Abstract
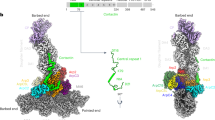
The Arp2/3 complex consists of seven evolutionarily conserved subunits (Arp2, Arp3 and ARPC1–5) and plays an essential role in generating branched actin filament networks during many different cellular processes. In mammals, however, the ARPC1 and ARPC5 subunits are each encoded by two isoforms that are 67% identical. This raises the possibility that Arp2/3 complexes with different properties may exist. We found that Arp2/3 complexes containing ARPC1B and ARPC5L are significantly better at promoting actin assembly than those with ARPC1A and ARPC5, both in cells and in vitro. Branched actin networks induced by complexes containing ARPC1B or ARPC5L are also disassembled ∼2-fold slower than those formed by their counterparts. This difference reflects the ability of cortactin to stabilize ARPC1B- and ARPC5L- but not ARPC1A- and ARPC5-containing complexes against coronin-mediated disassembly. Our observations demonstrate that the Arp2/3 complex in higher eukaryotes is actually a family of complexes with different properties.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Machesky, L. M., Atkinson, S. J., Ampe, C., Vandekerckhove, J. & Pollard, T. D. Purification of a cortical complex containing two unconventional actins from Acanthamoeba by affinity chromatography on profilin-agarose. J. Cell Biol. 127, 107–115 (1994).
Beltzner, C. C. & Pollard, T. D. Identification of functionally important residues of Arp2/3 complex by analysis of homology models from diverse species. J. Mol. Biol. 336, 551–565 (2004).
Goley, E. D. & Welch, M. D. The ARP2/3 complex: an actin nucleator comes of age. Nat. Rev. Mol. Cell Biol. 7, 713–726 (2006).
Svitkina, T. M. & Borisy, G. G. Arp2/3 complex and actin depolymerizing factor/cofilin in dendritic organization and treadmilling of actin filament array in lamellipodia. J. Cell Biol. 145, 1009–1026 (1999).
Vinzenz, M. et al. Actin branching in the initiation and maintenance of lamellipodia. J. Cell Sci. 125, 2775–2785 (2012).
Mullins, D. R., Heuser, J. A. & Pollard, T. D. The interaction of Arp2/3 complex with actin: nucleation, high affinity pointed end capping, and formation of branching networks of filaments. Proc. Natl Acad. Sci. USA 95, 6181–6186 (1998).
Blanchoin, L. et al. Direct observation of dendritic actin filament networks nucleated by Arp2/3 complex and WASP/Scar proteins. Nature 404, 1007–1011 (2000).
Welch, M. D., Rosenblatt, J., Skoble, J., Portnoy, D. A. & Mitchison, T. J. Interaction of human Arp2/3 complex and the Listeria monocytogenes ActA protein in actin filament nucleation. Science 281, 105–108 (1998).
Welch, M. D. & Way, M. Arp2/3-mediated actin-based motility: a tail of pathogen abuse. Cell Host Microbe 14, 242–255 (2013).
Rottner, K., Hanisch, J. & Campellone, K. G. WASH, WHAMM and JMY: regulation of Arp2/3 complex and beyond. Trends Cell Biol. 20, 650–661 (2010).
Campellone, K. G. & Welch, M. D. A nucleator arms race: cellular control of actin assembly. Nat. Rev. Mol. Cell Biol. 11, 237–251 (2010).
Egile, C. et al. Activation of the Cdc42 effector N-WASP by the Shigella flexneri IcsA protein promotes actin nucleation by Arp2/3 complex and bacterial actin-based motility. J. Cell Biol. 146, 1319–1332 (1999).
Le Clainche, C., Didry, D., Carlier, M. F. & Pantaloni, D. Activation of Arp2/3 complex by Wiskott-Aldrich Syndrome protein is linked to enhanced binding of ATP to Arp2. J. Biol. Chem. 276, 46689–46692 (2001).
Marchand, J. B., Kaiser, D. A., Pollard, T. D. & Higgs, H. N. Interaction of WASP/Scar proteins with actin and vertebrate Arp2/3 complex. Nat. Cell Biol. 3, 76–82 (2001).
Panchal, S. C., Kaiser, D. A., Torres, E., Pollard, T. D. & Rosen, M. K. A conserved amphipathic helix in WASP/Scar proteins is essential for activation of Arp2/3 complex. Nat. Struct. Biol. 10, 591–598 (2003).
Chereau, D. et al. Actin-bound structures of Wiskott-Aldrich syndrome protein (WASP)-homology domain 2 and the implications for filament assembly. Proc. Natl Acad. Sci. USA 102, 16644–16649 (2005).
Co, C., Wong, D. T., Gierke, S., Chang, V. & Taunton, J. Mechanism of actin network attachment to moving membranes: barbed end capture by N-WASP WH2 domains. Cell 128, 901–913 (2007).
Weed, S. A. et al. Cortactin localization to sites of actin assembly in lamellipodia requires interactions with F-actin and the Arp2/3 complex. J. Cell Biol. 151, 29–40 (2000).
Uruno, T. et al. Activation of Arp2/3 complex-mediated actin polymerization by cortactin. Nat. Cell Biol. 3, 259–266 (2001).
Weaver, A. M. et al. Cortactin promotes and stabilizes Arp2/3-induced actin filament network formation. Curr. Biol. 11, 370–374 (2001).
Kirkbride, K. C., Sung, B. H., Sinha, S. & Weaver, A. M. Cortactin: a multifunctional regulator of cellular invasiveness. Cell Adhes. Migr. 5, 187–198 (2011).
Helgeson, L. A. & Nolen, B. J. Mechanism of synergistic activation of Arp2/3 complex by cortactin and N-WASP. eLife 2, e00884 (2013).
Egile, C. et al. Mechanism of filament nucleation and branch stability revealed by the structure of the Arp2/3 complex at actin branch junctions. PLoS Biol. 3, e383 (2005).
Siton, O. et al. Cortactin releases the brakes in actin-based motility by enhancing WASP-VCA detachment from Arp2/3 branches. Curr. Biol. 21, 2092–2097 (2011).
Helgeson, L. A., Prendergast, J. G., Wagner, A. R., Rodnick-Smith, M. & Nolen, B. J. Interactions with actin monomers, actin filaments, and Arp2/3 complex define the roles of WASP family proteins and cortactin in coordinately regulating branched actin networks. J. Biol. Chem. 289, 28856–28869 (2014).
Cai, L., Makhov, A. M., Schafer, D. A. & Bear, J. E. Coronin 1B antagonizes cortactin and remodels Arp2/3-containing actin branches in lamellipodia. Cell 134, 828–842 (2008).
Chan, K. T., Creed, S. J. & Bear, J. E. Unraveling the enigma: progress towards understanding the coronin family of actin regulators. Trends Cell Biol. 21, 481–488 (2011).
Cai, L., Marshall, T. W., Uetrecht, A. C., Schafer, D. A. & Bear, J. E. Coronin 1B coordinates Arp2/3 complex and cofilin activities at the leading edge. Cell 128, 915–929 (2007).
Jansen, S. et al. Single-molecule imaging of a three-component ordered actin disassembly mechanism. Nat. Commun. 6, 7202 (2015).
Jay, P. et al. ARP3β, the gene encoding a new human actin-related protein, is alternatively spliced and predominantly expressed in brain neuronal cells. Eur. J. Biochem. 267, 2921–2928 (2000).
Balasubramanian, M. K., Feoktistova, A., McCollum, D. & Gould, K. L. Fission yeast Sop2p: a novel and evolutionarily conserved protein that interacts with Arp3p and modulates profilin function. EMBO J. 15, 6426–6437 (1996).
Millard, T. H., Behrendt, B., Launay, S., Futterer, K. & Machesky, L. M. Identification and characterisation of a novel human isoform of Arp2/3 complex subunit p16-ARC/ARPC5. Cell Motil. Cytoskeleton 54, 81–90 (2003).
Robinson, R. C. et al. Crystal structure of Arp2/3 complex. Science 294, 1679–1684 (2001).
Steffen, A. et al. Filopodia formation in the absence of functional WAVE- and Arp2/3-complexes. Mol. Biol. Cell 17, 2581–2591 (2006).
Suraneni, P. et al. The Arp2/3 complex is required for lamellipodia extension and directional fibroblast cell migration. J. Cell Biol. 197, 239–251 (2012).
Frischknecht, F. et al. Actin based motility of vaccinia virus mimics receptor tyrosine kinase signalling. Nature 401, 926–929 (1999).
Snapper, S. B. et al. N-WASP deficiency reveals distinct pathways for cell surface projections and microbial actin-based motility. Nat. Cell Biol. 3, 897–904 (2001).
Weisswange, I., Newsome, T. P., Schleich, S. & Way, M. The rate of N-WASP exchange limits the extent of ARP2/3-complex-dependent actin-based motility. Nature 458, 87–91 (2009).
Humphries, A. C. et al. Clathrin potentiates vaccinia-induced actin polymerization to facilitate viral spread. Cell Host Microbe 12, 346–359 (2012).
Donnelly, S. K., Weisswange, I., Zettl, M. & Way, M. WIP provides an essential link between Nck and N-WASP during Arp2/3-dependent actin polymerization. Curr. Biol. 23, 999–1006 (2013).
Humphries, A. C., Donnelly, S. K. & Way, M. Cdc42 and the Rho GEF intersectin-1 collaborate with Nck to promote N-WASP-dependent actin polymerisation. J. Cell Sci. 127, 673–685 (2014).
Le Clainche, C., Pantaloni, D. & Carlier, M. F. ATP hydrolysis on actin-related protein 2/3 complex causes debranching of dendritic actin arrays. Proc. Natl Acad. Sci. USA 100, 6337–6342 (2003).
Rosenblatt, J., Agnew, B. J., Abe, H., Bamburg, J. R. & Mitchison, T. J. Xenopus actin depolymerizing factor/cofilin (XAC) is responsible for the turnover of actin filaments in Listeria monocytogenes tails. J. Cell Biol. 136, 1323–1332 (1997).
Carlier, M.-F. et al. Actin depolymerizing factor (ADF/cofilin) enhances the rate of filament turnover: implication in actin-based motility. J. Cell Biol. 136, 1307–1323 (1997).
Brieher, W. M., Kueh, H. Y., Ballif, B. A. & Mitchison, T. J. Rapid actin monomer-insensitive depolymerization of Listeria actin comet tails by cofilin, coronin, and Aip1. J. Cell Biol. 175, 315–324 (2006).
Mueller, J. et al. Electron tomography and simulation of baculovirus actin comet tails support a tethered filament model of pathogen propulsion. PLoS Biol. 12, e1001765 (2014).
Xu, X. P. et al. Three-dimensional reconstructions of Arp2/3 complex with bound nucleation promoting factors. EMBO J. 31, 236–247 (2012).
Frischknecht, F. & Way, M. Surfing pathogens and the lessons learned for actin polymerization. Trends Cell Biol. 11, 30–38 (2001).
Lai, F. P. et al. Arp2/3 complex interactions and actin network turnover in lamellipodia. EMBO J. 27, 982–992 (2008).
Boczkowska, M. et al. X-ray scattering study of activated Arp2/3 complex with bound actin-WCA. Structure 16, 695–704 (2008).
Padrick, S. B., Doolittle, L. K., Brautigam, C. A., King, D. S. & Rosen, M. K. Arp2/3 complex is bound and activated by two WASP proteins. Proc. Natl Acad. Sci. USA 108, E472–E479 (2011).
Ti, S. C., Jurgenson, C. T., Nolen, B. J. & Pollard, T. D. Structural and biochemical characterization of two binding sites for nucleation-promoting factor WASp-VCA on Arp2/3 complex. Proc. Natl Acad. Sci. USA 108, E463–E471 (2011).
Boczkowska, M., Rebowski, G., Kast, D. J. & Dominguez, R. Structural analysis of the transitional state of Arp2/3 complex activation by two actin-bound WCAs. Nature Commun. 5, 3308 (2014).
Zalevsky, J., Lempert, L., Kranitz, H. & Mullins, R. D. Different WASP family proteins stimulate different Arp2/3 complex-dependent actin-nucleating activities. Curr. Biol. 11, 1903–1913 (2001).
Volkmann, N. et al. Structure of Arp2/3 complex in its activated state and in actin filament branch junctions. Science 293, 2456–2459 (2001).
Rouiller, I. et al. The structural basis of actin filament branching by the Arp2/3 complex. J. Cell Biol. 180, 887–895 (2008).
Uruno, T., Liu, J., Li, Y., Smith, N. & Zhan, X. Sequential interaction of actin-related proteins 2 and 3 (Arp2/3) complex with neural Wiscott-Aldrich syndrome protein (N-WASP) and cortactin during branched actin filament network formation. J. Biol. Chem. 278, 26086–26093 (2003).
Smith, B. A. et al. Three-color single molecule imaging shows WASP detachment from Arp2/3 complex triggers actin filament branch formation. eLife 2, e01008 (2013).
Hiller, G. & Weber, K. Golgi-derived membranes that contain an acylated viral polypeptide are used for vaccinia virus envelopment. J. Virol. 55, 651–659 (1985).
Cai, L., Holoweckyj, N., Schaller, M. D. & Bear, J. E. Phosphorylation of coronin 1B by protein kinase C regulates interaction with Arp2/3 and cell motility. J. Biol. Chem. 280, 31913–31923 (2005).
Chan, K. T., Roadcap, D. W., Holoweckyj, N. & Bear, J. E. Coronin 1C harbours a second actin-binding site that confers co-operative binding to F-actin. Biochem. J. 444, 89–96 (2012).
Arakawa, Y., Cordeiro, J. V., Schleich, S., Newsome, T. & Way, M. The release of vaccinia virus from infected cells requires RhoA-mDia modulation of cortical actin. Cell Host Microbe 1, 227–240 (2007).
Welman, A., Serrels, A., Brunton, V. G., Ditzel, M. & Frame, M. C. Two-color photoactivatable probe for selective tracking of proteins and cells. J. Biol. Chem. 285, 11607–11616 (2010).
Fitzgerald, D. J. et al. Protein complex expression by using multigene baculoviral vectors. Nat. Methods 3, 1021–1032 (2006).
Goley, E. D., Rodenbusch, S. E., Martin, A. C. & Welch, M. D. Critical conformational changes in the Arp2/3 complex are induced by nucleotide and nucleation promoting factor. Mol. Cell 16, 269–279 (2004).
Pernier, J. et al. Dimeric WH2 domains in Vibrio VopF promote actin filament barbed-end uncapping and assisted elongation. Nat. Struct. Mol. Biol. 20, 1069–1076 (2013).
Churchman, L. S., Okten, Z., Rock, R. S., Dawson, J. F. & Spudich, J. A. Single molecule high-resolution colocalization of Cy3 and Cy5 attached to macromolecules measures intramolecular distances through time. Proc. Natl Acad. Sci. USA 102, 1419–1423 (2005).
Sbalzarini, I. F. & Koumoutsakos, P. Feature point tracking and trajectory analysis for video imaging in cell biology. J. Struct. Biol. 151, 182–195 (2005).
Acknowledgements
We would like to thank members of the Way laboratory, R. Treisman (The Francis Crick Institute) and H. Walden (University of Dundee) for comments on the manuscript, M. Howell (The Francis Crick Institute) for his helpful discussions, J. Cockburn (University of Leeds) for his structural insights into Arp2/3, R. George (The Francis Crick Institute) for his help with Arp2/3 purification, and B. Wharam for his contribution to this project during his summer studentship. We would like to thank J. Bear (University of North Carolina, Chapel Hill, USA) for coronin antibodies, I. Berger (University of Bristol, UK) for the pFL vector, and E. Benanti and M. Welch (University of California, Berkeley, USA) for helpful discussions concerning production of recombinant Arp2/3. This work was supported by Cancer Research UK (C.G. and M.W.) and by postdoctoral fellowships from FRQS (Fonds de recherche du Québec - Santé), EMBO and the Canadian Institutes of Health Research (CIHR) to J.V.G.A. M.-F.C. was supported by ERC advanced grant no. 249982 and FP7 241548.
Author information
Authors and Affiliations
Contributions
J.V.G.A. and M.W. designed the study and wrote the manuscript. J.V.G.A. performed and analysed the experiments with help from C.G. S.K. purified the recombinant Arp2/3 complexes. D.J.B. provided quantitative analysis of protein localizations in actin tails. J.P. performed the in vitro actin assays with input from M.-F.C. All authors discussed the results and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
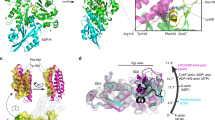
Supplementary Figure 1 HeLa cells express both isoforms of ARPC1 and ARPC5.
(A) Sequence alignment of human ARPC5 (NP_005708.1) and ARPC5L (NP_112240.1) (top) and ARPC1A (NP_ 006400.2) and ARPC1B (NP_005711.1) (bottom). Pink lines denote residues that are not present in the bovine crystal structure (PDB 1K8K), green line denotes residues involved in the interface between ARPC5 and Arp2. (B) Isoform specific antibodies do not cross react with the opposite endogenous (upper panels) or GFP-tagged (lower panels) ARPC1 or ARPC5 isoforms in HeLa cells. (C) The graphs show the relative abundance of the different ARPC1 or ARPC5 isoforms in a panel of cell lines normalized to their own ARPC2 levels relative to HeLa cells. Error bars represent s.e.m. from n = 3 independent experiments. (D) Immunoblot analysis of HeLa cells treated with siRNA against ARPC2 results in concomitant loss of all other complex subunits.
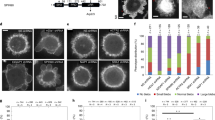
Supplementary Figure 2 ARPC1 and ARPC5 isoforms differentially affect actin tail lengths.
(a) Immunofluorescent images from which insets in Fig. 2b were taken. An image of a HeLa cell treated with siRNA against ARPC2 is also included. The virus is labelled in green and actin (phalloidin) in red. Scale bar is 10 μm. (B) Quantification of actin tail lengths of individual siRNA oligonucleotides from the siGenome pools against ARPC5, ARPC5L, ARPC1A and ARPC1B used in Fig. 2. Data from 3 independent experiments were combined and error bars represent s.e.m. from n = 360 tails. (C) Immunoblot analysis of RNAi treated Lifeact-RFP cells used in Fig. 2d to measure virus speeds. (D) HeLa cells stably expressing GFP-tagged ARPC1 and ARPC5 isoforms were treated with 500 nM Latrunculin A for 20 min before imaging. GFP-tagged isoforms recruited to vaccinia virus (red) were bleached and their recovery was measured (ARPC1B is shown). Scale bar is 5 μm. The graph shows a comparison of the recovery kinetics of photobleached GFP-tagged isoforms. Data from 3 independent experiments were combined and error bars represent s.e.m. for the indicated number of virus (n).
Supplementary Figure 3 Cofilin promotes actin tail disassembly independently of either ARPC5 isoform.
(A) Immunofluorescence images of virus (red) induced actin tails (green) in HeLa cells treated with the indicated siRNA. (B) Quantification of actin tail lengths in cells treated with the indicated siRNA (n = 360 tails). The immunoblot shows the degree of knockdown. Scale bar is 5 μm. Data from 3 independent experiments were combined and error bars represent s.e.m. from n = 360 tails. Tukey’s multiple comparisons test was used to determine Statistical significances, where ∗∗∗P < 0.001. Mean and s.e.m. are indicated below graph.
Supplementary Figure 4 Actin tail intensity profiles.
Average fluorescence intensity of GFP-tagged proteins in virus-induced actins tails in HeLa cells as a function of distance from the virus. Each profile represents the average of individual ‘snapshots’ of tails taken from live cell imaging. The data are fit with either a Gaussian (vaccinia), an exponentially modified Gaussian (lifeact, ArpC2, cortactin, coronin1C), or a sum of the two (N-WASP). The vertical dotted line denotes the position of peak fluorescence intensity of the RFP-tagged viral protein, A3. Error bars represent 95% confidence intervals with ‘snapshots’ of Vaccinia Virus (n = 4,050), N-WASP (n = 4,342), Cortactin (n = 7,558), ARPC2 (n = 2,043), LifeAct (nt = 1,314) and Coronin 1C (nt = 2,302).
Supplementary Figure 5 Localisation of cortactin is not dependent on Arp2/3 complex isoforms.
(A–C) Immunoblot analysis of HeLa cells stably expresssing (A) photoactivatable β-actin, (B) photoactivatable ARPC2 or (C) photoactivable Cortactin and treated with the indicated siRNAs for 72 h and probed with the indicated antibodies. (D) Average fluorescence intensity of the Cortactin-GFP in virus-induced actins tails as a function of distance from the virus in cells treated with the indicated siRNAs. Each profile is generated by analysing 900–2,100 ‘snapshots’ of the indicated number of tails in live imaging of the indicated number of cells. The data are fit with exponentially modified Gaussian functions. Error bars represent 95% confidence intervals with cortactin-GFP ‘snapshots’ for Control (n = 2,147), siC5-C1A (n = 1,100) and siC5L-C1B (n = 935).
Supplementary information
Supplementary Information
Supplementary Information (PDF 1462 kb)
Actin based motility of vaccinia in cells lacking ARPC5 or ARPC5L.
The panel of three movies represent HeLa cells stably expressing Lifeact-RFP, infected with Vaccinia expressing YFP-A3 (a core viral protein) for 8 h, post siRNA transfection with either control or siRNA against ARPC5 or ARPC5L for 72 h. Movies correspond to 1 min at 1 s intervals. Movies are representative of the data presented in Fig. 2d. (MOV 382 kb)
Actin based motility of vaccinia in cells lacking ARPC1A or ARPC1B.
The panel of three movies represent HeLa cells stably expressing Lifeact-RFP, infected with Vaccinia expressing YFP-A3 (a core viral protein) for 8 h, post siRNA transfection with either control or siRNA against ARPC1A or ARPC1B for 72 h. Movies correspond to 1 min at 1 s intervals. Movies are representative of the data presented in Fig. 2d. (MOV 382 kb)
Photoactivation of actin in a vaccinia actin tail.
The movie shows a representative photoactivation experiment, from which the panels in Fig. 4a were extracted. HeLa cells stably expressing Cherry-GFPPAβ-actin (red before photoactivation) were infected with Vaccinia expressing YFP-A3 for 8 h, post transfection with RNAi against ARPC1A for 72 h. (MOV 651 kb)
Actin based motility of vaccinia in cells lacking Coronin 1B or IC.
The panel of three movies represent HeLa cells stably expressing LifeAct-RFP, infected with Vaccinia expressing YFP-A3 (a core viral protein) for 8 h, post siRNA transfection with either control or siRNA against coronin1B or coronin1C for 72 h. Movies correspond to 1 min at 2.4 s intervals. Movies are representative of the data presented in Fig. 5c. (MOV 437 kb)
Rights and permissions
About this article
Cite this article
Abella, J., Galloni, C., Pernier, J. et al. Isoform diversity in the Arp2/3 complex determines actin filament dynamics. Nat Cell Biol 18, 76–86 (2016). https://doi.org/10.1038/ncb3286
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb3286
This article is cited by
-
Cytokinetic abscission requires actin-dependent microtubule severing
Nature Communications (2024)
-
ARPC1A correlates with poor prognosis in prostate cancer and is up-regulated by glutamine metabolism to promote tumor cell migration, invasion and cytoskeletal changes
Cell & Bioscience (2023)
-
T cell migration requires ion and water influx to regulate actin polymerization
Nature Communications (2023)
-
ARPC5 is transcriptionally activated by KLF4, and promotes cell migration and invasion in prostate cancer via up-regulating ADAM17
Apoptosis (2023)
-
A proteomics analysis of 5xFAD mouse brain regions reveals the lysosome-associated protein Arl8b as a candidate biomarker for Alzheimer’s disease
Genome Medicine (2023)