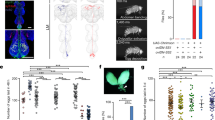
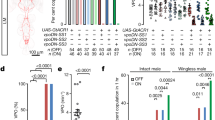
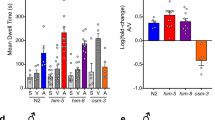
Abstract
Mating in many species induces a dramatic switch in female reproductive behaviour. In most insects, this switch is triggered by factors present in the male’s seminal fluid. How these factors exert such profound effects in females is unknown. Here we identify a receptor for the Drosophila melanogaster sex peptide (SP, also known as Acp70A), the primary trigger of post-mating responses in this species. Females that lack the sex peptide receptor (SPR, also known as CG16752), either entirely or only in the nervous system, fail to respond to SP and continue to show virgin behaviours even after mating. SPR is expressed in the female’s reproductive tract and central nervous system. The behavioural functions of SPR map to the subset of neurons that also express the fruitless gene, a key determinant of sex-specific reproductive behaviour. SPR is highly conserved across insects, opening up the prospect of new strategies to control the reproductive and host-seeking behaviours of agricultural pests and human disease vectors.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Accession codes
Primary accessions
GenBank/EMBL/DDBJ
Data deposits
Nucleotide sequences and translations of the reported SPRs have been deposited in the GenBank database under the following accession numbers: D. pseudoobscura, EU106873; A. aegypti, EU106874; A. gambiae, EU106875; B. mori, EU106876; and T. castaneum, EU106877.
References
Arthur, B. I., Jallon, J. M., Caflisch, B., Choffat, Y. & Nothiger, R. Sexual behaviour in Drosophila is irreversibly programmed during a critical period. Curr. Biol. 8, 1187–1190 (1998)
Morris, J. A., Jordan, C. L. & Breedlove, S. M. Sexual differentiation of the vertebrate nervous system. Nature Neurosci. 7, 1034–1039 (2004)
Munday, P. L., Buston, P. M. & Warner, R. R. Diversity and flexibility of sex-change strategies in animals. Trends Ecol. Evol. 21, 89–95 (2006)
Demir, E. & Dickson, B. J. fruitless splicing specifies male courtship behavior in Drosophila . Cell 121, 785–794 (2005)
Manoli, D. S. et al. Male-specific fruitless specifies the neural substrates of Drosophila courtship behaviour. Nature 436, 395–400 (2005)
Gillott, C. Male accessory gland secretions: modulators of female reproductive physiology and behavior. Annu. Rev. Entomol. 48, 163–184 (2003)
Chen, P. S. et al. A male accessory gland peptide that regulates reproductive behavior of female D. melanogaster . Cell 54, 291–298 (1988)
Chapman, T. et al. The sex peptide of Drosophila melanogaster: female post-mating responses analyzed by using RNA interference. Proc. Natl Acad. Sci. USA 100, 9923–9928 (2003)
Liu, H. & Kubli, E. Sex-peptide is the molecular basis of the sperm effect in Drosophila melanogaster . Proc. Natl Acad. Sci. USA 100, 9929–9933 (2003)
Kvitsiani, D. & Dickson, B. J. Shared neural circuitry for female and male sexual behaviours in Drosophila . Curr. Biol. 16, R355–R356 (2006)
Dietzl, G. et al. A genome-wide transgenic RNAi library for conditional gene inactivation in Drosophila . Nature 448, 151–156 (2007)
Parks, A. L. et al. Systematic generation of high-resolution deletion coverage of the Drosophila melanogaster genome. Nature Genet. 36, 288–292 (2004)
Santel, A., Winhauer, T., Blumer, N. & Renkawitz-Pohl, R. The Drosophila don juan (dj) gene encodes a novel sperm specific protein component characterized by an unusual domain of a repetitive amino acid motif. Mech. Dev. 64, 19–30 (1997)
Schmidt, T., Choffat, Y., Klauser, S. & Kubli, E. The Drosophila melanogaster Sex-peptide: a molecular analysis of structure–function relationships. J. Insect Physiol. 39, 361–368 (1993)
Le Poul, E., Hisada, S., Mizuguchi, Y., Dupriez, V. J., Burgeon, E. & Detheux, M. Adaptation of aequorin functional assay to high throughput screening. J. Biomol. Screen. 7, 57–65 (2002)
Harshman, L. G., Loeb, A. M. & Johnson & B. A Ecdysteroid titers in mated and unmated Drosophila melanogaster females. J. Insect Physiol. 45, 571–577 (1999)
Chapman, T., Choffat, Y., Lucas, W. E., Kubli, E. & Partridge, L. Lack of response to sex-peptide results in increased cost of mating in dunce Drosophila melanogaster females. J. Insect Physiol. 42, 1007–1015 (1996)
Conklin, B. R., Farfel, Z., Lustig, K. D., Julius, D. & Bourne, H. R. Substitution of three amino acids switches receptor specificity of Gqα to that of Giα. Nature 363, 274–276 (1993)
Conklin, B. R. et al. Carboxyl-terminal mutations of Gqα and Gsα that alter the fidelity of receptor activation. Mol. Pharmacol. 50, 885–890 (1996)
Meeusen, T. et al. Identification in Drosophila melanogaster of the invertebrate G protein-coupled FMRFamide receptor. Proc. Natl Acad. Sci. USA 99, 15363–15368 (2002)
Cazzamali, G. & Grimmelikhuijzen, C. J. Molecular cloning and functional expression of the first insect FMRFamide receptor. Proc. Natl Acad. Sci. USA 99, 12073–12078 (2002)
Park, Y., Kim, Y.-J. & Adams, M. E. Identification of G protein-coupled receptors for Drosophila PRXamide peptides, CCAP, corazonin, and AKH supports a theory of ligand–receptor coevolution. Proc. Natl Acad. Sci. USA 99, 11423–11428 (2002)
Rosenkilde, C. et al. Molecular cloning, functional expression, and gene silencing of two Drosophila receptors for the Drosophila neuropeptide pyrokinin-2. Biochem. Biophys. Res. Commun. 309, 485–494 (2003)
Saudan, P. et al. Ductus ejaculatorius peptide 99B (DUP99B), a novel Drosophila melanogaster sex-peptide pheromone. Eur. J. Biochem. 269, 989–997 (2002)
Bloch Qazi, M. C., Heifetz, Y. & Wolfner, M. F. The developments between gametogenesis and fertilization: ovulation and female sperm storage in Drosophila melanogaster . Dev. Biol. 256, 195–211 (2003)
Kubli, E. Sex-peptides: seminal peptides of the Drosophila male. Cell. Mol. Life Sci. 60, 1689–1704 (2003)
Peng, J. et al. Gradual release of sperm bound sex-peptide controls female postmating behavior in Drosophila . Curr. Biol. 15, 207–213 (2005)
Ottiger, M., Soller, M., Stocker, R. F. & Kubli, E. Binding sites of Drosophila melanogaster sex peptide pheromones. J. Neurobiol. 44, 57–71 (2000)
Stockinger, P., Kvitsiani, D., Rotkopf, S., Tirian, L. & Dickson, B. J. Neural circuitry that governs Drosophila male courtship behavior. Cell 121, 795–807 (2005)
Fan, Y., Rafaeli, A., Gileadi, C., Kubli, E. & Applebaum, S. W. Drosophila melanogaster sex peptide stimulates juvenile hormone synthesis and depresses sex pheromone production in Helicoverpa armigera . J. Insect Physiol. 45, 127–133 (1999)
Fan, Y. et al. Common functional elements of Drosophila melanogaster seminal peptides involved in reproduction of Drosophila melanogaster and Helicoverpa armigera females. Insect Biochem. Mol. Biol. 30, 805–812 (2000)
Brand, A. H. & Perrimon, N. Targeted gene expression as a means of altering cell fates and generating dominant phenotypes. Development 118, 401–415 (1993)
Groth, A. C., Fish, M., Nusse, R. & Calos, M. P. Construction of transgenic Drosophila by using the site-specific integrase from phage phiC31. Genetics 166, 1775–1782 (2004)
Luo, L., Liao, Y. J., Jan, L. Y. & Jan, Y. N. Distinct morphogenetic functions of similar small GTPases: Drosophila Drac1 is involved in axonal outgrowth and myoblast fusion. Genes Dev. 8, 1787–1802 (1994)
Kim, Y.-J. et al. Corazonin receptor signaling in ecdysis initiation. Proc. Natl Acad. Sci. USA 101, 6704–6709 (2004)
Aza-Blanc, P., Lin, H. Y., Ruiz i Altaba, A. & Kornberg, T. B. Expression of the vertebrate Gli proteins in Drosophila reveals a distribution of activator and repressor activities. Development 127, 4293–4301 (2000)
Backhaus, B., Sulkowski, E. & Schlote, F. W. A semi-synthetic, general-purpose medium for Drosophila melanogaster . Dros. Inf. Serv. 60, 210–212 (1984)
Johnson, E. C. et al. Identification of Drosophila neuropeptide receptors by G protein-coupled receptors–β-arrestin2 interactions. J. Biol. Chem. 278, 52172–52178 (2003)
Vernon, W. I. & Printen, J. A. Assay for intracellular calcium using a codon-optimized aequorin. Biotechniques 33, 730–734 (2002)
O’Neill, E. M., Rebay, I., Tjian, R. & Rubin, G. M. The activities of two Ets-related transcription factors required for Drosophila eye development are modulated by the Ras/MAPK pathway. Cell 78, 137–147 (1994)
Altschul, S. F., Gish, W., Miller, W., Myers, E. W. & Lipman, D. J. Basic local alignment search tool. J. Mol. Biol. 215, 403–410 (1990)
Eddy, S. R. Profile hidden Markov models. Bioinformatics 14, 755–763 (1998)
Edgar, R. C. MUSCLE: a multiple sequence alignment method with reduced time and space complexity. BMC Bioinformatics 5, 113 (2004)
Jeanmougin, F., Thompson, J. D., Gouy, M., Higgins, D. G. & Gibson, T. J. Multiple sequence alignment with Clustal X. Trends Biochem. Sci. 23, 403–405 (1998)
Felsenstein, J. PHYLIP (Phylogeny Inference Package) version 3.6. Distributed by the author. Department of Genome Sciences, University of Washington, Seattle. (2005)
Acknowledgements
We thank A. Schleiffer for preparation of the phylogenetic tree, R. Fuchs and G. Krssakova for technical assistance, M. Calos, K-C. Su and S. Oppel for φC31 reagents, and G. Bucher and D. Zitnan for insect stocks. We particularly thank E. Kubli for many reagents and discussions. Y-J.K. was supported by a Lise Meitner postdoctoral fellowship from the Austrian Science Fund, and C.R. was supported by an EMBO postdoctoral fellowship and an Advanced Researcher fellowship from the Swiss National Science Foundation. Basic research at the IMP is funded by Boehringer Ingelheim GmbH.
Author Contributions N.Y. and C.R. identified D. melanogaster SPR in the RNAi screen, N.Y. performed the initial molecular analysis and all behavioural assays, and Y.-J.K. performed the cellular assays and immunohistochemistry and cloned SPR orthologues from other insects. B.J.D. supervised the project and wrote the manuscript together with N.Y. and Y-J.K.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Information
The file contains Supplementary Figures S1-S3 with Legends (PDF 2146 kb)
Rights and permissions
About this article
Cite this article
Yapici, N., Kim, YJ., Ribeiro, C. et al. A receptor that mediates the post-mating switch in Drosophila reproductive behaviour. Nature 451, 33–37 (2008). https://doi.org/10.1038/nature06483
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature06483
This article is cited by
-
Transcriptional programs are activated and microRNAs are repressed within minutes after mating in the Drosophila melanogaster female reproductive tract
BMC Genomics (2023)
-
Heterogeneous receptor expression underlies non-uniform peptidergic modulation of olfaction in Drosophila
Nature Communications (2023)
-
Flexible neural control of transition points within the egg-laying behavioral sequence in Drosophila
Nature Neuroscience (2023)
-
Female factors modulate Sex Peptide’s association with sperm in Drosophila melanogaster
BMC Biology (2022)
-
Ethology of Sunn-pest oviposition in interaction with deltamethrin loaded on mesoporous silica nanoparticles as a nanopesticide
Chemical and Biological Technologies in Agriculture (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.