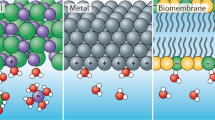
Abstract
The unique structural, dynamical and chemical properties of air/water and oil/water interfaces are thought to play a key role in various biological, geological and environmental processes. For example, non-hydrogen-bonded (‘dangling’) OH groups—which create surface defects in water's hydrogen bonding network and are experimentally detected at both macroscopic (air/water or oil/water) and microscopic (dissolved hydrophobic molecule) interfaces—are thought to catalyse some chemical reactions. However, how the size, curvature or charge of the exposed hydrophobic surface influences water's propensity to form dangling OH defects has not yet been established quantitatively. Here we use Raman multivariate curve resolution to probe spectroscopically the hydrophobic hydration shell and, using a statistical multisite analysis, we show that such interfacial dangling OH structures are entropically stabilized and their formation is cooperative (the probability that a non-hydrogen-bonded OH group will form depends nonlinearly on the hydrophobic surface area). We thus expose an important difference between the chemical properties of molecular and macroscopic oil/water interfaces.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Chandler, D. Physical chemistry – oil on troubled waters. Nature 445, 831–832 (2007).
Ball, P. Water as an active constituent in cell biology. Chem. Rev. 108, 74–108 (2008).
Patel, A. J. et al. Sitting at the edge: how biomolecules use hydrophobicity to tune their interactions and function. J. Phys. Chem. B 116, 2498–2503 (2012).
Du, Q., Freysz, E. & Shen, Y. R. Surface vibrational spectroscopic studies of hydrogen bonding and hydrophobicity. Science 264, 826–828 (1994).
Scatena, L. F., Brown, M. G. & Richmond, G. L. Water at hydrophobic surfaces: weak hydrogen bonding and strong orientation effects. Science 292, 908–912 (2001).
Ji, N., Ostroverkhov, V., Tian, C. S. & Shen, Y. R. Characterization of vibrational resonances of water–vapor interfaces by phase-sensitive sum-frequency spectroscopy. Phys. Rev. Lett. 100, 096102 (2008).
Levering, L. M., Sierra-Hernandez, M. R. & Allen, H. C. Observation of hydronium ions at the air–aqueous acid interface: vibrational spectroscopic studies of aqueous HCl, HBr, and HI. J. Phys. Chem. C 111, 8814–8826 (2007).
Moore, F. G. & Richmond, G. L. Integration or segregation: how do molecules behave at oil/water interfaces? Acc. Chem. Res. 41, 739–748 (2008).
Jung, Y. & Marcus, R. A. On the theory of organic catalysis on water. J. Am. Chem. Soc. 129, 5492–5502 (2007).
Jung, Y. S. & Marcus, R. A. Protruding interfacial OH groups and ‘on-water' heterogeneous catalysis. J. Phys. Condens. Matter 22, 284117 (2010).
Chanda, A. & Fokin, V. V. Organic synthesis ‘on water'. Chem. Rev. 109, 725–748 (2009).
Butler, R. N. & Coyne, A. G. Water: nature's reaction enforcer – comparative effects for organic synthesis ‘in-water’ and ‘on-water’. Chem. Rev. 110, 6302–6337 (2010).
Perera, P. N. et al. Observation of water dangling OH bonds around dissolved nonpolar groups. Proc. Natl Acad. Sci. USA 106, 12230–12234 (2009).
Davis, J. G., Gierszal, K. P., Wang, P. & Ben-Amotz, D. Water structural transformation at molecular hydrophobic interfaces. Nature 491, 582–585 (2012).
Tomlinson-Phillips, J. et al. Structure and dynamics of water dangling OH bonds in hydrophobic hydration shells. Comparison of simulation and experiment. J. Phys. Chem. A 115, 6177–6183 (2011).
Eaves, J. D. et al. Hydrogen bonds in liquid water are broken only fleetingly. Proc. Natl Acad. Sci. USA 102, 13019–13022 (2005).
Laage, D. & Hynes, J. T. A molecular jump mechanism of water reorientation. Science 311, 832–835 (2006).
Laage, D., Stirnemann, G. & Hynes, J. T. Why water reorientation slows without iceberg formation around hydrophobic solutes. J. Phys. Chem. B 113, 2428–2435 (2009).
Zheng, Y. Y. & Zhang, J. P. Catalysis in the oil droplet/water interface for aromatic Claisen rearrangement. J. Phys. Chem. A 114, 4325–4333 (2010).
Freier, E., Wolf, S. & Gerwert, K. Proton transfer via a transient linear water-molecule chain in a membrane protein. Proc. Natl Acad. Sci. USA 108, 11435–11439 (2011).
Furutani, Y. et al. Dynamics of dangling bonds of water molecules in pharaonis halorhodopsin during chloride ion transportation. J. Phys. Chem. Lett. 3, 2964–2969 (2012).
Rankin, B. M. et al. Interactions between halide anions and a molecular hydrophobic interface. Faraday Discuss. 160, 255–270 (2013).
Perera, P., Wyche, M., Loethen, Y. & Ben-Amotz, D. Solute-induced perturbations of solvent-shell molecules observed using multivariate Raman curve resolution. J. Am. Chem. Soc. 130, 4576–4579 (2008).
Perera, P. N., Browder, B. & Ben-Amotz, D. Perturbations of water by alkali halide ions measured using multivariate Raman curve resolution. J. Phys. Chem. B 113, 1805–1809 (2009).
Gierszal, K. P. et al. π-Hydrogen bonding in liquid water. J. Phys. Chem. Lett. 2, 2930–2933 (2011).
Fega, K. R., Wilcox, D. S. & Ben-Amotz, D. Application of Raman multivariate curve resolution to solvation-shell spectroscopy. Appl. Spectrosc. 66, 282–288 (2012).
Lum, K., Chandler, D. & Weeks, J. D. Hydrophobicity at small and large length scales. J. Phys. Chem. B 103, 4570–4577 (1999).
Chandler, D. Interfaces and the driving force of hydrophobic assembly. Nature 437, 640–647 (2005).
Lawton, W. H. & Sylvestre, E. A. Self modeling curve resolution. Technometrics 13, 617–633 (1971).
Huang, N. D. et al. Microscopic probing of the size dependence in hydrophobic solvation. J. Chem. Phys. 136, 074507 (2012).
Slipchenko, L. V. & Gordon, M. S. Water–benzene interactions: an effective fragment potential and correlated quantum chemistry study. J. Phys. Chem. A 113, 2092–2102 (2009).
Rosenfeld, D. E., Kwak, K., Gengeliczki, Z. & Fayer, M. D. Hydrogen bond migration between molecular sites observed with ultrafast 2D IR chemical exchange spectroscopy. J. Phys. Chem. B 114, 2383–2389 (2010).
Rosenfeld, D. E., Gengeliczki, Z. & Fayer, M. D. Solvent control of the soft angular potential in hydroxyl–π hydrogen bonds: inertial orientational dynamics. J. Phys. Chem. B 113, 13300–13307 (2009).
Li, I. T. S. & Walker, G. C. Signature of hydrophobic hydration in a single polymer. Proc. Natl Acad. Sci. USA 108, 16527–16532 (2011).
Nelson, N., Walder, R. & Schwartz, D. K. Single molecule dynamics on hydrophobic self-assembled monolayers. Langmuir 28, 12108–12113 (2012).
Collins, M. D., Hummer, G., Quillin, M. L., Matthews, B. W. & Gruner, S. M. Cooperative water filling of a nonpolar protein cavity observed by high-pressure crystallography and simulation. Proc. Natl Acad. Sci. USA 102, 16668–16671 (2005).
Kuna, J. J. et al. The effect of nanometre-scale structure on interfacial energy. Nature Mater. 8, 837–842 (2009).
Luzar, A. & Leung, K. Dynamics of capillary evaporation. I. Effect of morphology of hydrophobic surfaces. J. Chem. Phys. 113, 5836–5844 (2000).
Giovambattista, N., Debenedetti, P. G. & Rossky, P. J. Hydration behavior under confinement by nanoscale surfaces with patterned hydrophobicity and hydrophilicity. J. Phys. Chem. C 111, 1323–1332 (2007).
Acharya, H., Vembanur, S., Jamadagni, S. N. & Garde, S. Mapping hydrophobicity at the nanoscale: applications to heterogeneous surfaces and proteins. Faraday Discuss. 146, 1–13 (2010).
Jamadagni, S. N., Godawat, R. & Garde, S. in Annual Review of Chemical and Biomolecular Engineering Vol. 2 (ed. J. M. Prausnitz) 147–171 (Annual Reviews, 2011).
Jana, B., Pal, S. & Bagchi, B. Enhanced tetrahedral ordering of water molecules in minor grooves of DNA: relative role of DNA rigidity, nanoconfinement, and surface specific interactions. J. Phys. Chem. B 114, 3633–3638 (2010).
Hummer, G. Molecular binding under water's influence. Nature Chem. 2, 906–907 (2010).
Setny, P., Baron, R. & McCammon, J. A. How can hydrophobic association be enthalpy driven? J. Chem. Theory Comput. 6, 2866–2871 (2010).
Baron, R., Setny, P. & McCammon, J. A. Water in cavity-ligand recognition. J. Am. Chem. Soc. 132, 12091–12097 (2010).
Abel, R., Young, T., Farid, R., Berne, B. J. & Friesner, R. A. Role of the active-site solvent in the thermodynamics of factor Xa ligand binding. J. Am. Chem. Soc. 130, 2817–2831 (2008).
Corcelli, S. A. & Skinner, J. L. Infrared and Raman line shapes of dilute HOD in liquid H2O and D2O from 10 to 90 °C. J. Phys. Chem. A 109, 6154–6165 (2005).
Abe, N. & Ito, M. Effects of hydrogen-bonding on the Raman intensities of methanol, ethanol and water. J. Raman Spectrosc. 7, 161–167 (1978).
Wu, D. Y. et al. Theoretical study of binding interactions and vibrational Raman spectra of water in hydrogen-bonded anionic complexes: (H2O)n− (n = 2 and 3), H2O···X− (X=F, Cl, Br and I), and H2O···M− (M=Cu, Ag, and Au). J. Phys. Chem. A 112, 1313–1321 (2008).
Acknowledgements
This work was supported by the National Science Foundation (CHE-1213338). The authors thank Lyudmila Slipchenko for sharing the results of her QM-MM dimer calculations, as well as Jieke Chen for his assistance in collecting experimental results in this work.
Author information
Authors and Affiliations
Contributions
J.G.D., B.M.R. and K.P.G performed the experimental measurement, J.G.D. and B.M.R. contributed to the analysis of the experimental results, J.G.D. wrote the first draft of the manuscript and D.B-A. conceived the experimental and statistical analysis strategy, and wrote the final draft of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary information (PDF 332 kb)
Rights and permissions
About this article
Cite this article
Davis, J., Rankin, B., Gierszal, K. et al. On the cooperative formation of non-hydrogen-bonded water at molecular hydrophobic interfaces. Nature Chem 5, 796–802 (2013). https://doi.org/10.1038/nchem.1716
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchem.1716
This article is cited by
-
Nonthermal acceleration of protein hydration by sub-terahertz irradiation
Nature Communications (2023)
-
Isotope effects observed in diluted D2O/H2O mixtures identify HOD-induced low-density structures in D2O but not H2O
Scientific Reports (2022)
-
Motion of water monomers reveals a kinetic barrier to ice nucleation on graphene
Nature Communications (2021)
-
Plasmon-Activated Water can Prolong Existing Sea-Ice Habitats to Potentially Save Polar Bears
Scientific Reports (2019)
-
Direct visualization of both DNA and RNA quadruplexes in human cells via an uncommon spectroscopic method
Scientific Reports (2016)