Abstract
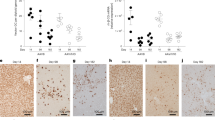
We used the integrase from phage φC31 to integrate the human Factor IX (hFIX) gene permanently into specific sites in the mouse genome. A plasmid containing attB and an expression cassette for hFIX was delivered to the livers of mice by using high-pressure tail vein injection. When an integrase expression plasmid was co-injected, hFIX serum levels increased more than tenfold to ∼4 μg/ml, similar to normal FIX levels, and remained stable throughout the more than eight months of the experiment. hFIX levels persisted after partial hepatectomy, suggesting genomic integration of the vector. Site-specific integration was proven by characterizing and quantifying genomic integration in the liver at the DNA level. Integration was documented at two pseudo-attP sites, native sequences with partial identity to attP, with one site highly predominant. This study demonstrates in vivo gene transfer in an animal by site-specific genomic integration.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Kuhstoss, S. & Rao, R.N. Analysis of the integration function of the streptomycete bacteriophage φC31. J. Mol. Biol. 222, 897–908 (1991).
Rausch, H. & Lehmann, M. Structural analysis of the actinophage φC31 atachment site. Nucleic Acids Res. 19, 5187–5189 (1991).
Groth, A.C., Olivares, E.C., Thyagarajan, B. & Calos, M.P. A phage integrase directs efficient site-specific integration in human cells. Proc. Natl. Acad. Sci. USA 97, 5995–6000 (2000).
Thyagarajan, B., Olivares, E.C., Hollis, R.P., Ginsburg, D.S. & Calos, M.P. Site-specific genomic integration in mammalian cells mediated by phage φC31 integrase. Mol. Cell. Biol. 21, 3926–3934 (2001).
Yant, S. et al. Somatic integration and long-term transgene expression in normal and haemophilic mice using a DNA transposon system. Nat. Genet. 25, 35–41 (2000).
Miao, C.H. et al. Inclusion of the hepatic locus control region, an intron, and untranslated region increases and stabilizes hepatic Factor IX gene expression in vivo but not in vitro. Mol. Ther. 1, 522–532 (2000).
Zhang, G., Budker, V. & Wolff, J.A. High levels of foreign gene expression in hepatocytes after tail vein injections of naked plasmid DNA. Hum. Gene Ther. 10, 1735–1737 (1999).
Liu, F., Song, Y.K. & Liu, D. Hydrodynamics-based transfection in animals by systemic administration of plasmid DNA. Gene Ther. 6, 1258–1266 (1999).
Stoll, S.M. et al. Epstein–Barr virus/human vector provides high-level, long-term expression of α1-antitrypsin in mice. Mol. Ther. 4, 122–129 (2001).
Morral, N. et al. High doses of a helper-dependent adenoviral vector yield supraphysiological levels of α1-antitrypsin with negligible toxicity. Hum. Gene Ther. 9, 2709–2716 (1998).
Thorpe, H.M. & Smith, M.C.M. In vitro site-specific integration of bacteriophage DNA catalyzed by a recombinase of the resolvase/invertase family. Proc. Natl. Acad. Sci. USA 95, 5505–5510 (1998).
Chen, Z.Y. et al. Linear DNAs concatemerize in vivo and result in sustained transgene expression in mouse liver. Mol. Ther. 3, 403–410 (2001).
Heid, C.A., Stevens, J., Livak, K.J. & Williams, P.M. Real-time quantitative PCR. Genome Res. 6, 986–994 (1996).
Zhang, G., Song, Y.K. & Liu, D. Long-term expression of human α1-antitrypsin gene in mouse liver achieved by intravenous adminstration of plasmid DNA using a hydrodynamics-based procedure. Gene Ther. 7, 1344–1349 (2000).
Zhang, G., Budker, V., Williams, P., Subbotin, V. & Wolff. J.A. Efficient expression of naked DNA delivered intraarterially to limb muscles of nonhuman primates. Hum. Gene Ther. 12, 427–438 (2001).
Tsui, L.V. et al. Production of human clotting Factor IX without toxicity in mice after vascular delivery of a lentiviral vector. Nat. Biotechnol. 20, 53–57 (2002).
Sclimenti, C.R., Thyagarajan, B. & Calos, M.P. Directed evolution of a recombinase for improved genomic integration at a native human sequence. Nucleic Acids Res. 29, 5044–5051 (2001).
Thyagarajan, B., Guimaraes, M.J., Groth, A.C. & Calos, M.P. Mammalian genomes contain active recombinase recognition sites. Gene 244, 47–54 (2000).
Acknowledgements
This work was supported by NIH grants HL68112 to M.P.C. and DK49022 to M.A.K.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Olivares, E., Hollis, R., Chalberg, T. et al. Site-specific genomic integration produces therapeutic Factor IX levels in mice. Nat Biotechnol 20, 1124–1128 (2002). https://doi.org/10.1038/nbt753
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nbt753
This article is cited by
-
SPARC enables genetic manipulation of precise proportions of cells
Nature Neuroscience (2020)
-
Gene Therapy for Hemophilia: Progress to Date
BioDrugs (2018)
-
Blastocyst Formation Rate and Transgene Expression are Associated with Gene Insertion into Safe and Non-Safe Harbors in the Cattle Genome
Scientific Reports (2017)
-
CRISPR/Cas9-mediated correction of human genetic disease
Science China Life Sciences (2017)