Abstract
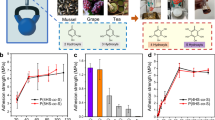
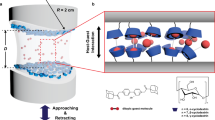
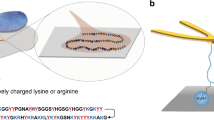
Cation–π interactions drive the self-assembly and cohesion of many biological molecules, including the adhesion proteins of several marine organisms. Although the origin of cation–π bonds in isolated pairs has been extensively studied, the energetics of cation–π-driven self-assembly in molecular films remains uncharted. Here we use nanoscale force measurements in combination with solid-state NMR spectroscopy to show that the cohesive properties of simple aromatic- and lysine-rich peptides rival those of the strong reversible intermolecular cohesion exhibited by adhesion proteins of marine mussel. In particular, we show that peptides incorporating the amino acid phenylalanine, a functional group that is conspicuously sparing in the sequences of mussel proteins, exhibit reversible adhesion interactions significantly exceeding that of analogous mussel-mimetic peptides. More broadly, we demonstrate that interfacial confinement fundamentally alters the energetics of cation–π-mediated assembly: an insight that should prove relevant for diverse areas, which range from rationalizing biological assembly to engineering peptide-based biomaterials.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Change history
08 June 2017
In the version of this Article originally published, the accept date was incorrect and should have read ‘9 December 2016’. This has now been corrected in the online versions of the Article.
References
Ma, J. C. & Dougherty, D. A. The cation–π interaction. Chem. Rev. 97, 1303–1324 (1997).
Gallivan, J. P. & Dougherty, D. A. Cation–π interactions in structural biology. Proc. Natl Acad. Sci. USA 96, 9459–9464 (1999).
Crowley, P. B. & Golovin, A. Cation–π interactions in protein–protein interfaces. Proteins. 59, 231–239 (2005).
Madahevi, A. S. & Sastry, G. N. Cation–π interaction: its role and relevance in chemistry, biology, and material science. Chem. Rev. 113, 2100–2138 (2013).
Zhong, W. et al. From ab initio quantum mechanics to molecular neurobiology: a cation–π binding site in the nicotinic receptor. Proc. Natl Acad. Sci. USA 95, 12088–12093 (1998).
Khademi, S. et al. Mechanism of ammonia transport by Amt/MEP/Rh: structure of AmtB at 1.35 Å. Science. 305, 1587–1594 (2004).
Meyer, E. A., Castellano, R. K. & Diederich, F. Interactions with aromatic rings in chemical and biological recognition. Angew. Chem. Int. Ed. 42, 1211–1250 (2003).
Hwang, D. S., Zeng, H., Lu, Q., Israelachvili, J. N. & Waite, J. H. Adhesion mechanism in a DOPA-deficient foot protein from green mussels. Soft Matter 8, 5640–5648 (2012).
Lu, Q. et al. Nanomechanics of cation–π interactions in aqueous solutions. Angew. Chem. 125, 4036–4040 (2013).
Kim, S. et al. Cation–π interaction in DOPA-deficient mussel adhesive protein mfp-1. J. Mater. Chem. B 3, 738–743 (2015).
Israelachvili, J. N. Intermolecular and Surface Forces Revised 3rd edn (Academic, 2011).
de Gennes, P. G. Soft adhesives. Langmuir 12, 4497–4500 (1996).
Rose, S. et al. Nanoparticle solutions as adhesives for gels and biological tissues. Nature 505, 382–385 (2014).
Sunner, J., Nishizawa, K. & Kebarle, P. Ion–solvent molecule interactions in the gas phase. The potassium ion and benzene. J. Phys. Chem. 85, 1814–1820 (1981).
Burley, S. K. & Petsko, G. A. Amino–aromatic interactions in proteins. FEBS Lett. 203, 139–143 (1986).
Deakyne, C. A. & Meot-Ner, M. Unconventional hydrogen bonds. 2. NH+–π complexes of onium ions with olefins and benzene derivatives. J. Am. Chem. Soc. 107, 474–479 (1985).
Lee, B. P., Messersmith, P. B., Israelachvili, J. N. & Waite, J. H. Mussel-inspired adhesives and coatings. Annu. Rev. Mater. Res. 41, 99–132 (2011).
Wong Po Foo, C. T. S., Lee, J. S., Mulyasasmita, W., Parisi-Amon, A. & Heilshorn, S. C. Two-component protein-engineered physical hydrogels for cell encapsulation. Proc. Natl Acad. Sci. USA 106, 22067–22072 (2009).
Norrby, P. & Liljefors, T. Strong decrease of the benzene–ammonium ion interaction upon complexation with a carboxylate anion. J. Am. Chem. Soc. 121, 2303–2306 (1999).
Bartoli, S. & Roelens, S. Binding of acetylcholine and tetramethylammonium to a cyclophane receptor: anion's contribution to the cation–π interaction. J. Am. Chem. Soc. 124, 8307–8315 (2002).
Hunter, C. A., Low, C. M. R., Rotger, C., Vinter, J. G. & Cristiano, Z. The role of the counterion in the cation–π interaction. Chem. Commun. 834–835 (2003).
Carrazana-García, J. A., Rodríguez-Otero, J. & Cabaleiro-Lago, E. M. A computational study of anion-modulated cation–π interactions. J. Phys. Chem. B 116, 5860–5871 (2012).
Carrazana-García, J. A., Cabaleiro-Lago, E. M., Campo-Caharrón, A. & Rodríguez-Otero, J. A theoretical study of ternary indole-cation-anion complexes. Org. Biomol. Chem. 12, 9145–9156 (2014).
Shao, H. & Stewart, R. J. Biomimetic underwater adhesives with environmentally triggered setting mechanisms. Adv. Mater. 22, 729–733 (2010).
Kamino, K., Nakano, M. & Kanai, S. Significance of the conformation of building blocks in curing of barnacle underwater adhesive. FEBS J. 279, 1750–1760 (2012).
Yamamoto, H. Synthesis and adhesive studies of marine polypeptides. J. Chem. Soc. Perkin Trans. 1 613–618 (1987).
Yu, M. & Deming, T. J. Synthetic polypeptide mimics of marine adhesives. Macromolecules 31, 4739–4745 (1998).
Mattson, K. M. et al. A facile synthesis of catechol-functionalized poly(ethylene oxide) block and random copolymers. J. Polymer Sci. A 53, 2685–2692 (2015).
Lee, H., Dellatore, S. M., Miller, W. M. & Messersmith, P. B. Mussel-inspired surface chemistry for multifunctional coatings. Science 318, 426–430 (2007).
Wang, J. et al. Influence of binding-site density in wet bioadhesion. Adv. Mater. 20, 3872–3876 (2008).
Wei, W. et al. Bridging adhesion of mussel-inspired peptides: role of charge, chain length, and surface type. Langmuir 31, 1105–1112 (2015).
Maier, G. P., Rapp, M. V., Waite, J. H., Israelachvili, J. N. & Butler, A. Adaptive synergy between catechol and lysine promotes wet adhesion by surface salt displacement. Science. 349, 628–632 (2015).
Danner, E. W., Kan, Y., Hammer, M. U., Israelachvili, J. N. & Waite, J. H. Adhesion of mussel foot protein mefp-5 to mica: an underwater superglue. Biochemistry 51, 6511–6518 (2012).
Luckham, P. F. & Klein, J. Forces between mica surfaces bearing adsorbed polyelectrolyte, poly-L-lysine, in aqueous media. J. Chem. Soc. Faraday Trans. 1 80, 865–878 (1984).
Israelachvili, J. N. et al. Recent advances in the surface forces apparatus (SFA) technique. Rep. Prog. Phys. 73, 036601 (2010).
Guo, C. & Holland, G. Investigating lysine adsorption on fumed silica nanoparticles. J. Phys. Chem. C 118, 25792–25801 (2014).
De Vita, E. & Frydman, L. Spectral editing in 13C MAS NMR under moderately fast spinning conditions. J. Magn. Reson. 148, 327–337 (2001).
Ando, S. et al. Conformational characterization of glycine residues incorporated into some homopolypeptides by solid-state 13C NMR spectroscopy. J. Am. Chem. Soc. 107, 7648–7652 (1985).
Selection of non-protonated carbon resonances in solid-state nuclear magnetic-resonance. J. Am. Chem. Soc. 101, 5854–5856 (1979).
Gomes, J. & Mallion, R. Aromaticity and ring currents. Chem. Rev. 101, 1349–1383 (2001).
Sever, M. J., Weisser, J. T., Monahan, J., Srinivasan, S. & Wilker, J. J. Metal-mediated cross-linking in the generation of a marine mussel adhesive. Angew. Chem. Int. Ed. 43, 448–450 (2004).
Holten-Andersen, N. et al. pH-induced metal–ligand cross-links inspired by mussel yield self-healing polymer networks with near-covalent elastic moduli. Proc. Natl Acad. Sci. USA 108, 2651–2655 (2011).
Yu, M., Hwang, J. & Deming, T. J. Role of L-3-4-dihydroxyphenylalanine in mussel adhesive proteins. J. Am. Chem. Soc. 121, 5825–5826 (1999).
Lee, H., Scherer, N. F. & Messersmith, P. B. Single-molecule mechanics of mussel adhesion. Proc. Natl Acad. Sci. USA 103, 12999–13003 (2006).
Martinez Rodriguez, N. R., Das, S., Kaufman, Y., Israelachvili, J. N. & Waite, J. H. Interfacial pH during mussel adhesive plaque formation. Biofouling 31, 221–227 (2015).
Liaqat, F. et al. High-performance TiO2 nanoparticle/DOPA polymer composites. Macromol. Rapid Commun. 36, 1129–1137 (2015).
Guardingo, M. et al. Bioinspired catechol-terminated self-assembled monolayers with enhanced adhesion properties. Small 10, 1594–1602 (2014).
Hediger, S., Meier, B. H., Kurur, N. D., Bodenhausen, G. & Ernst, R. R. NMR cross polarization by adiabatic passage through the Hartmann–Hahn condition (APHH). Chem. Phys. Lett. 223, 283–288 (1994).
Elena, B., de Paëpe, G. & Emsley, L. Direct spectral optimisation of proton–proton homonuclear dipolar decoupling in solid-state NMR. Chem. Phys. Lett. 398, 532–538 (2004).
Marion, D. & Wüthrich, K. Application of phase sensitive two-dimensional correlated spectroscopy (COSY) for measurements of 1H–1H spin-spin coupling constants in proteins. Biochem. Biophys. Res. Commun. 113, 967–974 (1983).
Fung, B. M., Khitrin, A. K. & Ermolaev, K. An improved broadband decoupling sequence for liquid crystals and solids. J. Magn. Reson. 142, 97–101 (2000).
Hayashi, S. & Hayamizu, K. Chemical shift standards in high-resolution solid-state NMR (1) 13C, 29Si, and 1H nuclei. Bull. Chem. Soc. Jpn 64, 685–687 (1991).
Acknowledgements
This research was supported by the Materials Research Science and Engineering Centers Program of the National Science Foundation under Award no. DMR 1121053. The authors acknowledge A. Griffin for assistance in characterizing the peptide adsorption.
Author information
Authors and Affiliations
Contributions
M.A.G. and W.W. contributed equally to this work. M.A.G., W.W., J.H.W. and J.N.I. conceived the research. M.A.G., A.M.S. and T.R.C. performed and analysed the force–distance measurements, W.W. synthesized and purified the peptides, M.A.G., H.A.D. and M.I. performed the NMR measurements, H.A.D., M.I. and B.F.C. analysed the NMR results, M.A.G. wrote the paper. All of the authors interpreted the data, discussed the results and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary information (PDF 1224 kb)
Rights and permissions
About this article
Cite this article
Gebbie, M., Wei, W., Schrader, A. et al. Tuning underwater adhesion with cation–π interactions. Nature Chem 9, 473–479 (2017). https://doi.org/10.1038/nchem.2720
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchem.2720
This article is cited by
-
Cation-π interactions enabled water-stable perovskite X-ray flat mini-panel imager
Nature Communications (2024)
-
Bi-terminal fusion of intrinsically-disordered mussel foot protein fragments boosts mechanical strength for protein fibers
Nature Communications (2023)
-
Vibrio cholerae biofilms use modular adhesins with glycan-targeting and nonspecific surface binding domains for colonization
Nature Communications (2023)
-
Functional Materials and Innovative Strategies for Wearable Thermal Management Applications
Nano-Micro Letters (2023)
-
Hydrogen-bonds mediate liquid-liquid phase separation of mussel derived adhesive peptides
Nature Communications (2022)