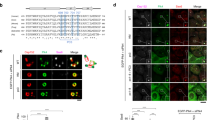
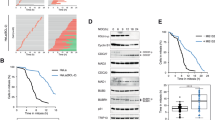
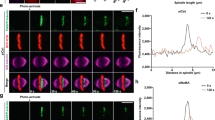
Abstract
Activation of mitosis-promoting factor (MPF) drives mitotic commitment1. In human cells active MPF appears first on centrosomes2. We show that local activation of MPF on the equivalent organelle of fission yeast, the spindle pole body (SPB), promotes Polo kinase activity at the SPBs long before global MPF activation drives mitotic commitment. Artificially promoting MPF or Polo activity at various locations revealed that this local control of Plo1 activity on G2 phase SPBs dictates the timing of mitotic commitment. Cytokinesis of the rod-shaped fission yeast cell generates a naive, new, cell end. Growth is restricted to the experienced old end until a point in G2 phase called new end take off (NETO) when bipolar growth is triggered3. NETO coincided with MPF activation of Plo1 on G2 phase SPBs (ref. 4). Both MPF and Polo activities were required for NETO and both induced NETO when ectopically activated at interphase SPBs. NETO promotion by MPF required polo. Thus, local MPF activation on G2 SPBs directs polo kinase to control at least two distinct and temporally separated, cell-cycle transitions at remote locations.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Nurse, P. Universal control mechanism regulating onset of M-phase. Nature 344, 503–508 (1990).
Jackman, M., Lindon, C., Nigg, E. A. & Pines, J. Active cyclin B1-Cdk1 first appears on centrosomes in prophase. Nat. Cell Biol. 5, 143–148 (2003).
Mitchison, J. M. & Nurse, P. Growth in cell length in the fission yeast Schizosaccharomyces pombe. J. Cell Sci. 75, 357–376 (1985).
Mulvihill, D. P., Petersen, J., Ohkura, H., Glover, D. M. & Hagan, I. M. Plo1 kinase recruitment to the spindle pole body and its role in cell division in Schizosaccharomyces pombe. Mol. Biol. Cell 10, 2771–2785 (1999).
Hudson, J. D., Feilotter, H. & Young, P. G. stf1 : non wee mutations epistatic to cdc25 in the fission yeast Schizosaccharomyces pombe. Genetics 126, 309–315 (1990).
Bridge, A. J., Morphew, M., Bartlett, R. & Hagan, I. M. The fission yeast SPB component Cut12 links bipolar spindle formation to mitotic control. Genes Dev. 12, 927–942 (1998).
Tallada, V. A., Bridge, A. J., Emery, P. E. & Hagan, I. M. Suppression of the S. pombe cut12.1 cell cycle defect by mutations in cdc25 and genes involved in transcriptional and translational control. Genetics 176, 73–83 (2007).
MacIver, F. H., Tanaka, K., Robertson, A. M. & Hagan, I. M. Physical and functional interactions between polo kinase and the spindle pole component Cut12 regulate mitotic commitment in S. pombe. Genes Dev. 17, 1507–1523 (2003).
Tanaka, K. et al. The role of Plo1 kinase in mitotic commitment and septation in Schizosaccharomyces pombe. EMBO J. 20, 1259–1270 (2001).
Bähler, J. et al. Role of polo kinase and Mid1p in determining the site of cell division in fission yeast. J. Cell Biol. 143, 1603–1616 (1998).
Dischinger, S., Krapp, A., Xie, L., Paulson, J. R. & Simanis, V. Chemical genetic analysis of the regulatory role of Cdc2p in the S. pombe septation initiation network. J. Cell Sci. 121, 843–853 (2008).
May, J. & Mitchison, J. M. Length growth in fission yeast cells measured by two novel techniques. Nature 322, 752–754 (1986).
Zhang, C. et al. A second-site suppressor strategy for chemical genetic analysis of diverse protein kinases. Nat. Methods 2, 435–441 (2005).
Ohkura, H., Hagan, I. M. & Glover, D. M. The conserved Schizosaccharomyces pombe kinase Plo1, required to form a bipolar spindle, the actin ring, and septum, can drive septum formation in G1 and G2 cells. Genes Dev. 9, 1059–1073 (1995).
Kothe, M. et al. Structure of the catalytic domain of human polo-like kinase 1. Biochemistry 46, 5960–5971 (2007).
May, K. M., Reynolds, N., Cullen, F., Yanagida, M. & Ohkura, H. Polo boxes and Cut23 (Apc8) mediate an interaction between polo kinase and the anaphase-promoting complex for fission yeast. J. Cell Biol. 156, 23–28 (2002).
Archambault, V. & Glover, D. M. Polo-like kinases: conservation anddivergence in their functions and regulation. Nat. Rev. Mol. Cell Biol. 10, 265–275 (2009).
Elia, A. E., Cantley, L. C. & Yaffe, M. B. Proteomic screen finds pSer/pThr-binding domain localizing Plk1 to mitotic substrates. Science 299, 1228–231 (2003).
Rothbauer, U. et al. Targeting and tracing antigens in live cells with fluorescent nanobodies. Nat. Methods 3, 887–889 (2006).
Rothbauer, U. et al. A versatile nanotrap for biochemical and functional studies with fluorescent fusion proteins. Mol. Cell Prot.: MCP 7, 282–289 (2008).
Bertazzi, D. T., Kurtulmus, B. & Pereira, G. The cortical protein Lte1 promotes mitotic exit by inhibiting the spindle position checkpoint kinase Kin4. J. Cell Biol. 193, 1033–1048 (2011).
Alfa, C. E., Ducommun, B., Beach, D. & Hyams, J. S. Distinct nuclear andspindle pole body populations of cyclin-cdc2 in fission yeast. Nature 347, 680–682 (1990).
Decottingnies, A., Zarzov, P. & Nurse, P. In vivo localisation of fission yeast cyclin-dependent kinase cdc2p and cyclin B cdc13p during mitosis and meiosis. J. Cell Sci. 114, 2627–2640 (2001).
Gould, K. L. & Nurse, P. Tyrosine phosphorylation of the fission yeast Cdc2+ protein-kinase regulates entry into mitosis. Nature 342, 39–45 (1989).
Petersen, J. & Nurse, P. TOR signalling regulates mitotic commitment through the stress MAP kinase pathway and the Polo and Cdc2 kinases. Nat. Cell Biol. 9, 1263–1272 (2007).
Halova, L. & Petersen, J. Aurora promotes cell division during recovery from TOR-mediated cell cycle arrest by driving spindle pole body recruitment of Polo. J. Cell Sci. 124, 3441–3449 (2011).
Petersen, J. & Hagan, I. M. Polo kinase links the stress pathway to cell cycle control and tip growth in fission yeast. Nature 435, 507–512 (2005).
Hartmuth, S. & Petersen, J. Fission yeast Tor1 functions as part of TORC1 to control mitotic entry through the stress MAPK pathway following nutrient stress. J. Cell Sci. 122, 1737–1746 (2009).
Moseley, J. B., Mayeux, A., Paoletti, A. & Nurse, P. A spatial gradient coordinates cell size and mitotic entry in fission yeast. Nature 459, 857–860 (2009).
Martin, S. G. & Berthelot-Grosjean, M. Polar gradients of the DYRK-family kinase Pom1 couple cell length with the cell cycle. Nature 459, 852–856 (2009).
Das, M. et al. Oscillatory dynamics of Cdc42 GTPase in the control of polarized growth. Science 337, 239–243 (2012).
Petersen, J. TOR signalling regulates mitotic commitment through stress-activated MAPK and Polo kinase in response to nutrient stress. Biochem. Soc. Trans. 37, 273–277 (2009).
Simanis, V. Events at the end of mitosis in the budding and fission yeasts. J. Cell Sci. 116, 4263–4275 (2003).
Moreno, S., Hayles, J. & Nurse, P. Regulation of p34cdc2 protein-kinase during mitosis. Cell 58, 361–372 (1989).
Creanor, J. & Mitchison, J. M. Reduction of perturbations in leucine incorporation in synchronous cultures of Schizosaccharomyces pombe made by elutriation. J. Gen. Micro. 112, 385–388 (1979).
Cipak, L. et al. Generation of a set of conditional analog-sensitive alleles of essential protein kinases in the fission yeast Schizosaccharomyces pombe. Cell Cycle 10, 3527–3532 (2011).
Hagan, I. & Yanagida, M. The product of the spindle formation gene sad1+ associates with the fission yeast spindle pole body and is essential for viability. J. Cell Biol. 129, 1033–1047 (1995).
Petersen, J., Paris, J., Willer, M., Philippe, M. & Hagan, I. M. The S. pombe aurora related kinase Ark1 associates with mitotic structures in a stage dependent manner and is required for chromosome segregation. J. Cell Sci. 114, 4371–4384 (2001).
Marks, J. & Hyams, J. S. Localization of F-actin through the cell-division cycle of Schizosaccharomyces pombe. E. J. Cell Biol. 39, 27–32 (1985).
Woods, A. et al. Definition of individual components within the cytoskeleton of Trypanosoma brucei by a library of monoclonal-antibodies. J. Cell Sci. 93, 491–500 (1989).
Davis, F. M., Tsao, T. Y., Fowler, S. K. & Rao, P. N. Monoclonal antibodies to mitotic cells. Proc. Natl Acad. Sci. USA 80, 2926–2930 (1983).
Knutsen, J. H. et al. Cell-cycle analysis of fission yeast cells by flow cytometry. PloS ONE 6, e17175 (2011).
Simanis, V. & Nurse, P. The cell cycle control gene cdc2+ of fission yeast encodes a protein kinase potentially regulated by phosphorylation. Cell 45, 261–268 (1986).
MacIver, F. H., Glover, D. M. & Hagan, I. M. A ‘marker switch’ approach for targeted mutagenesis of genes in Schizosaccharomyces pombe. Yeast 20, 587–594 (2003).
Acknowledgements
We thank B. Hodgson for technical assistance; V. Doye (Insitut Jacques Monod, Paris, France), D. Mulvihill (University of Kent, UK), U. Fleig (Düsseldorf, Germany) and D. Feret (Paterson Institute, UK) for strains; K. Gull (Oxford University, UK) and H. Ohkura (Edinburgh University, UK) for antibodies; D. Feret (Paterson Institute, UK), G. Pereira (DKFZ, Germany) and A. Carr (GDSC, Sussex, UK) for plasmids; and D. Bitton (Paterson Institute, UK) for discussions during the design of plo1.as8. This work was supported by Cancer Research UK (CRUK) grant number C147/A6058. V.S. and A.K. were supported by the Swiss National Science Foundation and EPFL.
Author information
Authors and Affiliations
Contributions
I.M.H. conceived the study. A.K. and V.S. generated cdc2.as. A.P. generated the conditional polo kinase alleles, including Plo1.RL. V.A.T. and K.Y.C. carried out the imaging work in Fig. 1. A.G. performed the remainder of the experiments with occasional assistance from A.P. and K.Y.C. for more complex experiments and cloning. I.M.H. wrote the manuscript with input and discussions from all authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 556 kb)
Supplementary Table 1
Supplementary Information (XLSX 35 kb)
Supplementary Movie 1
Supplementary Information (MOV 719 kb)
Supplementary Movie 2
Supplementary Information (MOV 698 kb)
Supplementary Movie 3
Supplementary Information (MOV 3523 kb)
Supplementary Movie 4
Supplementary Information (MOV 9410 kb)
Supplementary Movie 5
Supplementary Information (MOV 5045 kb)
Rights and permissions
About this article
Cite this article
Grallert, A., Patel, A., Tallada, V. et al. Centrosomal MPF triggers the mitotic and morphogenetic switches of fission yeast. Nat Cell Biol 15, 88–95 (2013). https://doi.org/10.1038/ncb2633
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb2633
This article is cited by
-
An updated view on the centrosome as a cell cycle regulator
Cell Division (2022)
-
Core control principles of the eukaryotic cell cycle
Nature (2022)
-
Two-step interphase microtubule disassembly aids spindle morphogenesis
BMC Biology (2018)
-
RNA metabolism is the primary target of formamide in vivo
Scientific Reports (2017)
-
How do fission yeast cells grow and connect growth to the mitotic cycle?
Current Genetics (2017)