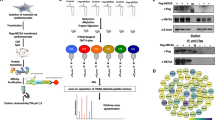
Abstract
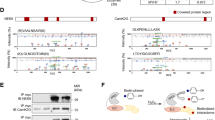
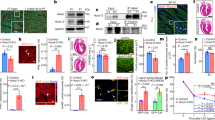
The calcium-activated protein phosphatase calcineurin is controlled by regulator of calcineurin (RCAN) in organisms ranging from yeast to mammals. Here we performed a yeast two-hybrid screen with RCAN1 as bait, identifying TAK1 binding protein 2 (TAB2) as an interacting partner. TAB2 interacted directly with RCAN1 in vitro and in vivo, recruiting TAK1, TAB1 and calcineurin, forming a macromolecular signalling complex. Overexpression of TAK1 and TAB1, or active TAK1ΔN, promoted direct phosphorylation of RCAN1 in vitro and in vivo. TAK1 phosphorylated RCAN1 at Ser 94 and Ser 136, converting RCAN1 from an inhibitor to a facilitator of calcineurin–NFAT signalling, and enhancing NFATc1 nuclear translocation, NFAT transcriptional activation and the hypertrophic growth of cultured cardiomyocytes. The TAK1–TAB1–TAB2 and the calcineurin–NFAT signalling modules did not interact in Rcan1/2- or Tab2-deficient mouse embryonic fibroblast (MEF) cultures. Calcineurin activation also dephosphorylated and inhibited TAK1 and TAB1, an effect that was absent in Rcan1/2 deficient MEFs. Functionally, TAK1 was indispensable for the cardiomyocyte growth response induced by pro-hypertrophic stimuli through calcineurin. These results describe a signalling relationship between two central regulatory pathways in which TAK1–TAB1–TAB2 selectively induces calcineurin–NFAT signalling through direct phosphorylation of RCAN1, while calcineurin activation diminishes TAK1 signalling by dephosphorylation of TAK1 and TAB1.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Wu, H., Peisley, A., Graef, I. A. & Crabtree G. R. NFAT signalling and the invention of vertebrates. Trends Cell Biol. 17, 251–260 (2007).
Hogan, P. G., Chen, L., Nardone, J. & Rao, A. Transcriptional regulation by calcium, calcineurin, and NFAT. Genes Dev. 17, 2205–2232 (2003).
Molkentin, J. D. A calcineurin-dependent transcriptional pathway for cardiac hypertrophy. Cell 93, 215–228 (1998).
Hilioti, Z. & Cunningham, K. W. The RCN family of calcineurin regulators. Biochem. Biophys. Res. Commun. 311, 1089–1093 (2003).
Davies, K. J. et al. Renaming the DSCR1/Adapt78 gene family as RCAN: regulators of calcineurin. FASEB J. 21, 3023–3028 (2007).
Chan, B., Greenan, G., McKeon, F. & Ellenberger, T. Identification of a peptide fragment of DSCR1 that competitively inhibits calcineurin activity in vitro and in vivo. Proc. Natl Acad. Sci. USA 102, 13075–13080 (2005).
Hilioti, Z. et al. GSK-3 kinases enhance calcineurin signalling by phosphorylation of RCNs. Genes Dev. 18, 35–47 (2004).
Kingsbury, T. J. & Cunningham, K. W. A conserved family of calcineurin regulators. Genes Dev. 14, 1595–1604 (2000).
Gorlach, J. et al. Identification and characterization of a highly conserved calcineurin binding protein, CBP1/calcipressin, in Cryptococcus neoformans. EMBO J. 19, 3618–3629 (2000).
Sanna, B. et al. Modulatory calcineurin-interacting proteins 1 and 2 function as calcineurin facilitators in vivo. Proc. Natl Acad. Sci. USA 103, 7327–7332 (2006).
Vega, R. B. et al. Dual roles of modulatory calcineurin-interacting protein 1 in cardiac hypertrophy. Proc. Natl. Acad. Sci. USA 100, 669–674 (2003).
Genesca, L. et al. Phosphorylation of calcipressin 1 increases its ability to inhibit calcineurin and decreases calcipressin half-life. Biochem. J. 374, 567–575 (2003).
Takaesu, G. et al. TAB2, a novel adaptor protein, mediates activation of TAK1 MAPKKK by linking TAK1 to TRAF6 in the IL-1 signal transduction pathway. Mol. Cell 5, 649–658 (2000).
Yamaguchi, K. et al. Identification of a member of the MAPKKK family as a potential mediator of TGF-β signal transduction. Science 270, 2008–2011 (1995).
Besse, A. et al. TAK1-dependent signalling requires functional interaction with TAB2/TAB3. J. Biol. Chem. 282, 3918–3928 (2007).
Shibuya, H. et al. TAB1: an activator of the TAK1 MAPKKK in TGF-β signal transduction. Science 272, 1179–1182 (1996).
Ninomiya-Tsuji J. et al. The kinase TAK1 can activate the NIK-I κB as well as the MAP kinase cascade in the IL-1 signalling pathway. Nature 398, 252–256 (1999).
Wang, C. et al. TAK1 is a ubiquitin-dependent kinase of MKK and IKK. Nature 412, 346–351 (2001).
Zhang, D. et al. TAK1 is activated in the myocardium after pressure overload and is sufficient to provoke heart failure in transgenic mice. Nature Med. 6, 556–563 (2000).
Vega, R. B., Yang, J., Rothermel, B. A., Bassel-Duby, R. & Williams R. S. Multiple domains of MCIP1 contribute to inhibition of calcineurin activity. J. Biol. Chem. 277, 30401–30407 (2002).
Rothermel, B. et al. A protein encoded within the Down syndrome critical region is enriched in striated muscles and inhibits calcineurin signalling. J. Biol. Chem. 275, 8719–8725 (2000).
Rothermel, B. A. et al. Myocyte-enriched calcineurin-interacting protein, MCIP1, inhibits cardiac hypertrophy in vivo. Proc. Natl Acad. Sci. USA 98, 3328–3333 (2001).
Watkins, S. J., Jonker, L. & Arthur, H. M. A direct interaction between TGFβ activated kinase 1 and the TGFβ type II receptor: implications for TGFβ signalling and cardiac hypertrophy. Cardiovasc. Res. 69, 432–439 (2006).
Tung, H. Y., Wangm W. & Chan, C. S. Regulation of chromosome segregation by Glc8p, a structural homologue of mammalian inhibitor 2 that functions as both an activator and an inhibitor of yeast protein phosphatase 1. Mol Cell Biol. 15, 6064–6074 (1995).
Greengard, P., Allen. P. B. & Nairn, A. C. Beyond the dopamine receptor: the DARPP-32/protein phosphatase-1 cascade. Neuron 23, 435–447 (1999).
Cohen, P. T. Protein phosphatase 1 — targeted in many directions. J. Cell. Sci. 115, 241–256 (2002).
Abbasi, S. et al. Protein kinase-mediated regulation of calcineurin through the phosphorylation of modulatory calcineurin-interacting protein 1. J. Biol. Chem. 281, 7717–7726 (2006).
Liu, Q., Wilkins, B. J., Lee, Y. J., Ichijo, H. & Molkentin, J. D. Direct interaction and reciprocal regulation between ASK1 and calcineurin-NFAT control cardiomyocyte death and growth. Mol. Cell. Biol. 26, 3785–3797 (2006).
Liang, Q. et al. The transcription factor GATA4 is activated by extracellular signal-regulated kinase 1- and 2-mediated phosphorylation of serine 105 in cardiomyocytes. Mol. Cell. Biol. 21, 7460–7469 (2001).
Godeny, M. D. et al. The N-terminal SH2 domain of the tyrosine phosphatase, SHP-2, is essential for Jak2-dependent signalling via the angiotensin II type AT1 receptor. Cell. Signal. 19, 600–609 (2007).
Acknowledgements
We are very grateful to Yang Xia for the generous gift of the S108/112A mutant RCAN1 plasmid, and Jun Tsuji for providing the Tak1−/− MEFs originated from Akira's laboratory. This work was supported by grants from the National Institutes of Health (J.D.M), the Fondation Leducq (Heart failure network grant to J.D.M), and the Howard Hughes Medical Institute.
Author information
Authors and Affiliations
Contributions
Q.L. performed the experiments; J.O.M. supervised all work and data interpretation; J.C.B. performed mass spectrometry of phosphorylated RCAN1.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 243 kb)
Rights and permissions
About this article
Cite this article
Liu, Q., Busby, J. & Molkentin, J. Interaction between TAK1–TAB1–TAB2 and RCAN1–calcineurin defines a signalling nodal control point. Nat Cell Biol 11, 154–161 (2009). https://doi.org/10.1038/ncb1823
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb1823
This article is cited by
-
Oxidative stress induces mitochondrial iron overload and ferroptotic cell death
Scientific Reports (2023)
-
RCAN1 knockout and overexpression recapitulate an ensemble of rest-activity and circadian disruptions characteristic of Down syndrome, Alzheimer’s disease, and normative aging
Journal of Neurodevelopmental Disorders (2022)
-
RCAN1-mediated calcineurin inhibition as a target for cancer therapy
Molecular Medicine (2022)
-
RCAN1 deficiency aggravates sepsis-induced cardiac remodeling and dysfunction by accelerating mitochondrial pathological fission
Inflammation Research (2022)
-
AAV-mediated YAP expression in cardiac fibroblasts promotes inflammation and increases fibrosis
Scientific Reports (2021)