Abstract
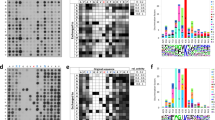
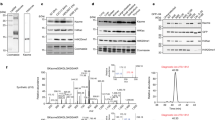
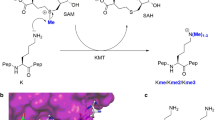
By methylation of peptide arrays, we determined the specificity profile of the protein methyltransferase G9a. We show that it mostly recognizes an Arg-Lys sequence and that its activity is inhibited by methylation of the arginine residue. Using the specificity profile, we identified new non-histone protein targets of G9a, including CDYL1, WIZ, ACINUS and G9a (automethylation), as well as peptides derived from CSB. We demonstrate potential downstream signaling pathways for methylation of non-histone proteins.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Kouzarides, T. Cell 128, 693–705 (2007).
Rea, S. et al. Nature 406, 593–599 (2000).
Cheng, X., Collins, R.E. & Zhang, X. Annu. Rev. Biophys. Biomol. Struct. 34, 267–294 (2005).
Tachibana, M. et al. Genes Dev. 16, 1779–1791 (2002).
Xin, Z. et al. J. Biol. Chem. 278, 14996–15000 (2003).
Ikegami, K. et al. Genes Cells 12, 1–11 (2007).
Tachibana, M. et al. Genes Dev. 19, 815–826 (2005).
Collins, R.E. et al. J. Biol. Chem. 280, 5563–5570 (2005).
Patnaik, D. et al. J. Biol. Chem. 279, 53248–53258 (2004).
Tachibana, M., Sugimoto, K., Fukushima, T. & Shinkai, Y. J. Biol. Chem. 276, 25309–25317 (2001).
Rathert, P., Zhang, X., Freund, C., Cheng, X. & Jeltsch, A. Chem. Biol. 15, 5–11 (2008).
Chin, H.G. et al. Biochemistry 44, 12998–13006 (2005).
Dacwag, C.S., Ohkawa, Y., Pal, S., Sif, S. & Imbalzano, A.N. Mol. Cell. Biol. 27, 384–394 (2007).
Kirmizis, A. et al. Nature 449, 928–932 (2007).
Guccione, E. et al. Nature 449, 933–937 (2007).
Sampath, S.C. et al. Mol. Cell 27, 596–608 (2007).
Jacobs, S.A. & Khorasanizadeh, S. Science 295, 2080–2083 (2002).
Frank, R. J. Immunol. Methods 267, 13–26 (2002).
Acknowledgements
The US National Institutes of Health (GM068680) and the Bundesministerium für Bildung und Forschung Biofuture program have supported this work. We thank M.T. Bedford (University of Texas M.D. Anderson Cancer Center) for the GST-HP1β expression construct and M. Yoshida (RIKEN) for technical advice on anti-pan methyllysine antibodies. Technical assistance by M. Schwerdtfeger (Jacobs University Bremen) is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–4, Supplementary Table 1, Supplementary Discussion and Supplementary Methods (PDF 421 kb)
Rights and permissions
About this article
Cite this article
Rathert, P., Dhayalan, A., Murakami, M. et al. Protein lysine methyltransferase G9a acts on non-histone targets. Nat Chem Biol 4, 344–346 (2008). https://doi.org/10.1038/nchembio.88
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchembio.88
This article is cited by
-
Comparison of partial vs. total ossicular chain reconstruction using titanium prosthesis: a retrospective cohort study
European Archives of Oto-Rhino-Laryngology (2023)
-
De novo methylation of histone H3K23 by the methyltransferases EHMT1/GLP and EHMT2/G9a
Epigenetics & Chromatin (2022)
-
Towards the understanding of the activity of G9a inhibitors: an activity landscape and molecular modeling approach
Journal of Computer-Aided Molecular Design (2020)
-
Monitoring the crosstalk between methylation and phosphorylation on histone peptides with host-assisted capillary electrophoresis
Analytical and Bioanalytical Chemistry (2020)
-
Structural basis for the target specificity of actin histidine methyltransferase SETD3
Nature Communications (2019)