Abstract
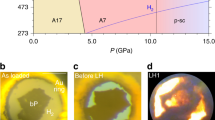
Closed electron shell systems, such as hydrogen, nitrogen or group 18 elements, can form weakly bound stoichiometric compounds at high pressures. An understanding of the stability of these van der Waals compounds is lacking, as is information on the nature of their interatomic interactions. We describe the formation of a stable compound in the Xe–H2 binary system, revealed by a suite of X-ray diffraction and optical spectroscopy measurements. At 4.8 GPa, a unique hydrogen-rich structure forms that can be viewed as a tripled solid hydrogen lattice modulated by layers of xenon, consisting of xenon dimers. Varying the applied pressure tunes the Xe–Xe distances in the solid over a broad range from that of an expanded xenon lattice to the distances observed in metallic xenon at megabar pressures. Infrared and Raman spectra indicate a weakening of the intramolecular covalent bond as well as persistence of semiconducting behaviour in the compound to at least 255 GPa.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Crabtree, G. W., Dresselhaus, M. S. & Buchanan, M. V. The hydrogen economy. Phys. Today 57, 39–44 (2004).
Hemley, R. J. Effects of high pressures on molecules. Ann. Rev. Phys. Chem. 51, 763–800 (2000).
Ashcroft, N. W. Metallic hydrogen: a high-temperature superconductor? Phys. Rev. Lett. 21, 1748–1749 (1968).
Ashcroft, N. W. Hydrogen dominant metallic alloys: high temperature superconductors? Phys. Rev. Lett. 92, 187002 (2004).
Carlsson, A. E. & Ashcroft, N. W. Approaches for reducing the insulator–metal transition pressure in hydrogen. Phys. Rev. Lett. 50, 1305–1308 (1983).
Goettel, K. A., Eggert, J. H. & Silvera, I. F. Optical evidence for the metallization of xenon at 132(5) GPa. Phys. Rev. Lett. 62, 665–668 (1989).
Reichlin, R. et al. Evidence for the insulator–metal transition in xenon from optical, X-ray and band-structure studies to 170 GPa. Phys. Rev. Lett. 62, 669–672 (1989).
Eremets, M. I., Gregoryanz, E. A., Struzhkin, V. V., Mao, H. K. & Hemley, R. J. Electrical conductivity of xenon at megabar pressures. Phys. Rev. Lett. 85, 2797–2800 (2000).
Khriachtchev, L., Lignell, A., Juselius, J., Rasanen, M. & Savchenko, E. Infrared absorption spectrum of matrix-isolated noble-gas hydride molecules: fingerprints of specific interactions and hindered rotation. J. Chem. Phys. 122, 14510–14517 (2005).
Khriachtchev, L., Pettersson, M., Runeberg, N., Lundell, J. & Rasanen, M. A stable argon compound. Nature 406, 874–876 (2000).
Grochala, W. Atypical compounds of gases, which have been called ‘noble’. Chem. Soc. Rev. 36, 1632–1655 (2007).
Wlodarczyk, A., McMillan, P. F. & Greenfield, S. A. High pressure effects in anaesthesia and narcosis. Chem. Soc. Rev. 35, 890–898 (2006).
Vos, W. L. et al. A high-pressure van der Waals compound in solid nitrogen–helium mixtures. Nature 358, 46–48 (1992).
Loubeyre, P., Jean-Louis, M., LeToullec, R. & Charon-Gérard, L. High pressure measurements of the He–Ne binary phase diagram at 296 K: evidence for the stability of a stoichiometric Ne(He)2 solid. Phys. Rev. Lett. 70, 178–181 (1993).
Hanni, H., Lantto, P., Runeberg, N., Jokisaari, J. & Vaara, J. Calculation of binary magnetic properties and potential energy curve in xenon dimer: second virial coefficient of 129Xe nuclear shielding. J. Chem. Phys. 121, 5908–5919 (2004).
Asaumi, K. High-pressure X-ray diffraction study of solid xenon and its equation of state in relation to metallization transition. Phys. Rev. B 29, 7026–7029 (1984).
Sears, D. R. & Harold, P. K. Density and expansivity of solid xenon. J. Chem. Phys. 37, 3002–3006 (1962).
Mao, H. K. & Hemley, R. J. Ultrahigh-pressure transitions in solid hydrogen. Rev. Mod. Phys. 66, 671–692 (1994).
Goncharov, A. F., Eggert, J. H., Mazin, I. I., Hemley, R. J. & Mao, H. K. Raman excitations and orientational ordering in deuterium at high pressure. Phys. Rev. B 54, R15590–R15593 (1996).
Loubeyre, P. et al. X-ray diffraction and equation of state of hydrogen at megabar pressures. Nature 383, 702–704 (1996).
LeBail, A. ESPOIR: a program for solving structures by Monte Carlo analysis of powder data. Mater. Sci. Forum 378–381, 65–70 (2001).
Brandenburg, K. & Putz, H. Crystal Impact GbR, ENDEAVOR 1.6 <http://www.crystalimpact.com/endeavor> (2008).
Proserpio, D. M., Hoffman, R. & Janda, K. C. The xenon–chlorine conundrum: van der Waals complex or linear molecule. J. Am. Chem. Soc. 113, 7184–7189 (1991).
Amarouche, M., Durand, G. & Malrieu, J. P. Structure and stability of Xen+ clusters. J. Chem. Phys. 88, 1010–1018 (1988).
Drews, T. & Seppelt, K. The Xe2+ ion—preparation and structure. Angew. Chem. Int. Ed. 36, 273–274 (1997).
Berry-Pusey, B. N., Anger, B. C., Laicher, G. & Saam, B. Nuclear spin relaxation of 129Xe due to persistent xenon dimers. Phys. Rev. A 74, 63408–63417 (2006).
Hanfland, M., Hemley, R. J., Mao, H. K. & Williams, G. P. Synchrotron infrared spectroscopy at megabar pressures: vibrational dynamics of hydrogen to 180 GPa. Phys. Rev. Lett. 69, 1129–1132 (1992).
Loubeyre, P., LeToullec, R. & Pinceaux, J. P. Raman measurements of the vibrational properties of H2 as a guest molecule in dense helium, neon, argon, and deuterium systems up to 40 GPa. Phys. Rev. B 45, 12844–12853 (1992).
Hemley, R. J., Mao, H.-K., Goncharov, A. F., Hanfland, M. & Struzhkin, V. Synchrotron infrared spectroscopy to 0.15 eV of H2 and D2 at megabar pressures. Phys. Rev. Lett. 76, 1667–1670 (1996).
Sheldrick, G. A short history of SHELX. Acta Cryst. Sec. A 64, 112–122 (2008).
Acknowledgements
The authors thank V. V. Struzhkin, G. Shen, Y. Meng and S. Sinogeikin for assistance and discussions. This work was supported by DOE-BES (DE-FG02-06ER46280), DOE-NNSA (CDAC), NSF-DMR (DMR-0805056), NSF-EAR (COMPRES) and the Balzan Foundation. A.P.S. is supported by DOE-BES under contract DE-AC02-06CH11357 and N.S.L.S. is supported by DOE-BES under contract no. DE-AC02-98CH10886.
Author information
Authors and Affiliations
Contributions
M.S. and R.J.H. designed the project. M.S., A.F.G. and S.A.G. conducted the sample loading, spectroscopic studies and analysis. M.S., P.D., P.L., W.Y. and H.K.M. conducted the synchrotron X-ray diffraction measurements and analysis. Z.L. performed the synchrotron IR measurements. M.S., P.D., R.J.H., A.F.G. and S.A.G. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary information (PDF 987 kb)
Rights and permissions
About this article
Cite this article
Somayazulu, M., Dera, P., Goncharov, A. et al. Pressure-induced bonding and compound formation in xenon–hydrogen solids. Nature Chem 2, 50–53 (2010). https://doi.org/10.1038/nchem.445
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchem.445
This article is cited by
-
Materials under high pressure: a chemical perspective
Applied Physics A (2022)
-
Xenon iron oxides predicted as potential Xe hosts in Earth’s lower mantle
Nature Communications (2020)
-
High pressure synthesis of phosphine from the elements and the discovery of the missing (PH3)2H2 tile
Nature Communications (2020)
-
Polyhydride CeH9 with an atomic-like hydrogen clathrate structure
Nature Communications (2019)
-
Synthesis of clathrate cerium superhydride CeH9 at 80-100 GPa with atomic hydrogen sublattice
Nature Communications (2019)