Abstract
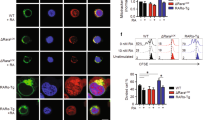
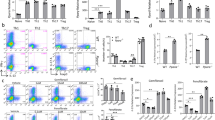
Retinoic acid receptor–related orphan receptor γ (RORγt) controls the differentiation of naive CD4+ T cells into the TH17 lineage, which are critical cells in the pathogenesis of autoimmune diseases. Here we report that during TH17 differentiation, cholesterol biosynthesis and uptake programs are induced, whereas their metabolism and efflux programs are suppressed. These changes result in the accumulation of the cholesterol precursor, desmosterol, which functions as a potent endogenous RORγ agonist. Generation of cholesterol precursors is essential for TH17 differentiation as blocking cholesterol synthesis with chemical inhibitors at steps before the formation of active precursors reduces differentiation. Upon activation, metabolic changes also lead to production of specific sterol-sulfate conjugates that favor activation of RORγ over the TH17-inhibiting sterol receptor LXR. Thus, TH17 differentiation is orchestrated by coordinated sterol synthesis, mobilization and metabolism to selectively activate RORγ.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Change history
15 July 2015
In the version of this article initially published, the zymosterol and zymostenol structures shown in Figure 3a were depicted with a double bond at C14-C15, where there should have been a single bond. The error has been corrected in the HTML and PDF versions of the article.
References
Yamane, H. & Paul, W.E. Early signaling events that underlie fate decisions of naive CD4+ T cells toward distinct T-helper cell subsets. Immunol. Rev. 252, 12–23 (2013).
Wang, R. & Green, D.R. Metabolic checkpoints in activated T cells. Nat. Immunol. 13, 907–915 (2012).
Jiang, S. & Dong, C. A complex issue on CD4+ T-cell subsets. Immunol. Rev. 252, 5–11 (2013).
Weaver, C.T., Hatton, R.D., Mangan, P.R. & Harrington, L.E. IL-17 family cytokines and the expanding diversity of effector T cell lineages. Annu. Rev. Immunol. 25, 821–852 (2007).
Zou, W. & Restifo, N.P.T. TH17 cells in tumour immunity and immunotherapy. Nat. Rev. Immunol. 10, 248–256 (2010).
Jones, S.A., Sutton, C.E., Cua, D. & Mills, K.H. Therapeutic potential of targeting IL-17. Nat. Immunol. 13, 1022–1025 (2012).
Ciofani, M. et al. A validated regulatory network for TH17 cell specification. Cell 151, 289–303 (2012).
Yosef, N. et al. Dynamic regulatory network controlling TH17 cell differentiation. Nature 496, 461–468 (2013).
Ivanov, I.I. et al. The orphan nuclear receptor RORγt directs the differentiation program of proinflammatory IL-17+ T helper cells. Cell 126, 1121–1133 (2006).
Manel, N., Unutmaz, D. & Littman, D.R. The differentiation of human TH-17 cells requires transforming growth factor-beta and induction of the nuclear receptor RORγt. Nat. Immunol. 9, 641–649 (2008).
Jin, L. et al. Structural basis for hydroxycholesterols as natural ligands of orphan nuclear receptor RORγ. Mol. Endocrinol. 24, 923–929 (2010).
Wang, Y. et al. Modulation of retinoic acid receptor-related orphan receptor α and gamma activity by 7-oxygenated sterol ligands. J. Biol. Chem. 285, 5013–5025 (2010).
Wang, Y., Kumar, N., Crumbley, C., Griffin, P.R. & Burris, T.P. A second class of nuclear receptors for oxysterols: Regulation of RORα and RORγ activity by 24S-hydroxycholesterol (cerebrosterol). Biochim. Biophys. Acta 1801, 917–923 (2010).
Spann, N.J. & Glass, C.K. Sterols and oxysterols in immune cell function. Nat. Immunol. 14, 893–900 (2013).
Bensinger, S.J. et al. LXR signaling couples sterol metabolism to proliferation in the acquired immune response. Cell 134, 97–111 (2008).
Armstrong, A.J., Gebre, A.K., Parks, J.S. & Hedrick, C.C. ATP-binding cassette transporter G1 negatively regulates thymocyte and peripheral lymphocyte proliferation. J. Immunol. 184, 173–183 (2010).
Cui, G. et al. Liver X receptor (LXR) mediates negative regulation of mouse and human TH17 differentiation. J. Clin. Invest. 121, 658–670 (2011).
Rathmell, J.C., Elstrom, R.L., Cinalli, R.M. & Thompson, C.B. Activated Akt promotes increased resting T cell size, CD28-independent T cell growth, and development of autoimmunity and lymphoma. Eur. J. Immunol. 33, 2223–2232 (2003).
Wilson, N.J. et al. Development, cytokine profile and function of human interleukin 17–producing helper T cells. Nat. Immunol. 8, 950–957 (2007).
Hakamada-Taguchi, R. et al. Inhibition of hydroxymethylglutaryl-coenzyme a reductase reduces TH1 development and promotes TH2 development. Circ. Res. 93, 948–956 (2003).
Zhang, X., Jin, J., Peng, X., Ramgolam, V.S. & Markovic-Plese, S. Simvastatin inhibits IL-17 secretion by targeting multiple IL-17–regulatory cytokines and by inhibiting the expression of IL-17 transcription factor RORC in CD4+ lymphocytes. J. Immunol. 180, 6988–6996 (2008).
Kagami, S. et al. Protein geranylgeranylation regulates the balance between TH17 cells and Foxp3+ regulatory T cells. Int. Immunol. 21, 679–689 (2009).
Ekins, S. et al. Three-dimensional quantitative structure-activity relationship analysis of human CYP51 inhibitors. Drug Metab. Dispos. 35, 493–500 (2007).
Solt, L.A. et al. Identification of a selective RORγ ligand that suppresses TH17 cells and stimulates T regulatory cells. ACS Chem. Biol. 7, 1515–1519 (2012).
Skepner, J. et al. Pharmacologic inhibition of RORγt regulates TH17 signature gene expression and suppresses cutaneous inflammation in vivo. J. Immunol. 192, 2564–2575 (2014).
Xu, T. et al. Ursolic acid suppresses interleukin-17 (IL-17) production by selectively antagonizing the function of RORγt protein. J. Biol. Chem. 286, 22707–22710 (2011).
Huh, J.R. et al. Digoxin and its derivatives suppress TH17 cell differentiation by antagonizing RORγt activity. Nature 472, 486–490 (2011).
Kassi, E. et al. Ursolic acid triggers apoptosis and Bcl-2 downregulation in MCF-7 breast cancer cells. Cancer Invest. 27, 723–733 (2009).
Yang, C. et al. Sterol intermediates from cholesterol biosynthetic pathway as liver X receptor ligands. J. Biol. Chem. 281, 27816–27826 (2006).
Bai, Q. et al. Sulfation of 25-hydroxycholesterol by SULT2B1b decreases cellular lipids via the LXR/SREBP-1c signaling pathway in human aortic endothelial cells. Atherosclerosis 214, 350–356 (2011).
Chen, W., Chen, G., Head, D.L., Mangelsdorf, D.J. & Russell, D.W. Enzymatic reduction of oxysterols impairs LXR signaling in cultured cells and the livers of mice. Cell Metab. 5, 73–79 (2007).
Ma, Y. et al. 25-Hydroxycholesterol-3-sulfate regulates macrophage lipid metabolism via the LXR/SREBP-1 signaling pathway. Am. J. Physiol. Endocrinol. Metab. 295, E1369–E1379 (2008).
Song, C., Hiipakka, R.A. & Liao, S. Auto-oxidized cholesterol sulfates are antagonistic ligands of liver X receptors: implications for the development and treatment of atherosclerosis. Steroids 66, 473–479 (2001).
Cook, I.T., Duniec-Dmuchowski, Z., Kocarek, T.A., Runge-Morris, M. & Falany, C.N. 24-hydroxycholesterol sulfation by human cytosolic sulfotransferases: formation of monosulfates and disulfates, molecular modeling, sulfatase sensitivity, and inhibition of liver X receptor activation. Drug Metab. Dispos. 37, 2069–2078 (2009).
Esplugues, E. et al. Control of TH17 cells occurs in the small intestine. Nature 475, 514–518 (2011).
Sutton, C.E. et al. Interleukin-1 and IL-23 induce innate IL-17 production from γΔ T cells, amplifying TH17 responses and autoimmunity. Immunity 31, 331–341 (2009).
Pantelyushin, S. et al. Rorγt+ innate lymphocytes and γΔ T cells initiate psoriasiform plaque formation in mice. J. Clin. Invest. 122, 2252–2256 (2012).
Hirayama, S. et al. Ezetimibe decreases serum oxidized cholesterol without impairing bile acid synthesis in Japanese hypercholesterolemic patients. Atherosclerosis 230, 48–51 (2013).
Brown, A.J. & Jessup, W. Oxysterols and atherosclerosis. Atherosclerosis 142, 1–28 (1999).
Ikonen, E. Cellular cholesterol trafficking and compartmentalization. Nat. Rev. Mol. Cell Biol. 9, 125–138 (2008).
Spann, N.J. et al. Regulated accumulation of desmosterol integrates macrophage lipid metabolism and inflammatory responses. Cell 151, 138–152 (2012).
Lim, H. et al. Proatherogenic conditions promote autoimmune T helper 17 cell responses in vivo. Immunity 40, 153–165 (2014).
Zhu, M., Hanyou, M., Dong, L., Xiaohong, L. & Lihua, Z. TH17/Treg imbalance induced by increased incidence of atherosclerosis in patients with systemic lupus erythematosus (SLE). Clin. Rheumatol. 32, 1045–1052 (2013).
Wu, S., Li, W.Q., Han, J., Sun, Q. & Qureshi, A.A. Hypercholesterolemia and risk of incident psoriasis and psoriatic arthritis in US women. Arthritis Rheumatol. 66, 304–310 (2014).
Kidani, Y. et al. Sterol regulatory element-binding proteins are essential for the metabolic programming of effector T cells and adaptive immunity. Nat. Immunol. 14, 489–499 (2013).
Zeng, H. et al. mTORC1 couples immune signals and metabolic programming to establish Treg cell function. Nature 499, 485–490 (2013).
Cua, D.J. & Tato, C.M. Innate IL-17–producing cells: the sentinels of the immune system. Nat. Rev. Immunol. 10, 479–489 (2010).
Cheng, H.Y. et al. Increased cholesterol content in γΔ T lymphocytes differentially regulates their activation. PLoS ONE 8, e63746 (2013).
Kallen, J., Schlaeppi, J.M., Bitsch, F., Delhon, I. & Fournier, B. Crystal structure of the human RORα ligand binding domain in complex with cholesterol sulfate at 2.2 Å. J. Biol. Chem. 279, 14033–14038 (2004).
Hanyu, O. et al. Cholesterol sulfate induces expression of the skin barrier protein filaggrin in normal human epidermal keratinocytes through induction of RORα. Biochem. Biophys. Res. Commun. 428, 99–104 (2012).
Acknowledgements
We thank X. Lu and D. Bousley for help on maintaining cells and mice and C. Van Huis for help on extraction with organic solvents. This work was supported in part by US National Institutes of Health grant AI-47450 to G.D.G.
Author information
Authors and Affiliations
Contributions
X.H. designed and conceptualized the research and wrote the manuscript. Y.W. performed ROR assays. Y.W. and X.H. conducted TH17 experiments. Y.W., L.-Y.H. and X.L. contributed to gene expression and flow cytometry analyses. J.M.W. analyzed sterol and sterol sulfate levels using LC/MS/MS. L.-Y.H., C.A.L. and B.M.S. contributed to in vivo experiments. R.W.M. established the ROR assays. T.D.A. provided structural insights. T.D.A., L.L.C., P.L.T. and G.D.G. provided constructive suggestions, helped analyze selected data and contributed to writing of the manuscript.
Corresponding author
Ethics declarations
Competing interests
All authors except J.M.W. are employees of Lycera Corp. J.M.W. is employed by Seventh Wave Laboratories.
Supplementary information
Supplementary Text and Figures
Supplementary Results and Supplementary Figures 1–6. (PDF 957 kb)
Rights and permissions
About this article
Cite this article
Hu, X., Wang, Y., Hao, LY. et al. Sterol metabolism controls TH17 differentiation by generating endogenous RORγ agonists. Nat Chem Biol 11, 141–147 (2015). https://doi.org/10.1038/nchembio.1714
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchembio.1714
This article is cited by
-
Shaping immune landscape of colorectal cancer by cholesterol metabolites
EMBO Molecular Medicine (2024)
-
Gut microbial metabolite deoxycholic acid facilitates Th17 differentiation through modulating cholesterol biosynthesis and participates in high-fat diet-associated colonic inflammation
Cell & Bioscience (2023)
-
Body mass index affects the association between plasma lipids and peripheral eosinophils in a general chinese population: a cross-sectional survey
Lipids in Health and Disease (2023)
-
Immunmodulation durch Ernährung bei kritisch kranken Patienten
Die Anaesthesiologie (2023)
-
CYP51-mediated cholesterol biosynthesis is required for the proliferation of CD4+ T cells in Sjogren’s syndrome
Clinical and Experimental Medicine (2022)