prog. Drug Res. 65, 329, 331–370 (2008).
Roberts, S.C. Production and engineering of terpenoids in plant cell culture. Nat. Chem. Biol. 3, 387–395 (2007).
St-Pierre, B., Vazquez-Flota, F.A. & De Luca, V. Multicellular compartmentation of catharanthus roseus alkaloid biosynthesis predicts intercellular translocation of a pathway intermediate. Plant Cell 11, 887–900 (1999).
Bhadra, R., Vani, S. & Shanks, J.V. Production of indole alkaloids by selected hairy root lines of Catharanthus roseus. Biotechnol. Bioeng. 41, 581–592 (1993).
Shanks, J.V., Bhadra, R., Morgan, J., Rijhwani, S. & Vani, S. Quantification of metabolites in the indole alkaloid pathways of Catharanthus roseus: implications for metabolic engineering. Biotechnol. Bioeng. 58, 333–338 (1998).
Morgan, J.A. & Shanks, J.V. Quantification of metabolic flux in plant secondary metabolism by a biogenetic organizational approach. Metab. Eng. 4, 257–262 (2002).
Rischer, H. & Oksman-Caldentey, K.M. Unintended effects in genetically modified crops: revealed by metabolomics? Trends Biotechnol. 24, 102–104 (2006).
Gantet, P. & Memelink, J. Transcription factors: tools to engineer the production of pharmacologically active plant metabolites. Trends Pharmacol. Sci. 23, 563–569 (2002).
Kizer, L., Pitera, D.J., Pfleger, B.F. & Keasling, J.D. Application of functional genomics to pathway optimization for increased isoprenoid production. Appl. Environ. Microbiol. 74, 3229–3241 (2008).
Alper, H., Moxley, J., Nevoigt, E., Fink, G.R. & Stephanopoulos, G. Engineering yeast transcription machinery for improved ethanol tolerance and production. Science 314, 1565–1568 (2006).
Pakrashi, S.C., Djerassi, C., Wasicky, R. & Neuss, N. Alkaloid studies. IX.1 Rauwolfia alkaloids. IV.2 Isolation of reserpine and other alkaloids from Rauwolfia sellowii muell. Argov.3. J. Am. Chem. Soc. 77, 6687–6689 (1955).
Kosalec, I. et al. Croatian barberry (Berberis croatica Horvat): a new source of berberine-analysis and antimicrobial activity. World J. Microbiol. Biotechnol. 25, 145–150 (2009).
Carvalho, A., Tango, J.S. & Monaco, L.C. Genetic control of caffeine in coffee. Nature 205, 314 (1965).
Wiedenfeld, H., Furmanowa, M., Roeder, E., Guzewska, J. & Gustowski, W. Camptothecin and 10-hydroxycamptothecin in callus and plantlets of Camptotheca acuminata. Plant Cell Tissue Organ Eng. 49, 213–218 (1997).
Johnson, E. & Emche, S. Variation of alkaloid content in Erythroxylum coca leaves from leaf bud to leaf drop. Ann. Bot. (Lond.) 73, 645–650 (1994).
Kalant, H. Opium revisited: a brief review of its nature, composition, non-medical use and relative risks. Addiction 92, 267–277 (1997).
Mateus, L., Cherkaoui, S., Christen, P. & Oksman-Caldentey, K.M. Simultaneous determination of scopolamine, hyoscyamine and littorine in plants and different hairy root clones of Hyoscyamus muticus by micellar electrokinetic chromatography. Phytochemistry 54, 517–523 (2000).
Saunders, J.W. & Bush, L.P. Nicotine biosynthetic enzyme activities in Nicotiana tabacum L. genotypes with different alkaloid levels. Plant Physiol. 64, 236–240 (1979).
Hagel, J.M., Weljie, A.M., Vogel, H.J. & Facchini, P.J. Quantitative 1H nuclear magnetic resonance metabolite profiling as a functional genomics platform to investigate alkaloid biosynthesis in opium poppy. Plant Physiol. 147, 1805–1821 (2008).
McCalley, D. Quantitative analysis of alkaloids from Cinchona bark by high-performance liquid chromatography. Analyst (Lond.) 115, 1355–1358 (1990).
Amer, M.A. & Court, W.E. Alkaloids of Rauwolfia nitida root bark. Phytochemistry 20, 2569–2573 (1981).
Baser, K.H., Bisset, N.G. & Hylands, P.J. Protostrychnine, a new alkaloid from Strychnos nux-vomica. Phytochemistry 18, 512–514 (1979).
Datta, A. & Srivastava, P.S. Variation in vinblastine production by Catharanthus roseus, during in vivo and in vitro differentiation. Phytochemistry 46, 135–137 (1997).
Chen, Q. et al. Analysis of yohimbine alkaloid from Pausinystalia yohimbe by non-aqueous capillary electrophoresis and gas chromatography-mass spectrometry. J. Sep. Sci. 31, 2211–2218 (2008).
Acknowledgements
The authors are grateful for the constructive discussion and contribution of M. Tackett (Whitehead Institute for Biomedical Research and Department of Biology, MIT). Research in the Prather laboratory is supported by the US National Science Foundation/Synthetic Biology Engineering Research Center (SynBERC; grant no. 0540879) and the MIT Energy Initiative (grant no. 6917278). Research in the O'Connor laboratory is funded by the US National Institutes of Health (GM074820) and the US National Science Foundation (MCB-0719120).
Author information
Rights and permissions
About this article
Cite this article
Leonard, E., Runguphan, W., O'Connor, S. et al. Opportunities in metabolic engineering to facilitate scalable alkaloid production. Nat Chem Biol 5, 292–300 (2009). https://doi.org/10.1038/nchembio.160
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchembio.160
This article is cited by
-
Mining cancer genomes for change-of-metabolic-function mutations
Communications Biology (2023)
-
Successful regeneration of fertile stably transformed tropane alkaloid-producing plant (Hyoscyamus muticus L.) with PVX-gus-(astr1 or astr2)-nptII constructs
Plant Cell, Tissue and Organ Culture (PCTOC) (2021)
-
Broadening the scope of biocatalytic C–C bond formation
Nature Reviews Chemistry (2020)
-
Molecular characterization and overexpression analyses of secologanin synthase to understand the regulation of camptothecin biosynthesis in Nothapodytes nimmoniana (Graham.) Mabb.
Protoplasma (2020)
-
Metabolic and transcriptional analyses in response to potent inhibitors establish MEP pathway as major route for camptothecin biosynthesis in Nothapodytes nimmoniana (Graham) Mabb
BMC Plant Biology (2019)