Abstract
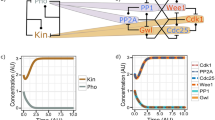
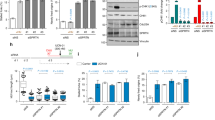
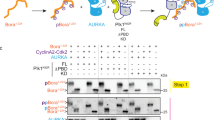
In vertebrates, the checkpoint-regulatory kinase Chk1 mediates cell-cycle arrest in response to a block in DNA replication or to DNA damaged by ultraviolet radiation. The activation of Chk1 depends on both Claspin and the upstream regulatory kinase ATR. Claspin is a large acidic protein that becomes phosphorylated and binds to Chk1 in the presence of checkpoint-inducing DNA templates in Xenopus egg extracts. Here we identify, by means of deletion analysis, a region of Claspin of 57 amino acids that is both necessary and sufficient for binding to Xenopus Chk1. This Chk1-binding domain contains two highly conserved repeats of approximately ten amino acids. A serine residue in each repeat (serine 864 and serine 895) undergoes phosphorylation during a checkpoint response. A mutant of Claspin containing non-phosphorylatable amino acids at positions 864 and 895 cannot bind to Chk1 and is unable to mediate its activation. Our results indicate that two phosphopeptide motifs in Claspin are essential for checkpoint signalling.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Melo, J. & Toczyski, D. Curr. Opin. Cell Biol. 14, 237–245 (2002).
Guo, Z., Kumagai, A., Wang, S.X. & Dunphy, W.G. Genes Dev. 14, 2745–2756 (2000).
Hekmat-Nejad, M., You, Z., Yee, M., Newport, J.W. & Cimprich, K.A. Curr. Biol. 10, 1565–1573 (2000).
Liu, Q. et al. Genes Dev. 14, 1448–1459 (2000).
Zhao, H. & Piwnica-Worms, H. Mol. Cell. Biol. 21, 4129–4139 (2001).
Peng, C.Y. et al. Science 277, 1501–1505 (1997).
Sanchez, Y. et al. Science 277, 1497–1501 (1997).
Furnari, B., Rhind, N. & Russell, P. Science 277, 1495–1497 (1997).
Kumagai, A., Guo, Z., Emami, K.H., Wang, S.X. & Dunphy, W.G. J. Cell Biol. 142, 1559–1569 (1998).
Lee, J., Kumagai, A. & Dunphy, W.G. Mol. Biol. Cell 12, 551–563 (2001).
Kumagai, A. & Dunphy, W.G. Mol. Cell 6, 839–849 (2000).
Alcasabas, A.A. et al. Nature Cell Biol. 3, 958–965 (2001).
Tanaka, K. & Russell, P. Nature Cell Biol. 3, 966–972 (2001).
Sun, Z., Hsiao, J., Fay, D.S. & Stern, D.F. Science 281, 272–274 (1998).
de la Torre-Ruiz, M.A., Green, C.M. & Lowndes, N.F. EMBO J. 17, 2687–2698 (1998).
Gilbert, C.S., Green, C.M. & Lowndes, N.F. Mol. Cell 8, 129–136 (2001).
Saka, Y., Esashi, F., Matsusaka, T., Mochida, S. & Yanagida, M. Genes Dev. 11, 3387–3400 (1997).
Willson, J., Wilson, S., Warr, N. & Watts, F.Z. Nucleic Acids Res. 25, 2138–2146 (1997).
Longhese, M.P., Foiani, M., Muzi-Falconi, M., Lucchini, G. & Plevani, P. EMBO J. 17, 5525–5528 (1998).
Pawson, T. & Nash, P. Genes Dev. 14, 1027–1047 (2000).
Yaffe, M.B. & Elia, A.E. Curr. Opin. Cell Biol. 13, 131–138 (2001).
Durocher, D., Henckel, J., Fersht, A.R. & Jackson, S.P. Mol. Cell 4, 387–394 (1999).
Emili, A. Mol. Cell 2, 183–189 (1998).
Vialard, J.E., Gilbert, C.S., Green, C.M. & Lowndes, N.F. EMBO J. 17, 5679–5688 (1998).
Huse, M. & Kuriyan, J. Cell 109, 275–282 (2002).
Hunter, T. Cell 100, 113–127 (2000).
Shiloh, Y. & Kastan, M.B. Adv. Cancer Res. 83, 209–254 (2001).
Abraham, R.T. Genes Dev. 15, 2177–2196 (2001).
Acknowledgements
We thank T. Pogodin for technical assistance, S. Horvath for phosphopeptide synthesis, and members of the laboratory for comments on the manuscript. This work was supported in part by a grant from the NIH. W.G.D. is an investigator of the Howard Hughes Medical Institute.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Figures
Figure S1 Assay for identification of the CKBD. (PDF 533 kb)
Figure S2 Ser-864 and Ser-895 in the CKBD are phosphorylated in egg extracts.
Figure S3 Xatr regulates phosphorylation of Claspin and binding of Claspin to Xchk1 in egg extracts.
Rights and permissions
About this article
Cite this article
Kumagai, A., Dunphy, W. Repeated phosphopeptide motifs in Claspin mediate the regulated binding of Chk1. Nat Cell Biol 5, 161–165 (2003). https://doi.org/10.1038/ncb921
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb921
This article is cited by
-
A multi-omics analysis reveals CLSPN is associated with prognosis, immune microenvironment and drug resistance in cancers
Biological Procedures Online (2023)
-
Essential role of CK2α for the interaction and stability of replication fork factors during DNA synthesis and activation of the S-phase checkpoint
Cellular and Molecular Life Sciences (2022)
-
Mrc1/Claspin: a new role for regulation of origin firing
Current Genetics (2017)
-
Claspin recruits Cdc7 kinase for initiation of DNA replication in human cells
Nature Communications (2016)
-
Exploiting replicative stress to treat cancer
Nature Reviews Drug Discovery (2015)