Abstract
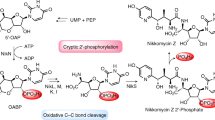
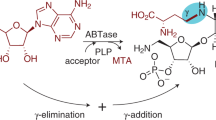
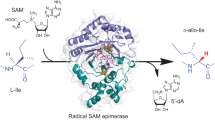
Nikkomycins and polyoxins are antifungal peptidylnucleoside antibiotics active against human and plant pathogens. Here we report that during peptidylnucleoside biosynthesis in Streptomyces cacaoi and S. tendae, the C5′ extension of the nucleoside essential for downstream structural diversification is catalyzed by a conserved radical S-adenosyl-L-methionine (SAM) enzyme, PolH or NikJ. This is distinct from the nucleophilic mechanism reported for antibacterial nucleosides and represents a new mechanism of nucleoside natural product biosynthesis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Isono, K. J. Antibiot. (Tokyo) 41, 1711–1739 (1988).
Munro, C.A. Adv. Appl. Microbiol. 83, 145–172 (2013).
Reuveni, M., Cohen, H., Zahavi, T. & Venezian, A. Crop Prot. 19, 393–399 (2000).
Shubitz, L.F. et al. J. Infect. Dis. 209, 1949–1954 (2014).
Isono, K., Sato, T., Hirasawa, K., Funayama, S. & Suzuki, S. J. Am. Chem. Soc. 100, 3937–3939 (1978).
Schüz, T.C., Fiedler, H.P., Zähner, H., Rieck, M. & Konig, W.A. J. Antibiot. (Tokyo) 45, 199–206 (1992).
Isono, K., Crain, P.F. & Mccloskey, J.A. J. Am. Chem. Soc. 97, 943–945 (1975).
Chen, W. et al. J. Biol. Chem. 284, 10627–10638 (2009).
Ginj, C., Rüegger, H., Amrhein, N. & Macheroux, P. ChemBioChem 6, 1974–1976 (2005).
Chen, W. et al. J. Ind. Microbiol. Biotechnol. 43, 401–417 (2016).
Sofia, H.J., Chen, G., Hetzler, B.G., Reyes-Spindola, J.F. & Miller, N.E. Nucleic Acids Res. 29, 1097–1106 (2001).
Frey, P.A. Acc. Chem. Res. 47, 540–549 (2014).
Oberdorfer, G., Binter, A., Ginj, C., Macheroux, P. & Gruber, K. J. Biol. Chem. 287, 31427–31436 (2012).
Zhang, Y. et al. Nature 465, 891–896 (2010).
Hover, B.M., Loksztejn, A., Ribeiro, A.A. & Yokoyama, K. J. Am. Chem. Soc. 135, 7019–7032 (2013).
Mahanta, N., Fedoseyenko, D., Dairi, T. & Begley, T.P. J. Am. Chem. Soc. 135, 15318–15321 (2013).
Broderick, J.B., Duffus, B.R., Duschene, K.S. & Shepard, E.M. Chem. Rev. 114, 4229–4317 (2014).
Kudo, F., Hoshi, S., Kawashima, T., Kamachi, T. & Eguchi, T. J. Am. Chem. Soc. 136, 13909–13915 (2014).
Wyszynski, F.J. et al. Nat. Chem. 4, 539–546 (2012).
Yang, Z. et al. J. Biol. Chem. 286, 7885–7892 (2011).
Barnard-Britson, S. et al. J. Am. Chem. Soc. 134, 18514–18517 (2012).
Hänzelmann, P. & Schindelin, H. Proc. Natl. Acad. Sci. USA 101, 12870–12875 (2004).
Edelhoch, H. Biochemistry 6, 1948–1954 (1967).
Fish, W.W. Methods Enzymol. 158, 357–364 (1988).
Beinert, H. Anal. Biochem. 131, 373–378 (1983).
Palmer, G. Methods Enzymol. 10, 594–609 (1967).
Stoll, S. & Schweiger, A. J. Magn. Reson. 178, 42–55 (2006).
Acknowledgements
We thank G.R. Dubay for assistance with the MS measurements. We thank A.A. Ribeiro at the Duke NMR center for the assistance in collecting NMR data. This work was supported by the Duke University Medical Center and National Institute of General Medical Sciences R01 GM115729 (to K.Y.). EPR spectrometer was supported by an Institutional Development Grant (ID 2014-IDG-1017) from the North Carolina Biotechnology Center.
Author information
Authors and Affiliations
Contributions
K.Y. conceived the project. K.Y. and E.A.L. designed the experiments. E.A.L. performed the experiments. K.Y. and E.A.L. analyzed the data and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Results, Supplementary Tables 1–11 and Supplementary Figures 1–31. (PDF 6129 kb)
Supplementary Note
Synthetic Procedures (PDF 311 kb)
Rights and permissions
About this article
Cite this article
Lilla, E., Yokoyama, K. Carbon extension in peptidylnucleoside biosynthesis by radical SAM enzymes. Nat Chem Biol 12, 905–907 (2016). https://doi.org/10.1038/nchembio.2187
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchembio.2187
This article is cited by
-
Cryptic phosphorylation in nucleoside natural product biosynthesis
Nature Chemical Biology (2021)
-
Recent advances in the biosynthesis of nucleoside antibiotics
The Journal of Antibiotics (2019)
-
Novel insights into nucleoamino acids: biomolecular recognition and aggregation studies of a thymine-conjugated l-phenyl alanine
Amino Acids (2018)
-
The antifungal pipeline: a reality check
Nature Reviews Drug Discovery (2017)
-
Biosynthesis and combinatorial biosynthesis of antifungal nucleoside antibiotics
Science China Life Sciences (2017)