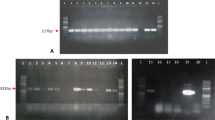
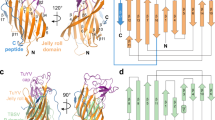
Abstract
The sap-sucking insects (order Hemiptera), including aphids, planthoppers, whiteflies and stink bugs, present one of the greatest challenges for pest management in global agriculture. Insect neurotoxins offer an alternative to chemical insecticides for controlling these pests, but require delivery into the insect hemocoel. Here we use the coat protein of a luteovirus, an aphid-vectored plant virus, to deliver a spider-derived, insect-specific toxin that acts within the hemocoel. The luteovirid coat protein is sufficient for delivery of fused proteins into the hemocoel of pea aphids, Acyrthosiphon pisum, without virion assembly. We show that when four aphid pest species—A. pisum, Rhopalosiphum padi, Aphis glycines and Myzus persicae—feed on a recombinant coat protein–toxin fusion, either in an experimental membrane sachet or in transgenic Arabidopsis plants, they experience significant mortality. Aphids fed on these fusion proteins showed signs of neurotoxin-induced paralysis. Luteovirid coat protein–insect neurotoxin fusions represent a promising strategy for transgenic control of aphids and potentially other hemipteran pests.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Sanahuja, G., Banakar, R., Twyman, R.M., Capell, T. & Christou, P. Bacillus thuringiensis: a century of research, development and commercial applications. Plant Biotechnol. J. 9, 283–300 (2011).
Stokstad, E. & Grullon, G. Infographic: pesticide planet. Science 341, 730–731 (2013).
Chougule, N.P. & Bonning, B.C. Toxins for transgenic resistance to hemipteran pests. Toxins 4, 405–429 (2012).
Li, H., Chougule, N.P. & Bonning, B.C. Interaction of the Bacillus thuringiensis delta endotoxins Cry1Ac and Cry3Aa with the gut of the pea aphid, Acyrthosiphon pisum (Harris). J. Invertebr. Pathol. 107, 69–78 (2011).
Lu, Y. et al. Mirid bug outbreaks in multiple crops correlated with wide-scale adoption of Bt cotton in China. Science 328, 1151–1154 (2010).
Li, G. et al. Effects of transgenic Bt cotton on the population density, oviposition behavior, development, and reproduction of a nontarget pest, Adelphocoris suturalis (Hemiptera: Miridae). Environ. Entomol. 39, 1378–1387 (2010).
Schmidt, N.R. & Bonning, B.C. in Molecular Biology and Genetics of Lepidoptera (eds. M. Goldsmith & F. Marc) 307–319 (Taylor and Francis, Baton Rouge, FL, 2010).
King, G.F. & Hardy, M.C. Spider-venom peptides: structure, pharmacology, and potential for control of insect pests. Annu. Rev. Entomol. 58, 475–496 (2013).
Reavy, B. & Mayo, M.A. Persistent transmission of luteoviruses by aphids. Adv. Bot. Res. 36, 21–46 (2002).
Gray, S. & Gildow, F.E. Luteovirus-aphid interactions. Annu. Rev. Phytopathol. 41, 539–566 (2003).
Liu, S., Sivakumar, S., Wang, Z., Bonning, B.C. & Miller, W.A. The readthrough domain of Pea enation mosaic virus coat protein is not essential for virus stability in the hemolymph of the pea aphid. Arch. Virol. 154, 469–479 (2009).
Fletcher, J.I. et al. The structure of a novel insecticidal neurotoxin, ω-atracotoxin-HV1, from the venom of an Australian funnel web spider. Nat. Struct. Biol. 4, 559–566 (1997).
Pal, N., Yamamoto, T., King, G.F., Waine, C. & Bonning, B.C. Aphicidal efficacy of scorpion- and spider-derived neurotoxins. Toxicon 70, 114–122 (2013).
Sivakumar, S. et al. Baculovirus-expressed virus-like particles of Pea enation mosaic virus vary in size and encapsidate baculovirus mRNAs. Virus Res. 139, 54–63 (2009).
Lamb, J.W. et al. Assembly of virus-like particles in insect cells infected with a baculovirus containing a modified coat protein gene of potato leafroll luteovirus. J. Gen. Virol. 77, 1349–1358 (1996).
Locke, M. & Russell, V.W. in Microscopic Anatomy of Invertebrates, vol. 11B (eds. F.W. Harrison & M. Locke) 687–709 (Wiley-Liss Inc., New York, 1998).
Crossley, A.C. in Comprehensive Insect Physiology, Biochemistry, and Pharmacology (eds. G.A. Kerkut & L.I. Gilbert) 487–515 (Pergamon Press, New York, 1985).
Wang, X. et al. Structure-function studies of ω-atracotoxin, a potent antagonist of insect voltage-gated calcium channels. Eur. J. Biochem. 264, 488–494 (1999).
Tedford, H.W., Fletcher, J.I. & King, G.F. Functional significance of the beta hairpin in the insecticidal neurotoxin ω-atracotoxin-Hv1a. J. Biol. Chem. 276, 26568–26576 (2001).
Jeffers, L.A. & Michael Roe, R. The movement of proteins across the insect and tick digestive system. J. Insect Physiol. 54, 319–332 (2008).
Maiti, I.B., Oey, N., Dahlman, D.L. & Webb, B.A. Antibiosis-type resistance in transgenic plants expressing a teratocyte secretory peptide (TSP) gene from a hymenopteran endoparasite (Microplitis croceipes). Plant Biotechnol. J. 1, 209–219 (2003).
Tortiglione, C. et al. The expression in tobacco plants of Aedes aegypti Trypsin Modulating Oostatic Factor (Aea-TMOF) alters growth and development of the tobacco budworm, Heliothis virescens. Mol. Breed. 9, 159–169 (2002).
Gray, S.M. & Banerjee, N. Mechanisms of arthropod transmission of plant and animal viruses. Microbiol. Mol. Biol. Rev. 63, 128–148 (1999).
Fitches, E.C., Pyati, P., King, G.F. & Gatehouse, J.A. Fusion to snowdrop lectin magnifies the oral activity of insecticidal ω-hexatoxin-Hv1a peptide by enabling its delivery to the central nervous system. PLoS ONE 7, e39389 (2012).
Fitches, E.C. et al. Insecticidal activity of scorpion toxin (ButaIT) and snowdrop lectin (GNA) containing fusion proteins towards pest species of different orders. Pest Manag. Sci. 66, 74–83 (2010).
Bonning, B.C. & Chougule, N.P. Delivery of intrahemocoelic peptides for insect pest management. Trends Biotechnol. (in the press) (2013).
Pennacchio, F., Giordana, B. & Rao, R. in Parasitoid Viruses: Symbionts and Pathogens (eds. N.E. Beckage & J.M. Drezen) 269–283 (Academic Press, San Diego, 2012).
Demler, S.A., Rucker-Feeney, D.G., Skaf, J.S. & deZoeten, G.A. Expression and suppression of circulative aphid transmission in pea enation mosaic virus. J. Gen. Virol. 78, 511–523 (1997).
Fukatsu, T., Tsuchida, T., Nikoh, N. & Koga, R. Spiroplasma symbiont of the pea aphid, Acyrthosiphon pisum (Insecta: Homoptera). Appl. Environ. Microbiol. 67, 1284–1291 (2001).
Tedford, H.W. et al. Scanning mutagenesis of ω-atracotoxin-Hv1a reveals a spatially restricted epitope that confers selective activity against insect calcium channels. J. Biol. Chem. 279, 44133–44140 (2004).
Febvay, G., Delobel, B. & Rahbe, Y. Influence of the amino-acid balance on the improvement of an artificial diet for a biotype of Acyrthosiphon pisum (Homoptera, Aphididae). Can. J. Zool. 66, 2449–2453 (1988).
Liu, S., Bonning, B.C. & Miller, W.A. A simple wax-embedding method for isolation of aphid hemolymph for detection of luteoviruses in the hemocoel. J. Virol. Methods 132, 174–180 (2006).
Clough, S.J. & Bent, A.F. Floral dip: a simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J. 16, 735–743 (1998).
Collins, T.J. ImageJ for microscopy. Biotechniques 43, S25–S30 (2007).
Hughes, P.R., Van Beek, N.A.M. & Wood, H.A. A modified droplet feeding method for rapid assay of Bacillus thuringiensis and baculoviruses in noctuid larvae. J. Invertebr. Pathol. 48, 187–192 (1986).
SAS Institute. SAS/STAT(R) 9.3 User's Guide (SAS Institute Inc., Cary NC, 2013).
LeOra-Software POLO-PC. A User's Guide to Probit and Logit Analysis (LeOra Software, Berkeley, California, 1987).
Acknowledgements
We thank R. Harrison, US Department of Agriculture (USDA), Beltsville, Maryland, for construction of the baculovirus expressing CP-GFP and for production of the PEMV coat protein antiserum. This work was supported by the USDA North Central Biotechnology Initiative grant number 97-34340-3987, USDA National Research Initiative grant number 02-01280, Australian Research Council Discovery Grant DP0774245, Bayer Crop Science, Syngenta, DuPont Pioneer, Grow Iowa Values Funds, Iowa State University Plant Sciences Institute, and by Hatch Act and State of Iowa funds. Mention of trade names or commercial products in this publication is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the US Department of Agriculture.
Author information
Authors and Affiliations
Contributions
W.A.M. devised the coat protein–toxin fusion strategy; B.C.B. and W.A.M. planned the experiments, interpreted results and wrote the manuscript; N.P. conducted E. coli expression of fusion proteins, generation and molecular testing of transgenic plants, membrane feeding assays and aphid feeding bioassays with transgenic plants; S.L. conducted aphid microinjections and preliminary experiments; Z.W. constructed plasmids and performed preliminary experiments; S.S. conducted research with baculovirus expressed fusions; G.F.K. provided oversight for use of the Hv1a toxin. P.M.D. conducted the statistical analyses.
Corresponding authors
Ethics declarations
Competing interests
The coat protein-toxin fusion strategy has been patented by B.C.B. and W.A.M.: US Patent No. 7,312,080 (2007). "Plant resistance to insect pests mediated by viral proteins." G.F.K. is a member of the Scientific Advisory Board of Vestaron Corporation, a company that is developing spider peptides as insecticides.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–4 and Supplementary Tables 1–3 (PDF 788 kb)
Rights and permissions
About this article
Cite this article
Bonning, B., Pal, N., Liu, S. et al. Toxin delivery by the coat protein of an aphid-vectored plant virus provides plant resistance to aphids. Nat Biotechnol 32, 102–105 (2014). https://doi.org/10.1038/nbt.2753
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nbt.2753
This article is cited by
-
Drosophila X virus-like particles as efficient dsRNA carriers for improved RNAi against the invasive species, Drosophila suzukii
Journal of Pest Science (2024)
-
Demonstrating the potential of a novel spider venom-based biopesticide for target-specific control of the small hive beetle, a serious pest of the European honeybee
Journal of Pest Science (2020)
-
Designing macrocyclic disulfide-rich peptides for biotechnological applications
Nature Chemical Biology (2018)
-
Assigning biological function using hidden signatures in cystine-stabilized peptide sequences
Scientific Reports (2018)
-
The genus Luteovirus from infection to disease
European Journal of Plant Pathology (2018)