Abstract
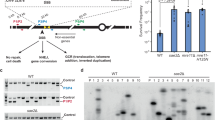
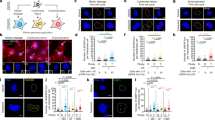
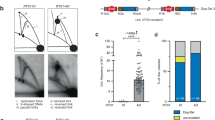
We have investigated the ability of different regions of the left arm of Saccharomyces cerevisiae chromosome V to participate in the formation of gross chromosomal rearrangements (GCRs). We found that the 4.2-kilobase HXT13-DSF1 region sharing divergent homology with chromosomes IV, X and XIV, similar to mammalian segmental duplications, was ‘at risk’ for participating in duplication-mediated GCRs generated by homologous recombination. Numerous genes and pathways, including SGS1, TOP3, RMI1, SRS2, RAD6, SLX1, SLX4, SLX5, MSH2, MSH6, RAD10 and the DNA replication stress checkpoint requiring MRC1 and TOF1, were highly specific for suppressing these GCRs compared to GCRs mediated by single-copy sequences. These results indicate that the mechanisms for formation and suppression of rearrangements occurring in regions containing at-risk sequences differ from those occurring in regions of single-copy sequence. This explains how extensive genome instability is prevented in eukaryotic cells whose genomes contain numerous divergent repeated sequences.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
Accession codes
Primary accessions
ArrayExpress
Data deposits
Microarray data have been submitted to ArrayExpress (http://www.ebi.ac.uk/arrayexpress) with accession number E-TABM-714.
References
Online Mendelian Inheritance in Man, OMIM. Institute of Genetic Medicine, Johns Hopkins University and National Center for Biotechnology Information, National Library of Medicine. 〈http://www.ncbi.nlm.nih.gov/omim/〉 (1999)
Stankiewicz, P. & Lupski, J. R. The genomic basis of disease, mechanisms and assays for genomic disorders. Genome Dyn. 1, 1–16 (2006)
Mitelman, F. Catalog of Chromosome Aberrations in Cancer (Wiley Liss, 1991)
Gorringe, K. L. et al. Evidence that both genetic instability and selection contribute to the accumulation of chromosome alterations in cancer. Carcinogenesis 26, 923–930 (2005)
Lengauer, C., Kinzler, K. W. & Vogelstein, B. Genetic instability in colorectal cancers. Nature 386, 623–627 (1997)
Ribas, M. et al. The structural nature of chromosomal instability in colon cancer cells. FASEB J. 17, 289–291 (2003)
Hoeijmakers, J. H. Genome maintenance mechanisms for preventing cancer. Nature 411, 366–374 (2001)
Deininger, P. L. & Batzer, M. A. Alu repeats and human disease. Mol. Genet. Metab. 67, 183–193 (1999)
Gordenin, D. A. & Resnick, M. A. Yeast ARMs (DNA at-risk motifs) can reveal sources of genome instability. Mutat. Res. 400, 45–58 (1998)
Batzer, M. A. & Deininger, P. L. Alu repeats and human genomic diversity. Nature Rev. Genet. 3, 370–379 (2002)
Ji, Y., Eichler, E. E., Schwartz, S. & Nicholls, R. D. Structure of chromosomal duplicons and their role in mediating human genomic disorders. Genome Res. 10, 597–610 (2000)
Harris, S., Rudnicki, K. S. & Haber, J. E. Gene conversions and crossing over during homologous and homeologous ectopic recombination in Saccharomyces cerevisiae . Genetics 135, 5–16 (1993)
Umezu, K., Hiraoka, M., Mori, M. & Maki, H. Structural analysis of aberrant chromosomes that occur spontaneously in diploid Saccharomyces cerevisiae: retrotransposon Ty1 plays a crucial role in chromosomal rearrangements. Genetics 160, 97–110 (2002)
Lemoine, F. J., Degtyareva, N. P., Lobachev, K. & Petes, T. D. Chromosomal translocations in yeast induced by low levels of DNA polymerase a model for chromosome fragile sites. Cell 120, 587–598 (2005)
Lobachev, K. S. et al. Inverted Alu repeats unstable in yeast are excluded from the human genome. EMBO J. 19, 3822–3830 (2000)
Chen, C. & Kolodner, R. D. Gross chromosomal rearrangements in Saccharomyces cerevisiae replication and recombination defective mutants. Nature Genet. 23, 81–85 (1999)
Putnam, C. D., Pennaneach, V. & Kolodner, R. D. Saccharomyces cerevisiae as a model system to define the chromosomal instability phenotype. Mol. Cell. Biol. 25, 7226–7238 (2005)
Eichler, E. E. Recent duplication, domain accretion and the dynamic mutation of the human genome. Trends Genet. 17, 661–669 (2001)
Bosco, G. & Haber, J. E. Chromosome break-induced DNA replication leads to nonreciprocal translocations and telomere capture. Genetics 150, 1037–1047 (1998)
Deem, A. et al. Defective break-induced replication leads to half-crossovers in Saccharomyces cerevisiae . Genetics 179, 1845–1860 (2008)
Boulton, S. J. & Jackson, S. P. Components of the Ku-dependent non-homologous end-joining pathway are involved in telomeric length maintenance and telomeric silencing. EMBO J. 17, 1819–1828 (1998)
Smith, C. E., Llorente, B. & Symington, L. S. Template switching during break-induced replication. Nature 447, 102–105 (2007)
Schmidt, K. H., Wu, J. & Kolodner, R. D. Control of translocations between highly diverged genes by Sgs1, the Saccharomyces cerevisiae homolog of the Bloom’s syndrome protein. Mol. Cell. Biol. 26, 5406–5420 (2006)
Oh, S. D. et al. BLM ortholog, Sgs1, prevents aberrant crossing-over by suppressing formation of multichromatid joint molecules. Cell 130, 259–272 (2007)
Myung, K., Chen, C. & Kolodner, R. D. Multiple pathways cooperate in the suppression of genome instability in Saccharomyces cerevisiae . Nature 411, 1073–1076 (2001)
Krogh, B. O. & Symington, L. S. Recombination proteins in yeast. Annu. Rev. Genet. 38, 233–271 (2004)
Bai, Y. & Symington, L. S. A. Rad52 homolog is required for RAD51-independent mitotic recombination in Saccharomyces cerevisiae . Genes Dev. 10, 2025–2037 (1996)
Spell, R. M. & Jinks-Robertson, S. Examination of the roles of Sgs1 and Srs2 helicases in the enforcement of recombination fidelity in Saccharomyces cerevisiae . Genetics 168, 1855–1865 (2004)
Myung, K., Datta, A., Chen, C. & Kolodner, R. D. SGS1, the Saccharomyces cerevisiae homologue of BLM and WRN, suppresses genome instability and homeologous recombination. Nature Genet. 27, 113–116 (2001)
Sugawara, N., Goldfarb, T., Studamire, B., Alani, E. & Haber, J. E. Heteroduplex rejection during single-strand annealing requires Sgs1 helicase and mismatch repair proteins Msh2 and Msh6 but not Pms1. Proc. Natl Acad. Sci. USA 101, 9315–9320 (2004)
Mullen, J. R., Nallaseth, F. S., Lan, Y. Q., Slagle, C. E. & Brill, S. J. Yeast Rmi1/Nce4 controls genome stability as a subunit of the Sgs1-Top3 complex. Mol. Cell. Biol. 25, 4476–4487 (2005)
Lydeard, J. R., Jain, S., Yamaguchi, M. & Haber, J. E. Break-induced replication and telomerase-independent telomere maintenance require Pol32. Nature 448, 820–823 (2007)
Zou, H. & Rothstein, R. Holliday junctions accumulate in replication mutants via a RecA homolog-independent mechanism. Cell 90, 87–96 (1997)
Hwang, J. Y., Smith, S. & Myung, K. The Rad1-Rad10 complex promotes the production of gross chromosomal rearrangements from spontaneous DNA damage in Saccharomyces cerevisiae . Genetics 169, 1927–1937 (2005)
Roberts, T. M., Zaidi, I. W., Vaisica, J. A., Peter, M. & Brown, G. W. Regulation of Rtt107 recruitment to stalled DNA replication forks by the cullin Rtt101 and the Rtt109 acetyltransferase. Mol. Biol. Cell 19, 171–180 (2008)
Conaway, R. C. & Conaway, J. W. The INO80 chromatin remodeling complex in transcription, replication and repair. Trends Biochem. Sci. 34, 71–77 (2009)
Mayer, M. L., Gygi, S. P., Aebersold, R. & Hieter, P. Identification of RFC(Ctf18p, Ctf8p, Dcc1p): an alternative RFC complex required for sister chromatid cohesion in S. cerevisiae . Mol. Cell 7, 959–970 (2001)
Osborn, A. J. & Elledge, S. J. Mrc1 is a replication fork component whose phosphorylation in response to DNA replication stress activates Rad53. Genes Dev. 17, 1755–1767 (2003)
Katou, Y. et al. S-phase checkpoint proteins Tof1 and Mrc1 form a stable replication-pausing complex. Nature 424, 1078–1083 (2003)
Mullen, J. R., Kaliraman, V., Ibrahim, S. S. & Brill, S. J. Requirement for three novel protein complexes in the absence of the Sgs1 DNA helicase in Saccharomyces cerevisiae . Genetics 157, 103–118 (2001)
Schmidt, K. H. & Kolodner, R. D. Requirement of Rrm3 helicase for repair of spontaneous DNA lesions in cells lacking Srs2 or Sgs1 helicase. Mol. Cell. Biol. 24, 3213–3226 (2004)
Fabre, F., Chan, A., Heyer, W. D. & Gangloff, S. Alternate pathways involving Sgs1/Top3, Mus81/Mms4, and Srs2 prevent formation of toxic recombination intermediates from single-stranded gaps created by DNA replication. Proc. Natl Acad. Sci. USA 99, 16887–16892 (2002)
Torres, J. Z., Schnakenberg, S. L. & Zakian, V. A. Saccharomyces cerevisiae Rrm3p DNA helicase promotes genome integrity by preventing replication fork stalling: viability of rrm3 cells requires the intra-S-phase checkpoint and fork restart activities. Mol. Cell. Biol. 24, 3198–3212 (2004)
Sugawara, N., Paques, F., Colaiacovo, M. & Haber, J. E. Role of Saccharomyces cerevisiae Msh2 and Msh3 repair proteins in double-strand break-induced recombination. Proc. Natl Acad. Sci. USA 94, 9214–9219 (1997)
Fiorentini, P., Huang, K. N., Tishkoff, D. X., Kolodner, R. D. & Symington, L. S. Exonuclease I of Saccharomyces cerevisiae functions in mitotic recombination in vivo and in vitro . Mol. Cell. Biol. 17, 2764–2773 (1997)
Krejci, L. et al. DNA helicase Srs2 disrupts the Rad51 presynaptic filament. Nature 423, 305–309 (2003)
Prakash, R. et al. Yeast Mph1 helicase dissociates Rad51-made D-loops: implications for crossover control in mitotic recombination. Genes Dev. 23, 67–79 (2009)
Veaute, X. et al. The Srs2 helicase prevents recombination by disrupting Rad51 nucleoprotein filaments. Nature 423, 309–312 (2003)
Hoege, C., Pfander, B., Moldovan, G. L., Pyrowolakis, G. & Jentsch, S. RAD6-dependent DNA repair is linked to modification of PCNA by ubiquitin and SUMO. Nature 419, 135–141 (2002)
Chen, C. C. et al. Acetylated lysine 56 on histone H3 drives chromatin assembly after repair and signals for the completion of repair. Cell 134, 231–243 (2008)
Sikorski, R. S. & Hieter, P. A system of shuttle vectors and yeast host strains designed for efficient manipulation of DNA in Saccharomyces cerevisiae . Genetics 122, 19–27 (1989)
Alani, E., Cao, L. & Kleckner, N. A method for gene disruption that allows repeated use of URA3 selection in the construction of multiply disrupted yeast strains. Genetics 116, 541–545 (1987)
Acknowledgements
We thank the UCSD Microarray Core Facility for assistance in the aCGH experiments and C. Smith, S. Shell and J. Petrini for comments on the manuscript. This work was supported by NIH grant GM26017.
Author Contributions C.D.P., R.D.K. and T.K.H. designed the experiments. C.D.P. and T.K.H. performed the experiment. C.D.P. and R.D.K. analysed the data and wrote the manuscript.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Information
This file contains Supplementary Tables 1-2 and Supplementary Figures 1-3 with Legends. (PDF 310 kb)
PowerPoint slides
Rights and permissions
About this article
Cite this article
Putnam, C., Hayes, T. & Kolodner, R. Specific pathways prevent duplication-mediated genome rearrangements. Nature 460, 984–989 (2009). https://doi.org/10.1038/nature08217
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature08217
This article is cited by
-
Insufficient evidence for non-neutrality of synonymous mutations
Nature (2023)
-
Double-strand breaks induce inverted duplication chromosome rearrangements by a DNA polymerase δ-dependent mechanism
Nature Communications (2023)
-
Effects of the loss of mismatch repair genes on single-strand annealing between divergent sequences in Saccharomyces cerevisiae
Journal of Microbiology (2021)
-
Analyses of DNA double-strand break repair pathways in tandem arrays of HXT genes of Saccharomyces cerevisiae
Journal of Microbiology (2020)
-
The Swr1 chromatin-remodeling complex prevents genome instability induced by replication fork progression defects
Nature Communications (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.