Abstract
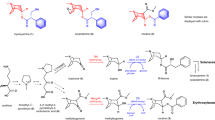
We have characterized four sequential enzymes that transform 1-hydroxy-N-methylcanadine to narcotoline hemiacetal, completing our elucidation of noscapine biosynthesis in opium poppy. Two cytochromes P450 catalyze hydroxylations at C13 and C8 on the protoberberine scaffold, the latter step inducing ring opening and the formation of an aldehyde moiety. Acetylation at C13 before C8 hydroxylation introduces a protective group subsequently hydrolyzed by a carboxylesterase, which triggers rearrangement to a cyclic hemiacetal.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Accession codes
References
Lettré, H. Ann. NY Acad. Sci. 58, 1264–1275 (1954).
Ye, K. et al. Proc. Natl. Acad. Sci. USA 95, 1601–1606 (1998).
Mahmoudian, M. & Rahimi-Moghaddam, P. Recent Pat. Anticancer Drug Discov. 4, 92–97 (2009).
Fang, Z.Z. et al. Br. J. Pharmacol. 167, 1271–1286 (2012).
Battersby, A.R. & Hirst, M. Tetrahedr. Lett. 11, 669–673 (1965).
Battersby, B.A.R., Staunton, J. & Wiltshire, H.R. J. Chem. Soc. Perkin Trans. I 1975, 1147–1156 (1975).
Dang, T.-T.T. & Facchini, P.J. Plant Physiol. 159, 618–631 (2012).
Winzer, T. et al. Science 336, 1704–1708 (2012).
Dang, T.-T.T. & Facchini, P.J. FEBS Lett. 588, 198–204 (2014).
Dang, T.-T.T. & Facchini, P.J. J. Biol. Chem. 289, 2013–2026 (2014).
Chen, X. & Facchini, P.J. Plant J. 77, 173–184 (2014).
Dinesh-Kumar, S.P., Anandalakshmi, R., Marathe, R., Schiff, M. & Liu, Y. Methods Mol. Biol. 236, 287–294 (2003).
D'Auria, J.C. Curr. Opin. Plant Biol. 9, 331–340 (2006).
Gershater, M.C. & Edwards, R. Plant Sci. 173, 579–588 (2007).
Ruppert, M. et al. Planta 222, 888–898 (2005).
Grothe, T., Lenz, R. & Kutchan, T.M. J. Biol. Chem. 276, 30717–30723 (2001).
Onoyovwe, A. et al. Plant Cell 25, 4110–4122 (2013).
McGary, K.L. et al. Proc. Natl. Acad. Sci. USA 110, 11481–11486 (2013).
Shulgin, A.T. & Perry, W.E. The Simple Plant Isoquinolines (Transform Press, Berkeley, CA, 2003).
Nguyen, D.T. et al. J. Biol. Chem. 285, 16588–16598 (2010).
Ro, D.-K. et al. BMC Biotechnol. 8, 83 (2008).
Dang, T.-T.T., Onoyovwe, A., Farrow, S.C. & Facchini, P.J. Methods Enzymol. 515, 231–266 (2012).
Hileman, L.C., Drea, S., Martino, G., Litt, A. & Irish, V.F. Plant J. 44, 334–341 (2005).
Acknowledgements
We thank D. Burns and J. Hagel for the NMR and LTQ-Orbitrap analyses, respectively. T.-T.T.D. is the recipient of the Alberta Innovates Technology Futures and University of Calgary Eyes High graduate scholarships. Funding was provided through grants from Genome Canada, Genome Alberta, the Government of Alberta (URSI-10-004-SEG), the Canada Foundation for Innovation (CFI-21695) and the Natural Sciences and Engineering Research Council of Canada (RGPIN/183573-2009) to P.J.F.
Author information
Authors and Affiliations
Contributions
T.-T.T.D. and X.C. contributed equally to all experiments. P.J.F. was responsible for experimental design. All authors contributed to the preparation of the manuscript.
Corresponding author
Ethics declarations
Competing interests
P.J.F., T.-T.T.D. and X.C. have filed a patent application (Patent Cooperation Treaty Application PCT/CA2014/050782) covering compositions and methods related to the manufacture of noscapine and synthesis intermediates thereof.
Supplementary information
Supplementary Text and Figures
Supplementary Results, Supplementary Figures 1–18 and Supplementary Tables 1–7. (PDF 20145 kb)
Supplementary Data Set 1
1H, 13C, COSY, HSQC, HMBC, and NOESY NMR spectral data for (13S,14R)-1,13-dihydroxy-N-methylcanadine (m/z 386) and 1H, COSY, HSQC and HMBC NMR spectral data for (13S,14R)-1-hydroxy-13-O-acetyl-N-methylcanadine (m/z 428). (PDF 898 kb)
Rights and permissions
About this article
Cite this article
Dang, TT., Chen, X. & Facchini, P. Acetylation serves as a protective group in noscapine biosynthesis in opium poppy. Nat Chem Biol 11, 104–106 (2015). https://doi.org/10.1038/nchembio.1717
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchembio.1717
This article is cited by
-
Plant cytochrome P450s directing monoterpene indole alkaloid (MIA) and benzylisoquinoline alkaloid (BIA) biosynthesis
Phytochemistry Reviews (2023)
-
Discovering and harnessing oxidative enzymes for chemoenzymatic synthesis and diversification of anticancer camptothecin analogues
Communications Chemistry (2021)
-
Gene clustering and copy number variation in alkaloid metabolic pathways of opium poppy
Nature Communications (2020)
-
High-throughput metabolomic and transcriptomic analyses vet the potential route of cerpegin biosynthesis in two varieties of Ceropegia bulbosa Roxb.
Planta (2020)
-
Transcriptomic profiles of 33 opium poppy samples in different tissues, growth phases, and cultivars
Scientific Data (2019)