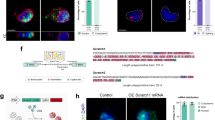
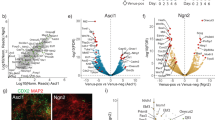
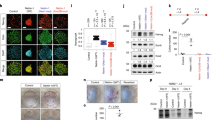
Abstract
Nuclear proteins are selectively imported into the nucleus by transport factors such as importin-α and importin-β1,2. Here, we show that the expression of importin-α subtypes is strictly regulated during neural differentiation of mouse embryonic stem (ES) cells, and that the switching of importin-α subtype expression is critical for neural differentiation. Moreover, reproducing the switching of importin-α subtype expression in undifferentiated ES cells induced neural differentiation in the presence of leukaemia inhibitory factor (LIF) and serum, coordinated with the regulated expression of Oct3/4, Brn2 and SOX2, which are involved in ES–neural identity determination. These transcription factors were selectively imported into the nucleus by specific subtypes of importin-α. Thus, importin-α subtype switching has a major impact on cell differentiation through the regulated nuclear import of a specific set of transcription factors. This is the first study to propose that transport factors should be considered as major players in cell-fate determination.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Görlich, D. & Mattaj, I. W. Nucleocytoplasmic transport. Science 271, 1513–1518 (1996).
Goldfarb, D. S. et al. Importin-α: a multipurpose nuclear-transport receptor. Trends Cell Biol. 14, 505–514 (2004).
Yoneda, Y. Nucleocytoplasmic protein traffic and its significance to cell function. Genes Cells 5, 777–787 (2000).
Köhler, M. et al. Evidence for distinct substrate specificities of importin-α family members in nuclear protein import. Mol. Cell. Biol. 11, 7782–7791 (1999).
Kawasaki, H. et al. Induction of midbrain dopaminergic neurons from ES cells by stromal cell-derived inducing activity. Neuron 28, 31–40 (2000).
Ying, Q. L. et al. Conversion of embryonic stem cells into neuroectodermal precursors in adherent monoculture. Nature Biotechnol. 2, 183–186 (2003).
Tanaka, S. et al. Interplay of SOX and POU factors in regulation of the Nestin gene in neural primordial cells. Mol. Cell Biol. 24, 8834–8846 (2004).
Kamachi, Y., Uchikawa, M. & Kondoh, H. Pairing SOX off: with partners in the regulation of embryonic development. Trends Genet. 16, 182–187 (2000).
Yuan, H., Corbi, N., Basilico, C. & Dailey, L. Developmental-specific activity of the FGF-4 enhancer requires the synergistic action of Sox2 and Oct-3. Genes Dev. 9, 2635–2645 (1995).
Chew, J. L. et. al. Reciprocal transcriptional regulation of Pou5f1 and Sox2 via the Oct4/Sox2 complex in embryonic stem cells. Mol Cell Biol. 25, 6031–6046 (2005).
Rodda, D. J. et. al. Transcriptional regulation of nanog by OCT4 and SOX2. J. Biol. Chem. 280, 24731–24737 (2005).
Kuroda, T. et. al. Octamer and Sox elements are required for transcriptional cis regulation of Nanog gene expression. Mol. Cell Biol. 25, 2475–2485 (2005).
Pan, G. et al. Identification of a nuclear localization signal in OCT4 and generation of a dominant negative mutant by its ablation. J. Biol. Chem. 279, 37013–37020 (2004).
Sock, E., Enderich, J., Rosenfeld, M. G. & Wegner, M. Identification of the nuclear localization signal of the POU domain protein Tst-1/Oct6. J. Biol. Chem. 271, 17512–17528 (1996).
Argentaro, A. et al. A SOX9 defect of calmodulin-dependent nuclear import in campomelic dysplasia/autosomal sex reversal. J. Biol. Chem. 278, 33839–33847 (2003).
Baranek, C., Sock, E. & Wegner, M. The POU protein Oct-6 is a nucleocytoplasmic shuttling protein. Nucleic Acids Res. 33, 6277–6286 (2005).
Ilia, M., Bazigou, E. & Price, J. Expression of the POU domain transcription factor, Oct-6, is attenuated in the adult mouse telencephalon, but increased by neurotoxic damage. Exp. Neurol. 181, 159–169 (2003).
Okumura-Nakanishi, S., Saito, M., Niwa, H. & Ishikawa, F. Oct-3/4 and Sox2 regulate Oct-3/4 gene in embryonic stem cells. J. Biol. Chem. 280, 5307–5317 (2005).
Tomioka, M. et al. Identification of Sox-2 regulatory region which is under the control of Oct-3/4-Sox-2 complex. Nucleic Acids Res. 30, 3202–3213 (2002).
Boyer, L. A. et al. Core transcriptional regulatory circuitry in human embryonic stem cells. Cell. 122, 947–956 (2005).
Niwa, H., Miyazaki, J. & Smith, A. G. Quantitative expression of Oct3/4 defines differentiation or self-renewal of ES cells. Nature Genet. 24, 372–376 (2000).
Rudnicki, M. A. & McBurney, M. W. in Teratocarcinomas and Embryonic Stem Cells: A partial Approach. (ed. Robertson E. J.) 19–49 (IRL Press, Oxford, 1987).
Kamikubo, Y. et al. Specific monoclonal antibody against nuclear import factor, importin-α1/Rch1. Hybrid Hybridomics 5, 301–304 (2004).
Sakaguchi, T. et al. Generation of a rat monoclonal antibody specific for importin-α 3/Qip1. Hybrid Hybridomics. 6, 397–400 (2003).
Imamoto, N. et al. Antibodies against 70-kD heat shock cognate protein inhibit mediated nuclear import of karyophilic proteins J. Cell Biol. 119, 1047–1061 (1992).
Yoneda, Y., Imamoto-Sonobe, N., Yamaizumi, M. & Uchida, T. Reversible inhibition of protein import into the nucleus by wheat germ agglutinin injected into cultured cells. Exp. Cell Res. 173, 586–595 (1987).
Adam, S. A., Marr, R. S. & Gerace, L. Nuclear protein import in permeabilized mammalian cells requires soluble cytoplasmic factors. J. Cell Biol. 111, 807–816 (1990).
Acknowledgements
We thank H. Niwa and T. Tachibana for providing the ES cell line and antibodies, A. G. Smith for the suggestion of using spreading culture conditions, and M. Raff, S. Nagata and T. Nakano for helpful comments. We also thank Y. Yoshida and T. Yamashita for help during the experiments. This work was supported by grants from the Ministry of Education, Culture, Sports, Science, and Technology of Japan and the Human Frontier Science Program.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary figures S1, S2, S3, S4 and Supplementary Table S1 (PDF 749 kb)
Rights and permissions
About this article
Cite this article
Yasuhara, N., Shibazaki, N., Tanaka, S. et al. Triggering neural differentiation of ES cells by subtype switching of importin-α. Nat Cell Biol 9, 72–79 (2007). https://doi.org/10.1038/ncb1521
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb1521
This article is cited by
-
Nucleocytoplasmic transport of active HER2 causes fractional escape from the DCIS-like state
Nature Communications (2023)
-
Structural basis for nuclear import selectivity of pioneer transcription factor SOX2
Nature Communications (2021)
-
Human retinal organoids release extracellular vesicles that regulate gene expression in target human retinal progenitor cells
Scientific Reports (2021)
-
Biological importance of OCT transcription factors in reprogramming and development
Experimental & Molecular Medicine (2021)
-
Disease-specific alteration of karyopherin-α subtype establishes feed-forward oncogenic signaling in head and neck squamous cell carcinoma
Oncogene (2020)