Abstract
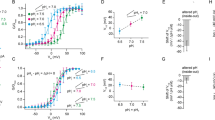
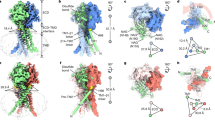
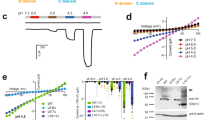
The ion selectivity of pumps and channels is central to their ability to perform a multitude of functions. Here we investigate the mechanism of the extraordinary selectivity of the human voltage-gated proton channel1, HV1 (also known as HVCN1). This selectivity is essential to its ability to regulate reactive oxygen species production by leukocytes2,3,4, histamine secretion by basophils5, sperm capacitation6, and airway pH7. The most selective ion channel known, HV1 shows no detectable permeability to other ions1. Opposing classes of selectivity mechanisms postulate that (1) a titratable amino acid residue in the permeation pathway imparts proton selectivity1,8,9,10,11, or (2) water molecules ‘frozen’ in a narrow pore conduct protons while excluding other ions12. Here we identify aspartate 112 as a crucial component of the selectivity filter of HV1. When a neutral amino acid replaced Asp 112, the mutant channel lost proton specificity and became anion-selective or did not conduct. Only the glutamate mutant remained proton-specific. Mutation of the nearby Asp 185 did not impair proton selectivity, indicating that Asp 112 has a unique role. Although histidine shuttles protons in other proteins, when histidine or lysine replaced Asp 112, the mutant channel was still anion-permeable. Evidently, the proton specificity of HV1 requires an acidic group at the selectivity filter.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
DeCoursey, T. E. Voltage-gated proton channels and other proton transfer pathways. Physiol. Rev. 83, 475–579 (2003)
Capasso, M. et al. HVCN1 modulates BCR signal strength via regulation of BCR-dependent generation of reactive oxygen species. Nature Immunol. 11, 265–272 (2010)
DeCoursey, T. E., Morgan, D. & Cherny, V. V. The voltage dependence of NADPH oxidase reveals why phagocytes need proton channels. Nature 422, 531–534 (2003)
Henderson, L. M., Chappell, J. B. & Jones, O. T. G. The superoxide-generating NADPH oxidase of human neutrophils is electrogenic and associated with an H+ channel. Biochem. J. 246, 325–329 (1987)
Musset, B. et al. A pH-stabilizing role of voltage-gated proton channels in IgE-mediated activation of human basophils. Proc. Natl Acad. Sci. USA 105, 11020–11025 (2008)
Lishko, P. V., Botchkina, I. L., Fedorenko, A. & Kirichok, Y. Acid extrusion from human spermatozoa is mediated by flagellar voltage-gated proton channel. Cell 140, 327–337 (2010)
Iovannisci, D., Illek, B. & Fischer, H. Function of the HVCN1 proton channel in airway epithelia and a naturally occurring mutation, M91T. J. Gen. Physiol. 136, 35–46 (2010)
Cherny, V. V., Markin, V. S. & DeCoursey, T. E. The voltage-activated hydrogen ion conductance in rat alveolar epithelial cells is determined by the pH gradient. J. Gen. Physiol. 105, 861–896 (1995)
DeCoursey, T. E. & Cherny, V. V. Deuterium isotope effects on permeation and gating of proton channels in rat alveolar epithelium. J. Gen. Physiol. 109, 415–434 (1997)
DeCoursey, T. E. & Cherny, V. V. Temperature dependence of voltage-gated H+ currents in human neutrophils, rat alveolar epithelial cells, and mammalian phagocytes. J. Gen. Physiol. 112, 503–522 (1998)
DeCoursey, T. E. & Cherny, V. V. Voltage-activated hydrogen ion currents. J. Membr. Biol. 141, 203–223 (1994)
Ramsey, I. S. et al. An aqueous H+ permeation pathway in the voltage-gated proton channel Hv1. Nature Struct. Mol. Biol. 17, 869–875 (2010)
Levitt, D. G., Elias, S. R. & Hautman, J. M. Number of water molecules coupled to the transport of sodium, potassium and hydrogen ions via gramicidin, nonactin or valinomycin. Biochim. Biophys. Acta 512, 436–451 (1978)
Nagle, J. F. & Morowitz, H. J. Molecular mechanisms for proton transport in membranes. Proc. Natl Acad. Sci. USA 75, 298–302 (1978)
Nelson, R. D., Kuan, G., Saier, M. H., Jr & Montal, M. Modular assembly of voltage-gated channel proteins: a sequence analysis and phylogenetic study. J. Mol. Microbiol. Biotechnol. 1, 281–287 (1999)
Ramsey, I. S., Moran, M. M., Chong, J. A. & Clapham, D. E. A voltage-gated proton-selective channel lacking the pore domain. Nature 440, 1213–1216 (2006)
Musset, B. et al. Zinc inhibition of monomeric and dimeric proton channels suggests cooperative gating. J. Physiol. (Lond.) 588, 1435–1449 (2010)
Musset, B. et al. Detailed comparison of expressed and native voltage-gated proton channel currents. J. Physiol. (Lond.) 586, 2477–2486 (2008)
Barry, P. H. The reliability of relative anion-cation permeabilities deduced from reversal (dilution) potential measurements in ion channel studies. Cell Biochem. Biophys. 46, 143–154 (2006)
Tao, X., Lee, A., Limapichat, W., Dougherty, D. A. & MacKinnon, R. A gating charge transfer center in voltage sensors. Science 328, 67–73 (2010)
Sansom, M. S. P., Kerr, I. D., Smith, G. R. & Son, H. S. The influenza A virus M2 channel: a molecular modeling and simulation study. Virology 233, 163–173 (1997)
Hu, F., Luo, W. & Hong, M. Mechanisms of proton conduction and gating in influenza M2 proton channels from solid-state NMR. Science 330, 505–508 (2010)
Venkataraman, P., Lamb, R. A. & Pinto, L. H. Chemical rescue of histidine selectivity filter mutants of the M2 ion channel of influenza A virus. J. Biol. Chem. 280, 21463–21472 (2005)
Acharya, R. et al. Structure and mechanism of proton transport through the transmembrane tetrameric M2 protein bundle of the influenza A virus. Proc. Natl Acad. Sci. USA 107, 15075–15080 (2010)
Starace, D. M. & Bezanilla, F. Histidine scanning mutagenesis of basic residues of the S4 segment of the Shaker K+ channel. J. Gen. Physiol. 117, 469–490 (2001)
Starace, D. M. & Bezanilla, F. A proton pore in a potassium channel voltage sensor reveals a focused electric field. Nature 427, 548–553 (2004)
Sokolov, S., Scheuer, T. & Catterall, W. A. Ion permeation and block of the gating pore in the voltage sensor of NaV1.4 channels with hypokalemic periodic paralysis mutations. J. Gen. Physiol. 136, 225–236 (2010)
Tu, C. K., Silverman, D. N., Forsman, C., Jonsson, B. H. & Lindskog, S. Role of histidine 64 in the catalytic mechanism of human carbonic anhydrase II studied with a site-specific mutant. Biochemistry 28, 7913–7918 (1989)
Leiding, T., Wang, J., Martinsson, J., DeGrado, W. F. & Årsköld, S. P. Proton and cation transport activity of the M2 proton channel from influenza A virus. Proc. Natl Acad. Sci. USA 107, 15409–15414 (2010)
Murata, Y., Iwasaki, H., Sasaki, M., Inaba, K. & Okamura, Y. Phosphoinositide phosphatase activity coupled to an intrinsic voltage sensor. Nature 435, 1239–1243 (2005)
Pei, J., Kim, B. H. & Grishin, N. V. PROMALS3D: a tool for multiple protein sequence and structure alignments. Nucleic Acids Res. 36, 2295–2300 (2008)
Guindon, S. & Gascuel, O. A simple, fast, and accurate algorithm to estimate large phylogenies by maximum likelihood. Syst. Biol. 52, 696–704 (2003)
Felsenstein, J. PROTPARS – Protein Sequence Parsimony Method http://cmgm.stanford.edu/phylip/protpars.html (1993)
Néron, B. et al. Mobyle: a new full web bioinformatics framework. Bioinformatics 25, 3005–3011 (2009)
Chevenet, F., Brun, C., Bañuls, A. L., Jacq, B. & Christen, R. TreeDyn: towards dynamic graphics and annotations for analyses of trees. BMC Bioinformatics 7, 439 (2006)
Letunic, I. & Bork, P. Interactive Tree Of Life (iTOL): an online tool for phylogenetic tree display and annotation. Bioinformatics 23, 127–128 (2007)
Cherny, V. V. & DeCoursey, T. E. pH-dependent inhibition of voltage-gated H+ currents in rat alveolar epithelial cells by Zn2+ and other divalent cations. J. Gen. Physiol. 114, 819–838 (1999)
Acknowledgements
We thank P. H. Barry, D. Gillespie, V. S. Markin, J. F. Nagle, R. Pomès, D. Silverman and V. Sokolov for discussions or comments on the manuscript. Supported by NSF grant MCB-0943362 (S.M.E.S. and T.E.D.) and NIH grant GM087507 (T.E.D.). The content is solely the responsibility of the authors and does not necessarily represent the views of the National Institute of General Medical Sciences or the National Institutes of Health.
Author information
Authors and Affiliations
Contributions
S.R. identified the similarity of C15orf27 to HV1 and cloned the C15orf27 gene; S.M.E.S. conceived the strategic approach based on molecular model, sequence and phylogenetic analysis; S.R. and S.M.E.S. created mutants; T.E.D., B.M. and V.V.C. designed experiments; B.M., D.M. and V.V.C. recorded, analysed and interpreted data; T.E.D. wrote the manuscript; all authors read and approved the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
The file contains Supplementary Figures 1-9 with legends, Supplementary Tables 1-4 and additional references. (PDF 2495 kb)
Rights and permissions
About this article
Cite this article
Musset, B., Smith, S., Rajan, S. et al. Aspartate 112 is the selectivity filter of the human voltage-gated proton channel. Nature 480, 273–277 (2011). https://doi.org/10.1038/nature10557
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature10557
This article is cited by
-
Ion currents through the voltage sensor domain of distinct families of proteins
Journal of Biological Physics (2023)
-
Role of the Voltage-Gated Proton Channel Hv1 in Nervous Systems
Neuroscience Bulletin (2023)
-
Intracellular pH regulation: characterization and functional investigation of H+ transporters in Stylophora pistillata
BMC Molecular and Cell Biology (2021)
-
Direct activation of the proton channel by albumin leads to human sperm capacitation and sustained release of inflammatory mediators by neutrophils
Nature Communications (2021)
-
Voltage-gated proton channels from fungi highlight role of peripheral regions in channel activation
Communications Biology (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.