Abstract
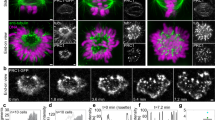
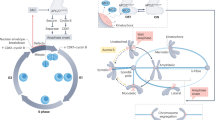
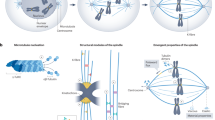
Accurate segregation of chromosomes, essential for the stability of the genome, depends on ‘bi-orientation’—simultaneous attachment of each individual chromosome to both poles of the mitotic spindle1. On bi-oriented chromosomes, kinetochores (macromolecular complexes that attach the chromosome to the spindle) reside on the opposite sides of the chromosome’s centromere2. In contrast, sister kinetochores shift towards one side of the centromere on ‘syntelic’ chromosomes that erroneously attach to one spindle pole with both sister kinetochores. Syntelic attachments often arise during spindle assembly and must be corrected to prevent chromosome loss3. It is assumed that restoration of proper centromere architecture occurs automatically owing to elastic properties of the centromere1,2. Here we test this assumption by combining laser microsurgery and chemical biology assays in cultured mammalian cells. We find that kinetochores of syntelic chromosomes remain juxtaposed on detachment from spindle microtubules. These findings reveal that correction of syntelic attachments involves an extra step that has previously been overlooked: external forces must be applied to move sister kinetochores to the opposite sides of the centromere. Furthermore, we demonstrate that the shape of the centromere is important for spindle assembly, because bipolar spindles do not form in cells lacking centrosomes when multiple chromosomes with juxtaposed kinetochores are present. Thus, proper architecture of the centromere makes an important contribution to achieving high fidelity of chromosome segregation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Nicklas, R. B. How cells get the right chromosomes. Science 275, 632–637 (1997)
Rieder, C. L. & Salmon, E. D. The vertebrate kinetochore and its roles during mitosis. Trends Cell Biol. 8, 310–318 (1998)
Cimini, D. & Degrassi, F. Aneuploidy: a matter of bad connections. Trends Cell Biol. 8, 442–451 (2005)
Ault, J. G. & Nicklas, R. B. Tension, microtubule rearrangements, and the proper distribution of chromosomes in mitosis. Chromosoma 98, 33–39 (1989)
Nicklas, R. B. & Ward, S. C. Elements of error correction in mitosis: microtubule capture, release, and tension. J. Cell Biol. 126, 1241–1253 (1994)
Lampson, M. A., Renduchitala, K., Khodjakov, A. & Kapoor, T. M. Correcting improper chromosome–spindle attachments during cell division. Nature Cell Biol. 6, 232–237 (2004)
Kline-Smith, S. L., Khodjakov, A., Hergert, P. & Walczak, C. E. Depletion of centromeric MCAK leads to chromosome congression and segregation defects due to improper kinetochore attachments. Mol. Biol. Cell 15, 1146–1159 (2004)
Andrews, P. D. et al. Aurora B regulates MCAK at the mitotic centromere. Dev. Cell 6, 253–268 (2004)
Sullivan, K. F. & Shelby, R. D. Using time-lapse confocal microscopy for analysis of centromere dynamics in human cells. Methods Cell Biol. 58, 183–202 (1999)
Poirier, M. G. & Marko, J. F. Micromechanical studies of mitotic chromosomes. Curr. Top. Dev. Biol. 55, 75–141 (2003)
Kapoor, T. M., Mayer, T. U., Coughlin, M. L. & Mitchison, T. J. Probing spindle assembly mechanisms with monastrol, a small molecule inhibitor of the mitotic kinesin, Eg5. J. Cell Biol. 150, 975–988 (2000)
Khodjakov, A., Copenagle, L., Gordon, M. B., Compton, D. A. & Kapoor, T. M. Minus-end capture of preformed kinetochore fibers contributes to spindle morphogenesis. J. Cell Biol. 160, 671–683 (2003)
Khodjakov, A. & Rieder, C. L. The sudden recruitment of γ-tubulin to the centrosome at the onset of mitosis and its dynamic exchange throughout the cell cycle, do not require microtubules. J. Cell Biol. 146, 585–596 (1999)
Khodjakov, A., Cole, R. W., Oakley, B. R. & Rieder, C. L. Centrosome-independent mitotic spindle formation in vertebrates. Curr. Biol. 10, 59–67 (2000)
Khodjakov, A. & Rieder, C. L. Centrosomes enhance the fidelity of cytokinesis in vertebrates and are required for cell cycle progression. J. Cell Biol. 153, 237–242 (2001)
Compton, D. A. Focusing on spindle poles. J. Cell Sci. 111, 1477–1481 (1998)
Rieder, C. L. & Alexander, S. P. Kinetochores are transported poleward along a single astral microtubule during chromosome attachment to the spindle in newt lung cells. J. Cell Biol. 110, 81–95 (1990)
Maiato, H., Rieder, C. L. & Khodjakov, A. Kinetochore-driven formation of kinetochore fibers contributes to spindle assembly during animal mitosis. J. Cell Biol. 167, 831–840 (2004)
Tulu, U. S., Fagerstrom, C., Ferenz, N. P. & Wadsworth, P. Molecular requirements for kinetochore-associated microtubule formation in mammalian cells. Curr. Biol. 16, 536–541 (2006)
Indjeian, V. B., Stern, B. M. & Murray, A. W. The centromeric protein Sgo1 is required to sense lack of tension on mitotic chromosomes. Science 307, 130–133 (2005)
Li, X. & Nicklas, R. B. Mitotic forces control a cell-cycle checkpoint. Nature 373, 630–632 (1995)
Waters, J. C., Chen, R.-H., Murray, A. W. & Salmon, E. D. Localization of Mad2 to kinetochores depends on microtubule attachment, not tension. J. Cell Biol. 141, 1181–1191 (1998)
Cimini, D. et al. Merotelic kinetochore orientation is a major mechanism of aneuploidy in mitotic mammalian tissue cells. J. Cell Biol. 153, 517–528 (2001)
Rieder, C. L., Cole, R. W., Khodjakov, A. & Sluder, G. The checkpoint delaying anaphase in response to chromosome monoorientation is mediated by an inhibitory signal produced by unattached kinetochores. J. Cell Biol. 130, 941–948 (1995)
Rieder, C. L., Schultz, A., Cole, R. & Sluder, G. Anaphase onset in vertebrate somatic cells is controlled by a checkpoint that monitors sister kinetochore attachment to the spindle. J. Cell Biol. 127, 1301–1310 (1994)
Cassimeris, L., Rieder, C. L. & Salmon, E. D. Microtubule assembly and kinetochore directional instability in vertebrate monopolar spindles: implications for the mechanism of chromosome congression. J. Cell Sci. 107, 285–297 (1994)
Kapoor, T. M. et al. Chromosomes can congress to the metaphase plate before biorientation. Science 311, 388–391 (2006)
Magidson, V., Loncarek, J., Hergert, P., Rieder, C. L. & Khodjakov, A. in Laser Manipulations of Cells and Tissues (eds Berns, M. W. & Greulich, K. O.) 237–266 (Elsevier, 2007)
Mayer, T. U. et al. Small molecule inhibitor of mitotic spindle bipolarity identified in a phenotype-based screen. Science 286, 971–974 (1999)
La Terra, S. et al. The de novo centriole assembly pathway in HeLa cells: cell cycle progression and centriole assembly/maturation. J. Cell Biol. 168, 713–720 (2005)
Khodjakov, A., Cole, R. W., McEwen, B. F., Buttle, K. F. & Rieder, C. L. Chromosome fragments possessing only one kinetochore can congress to the spindle equator. J. Cell Biol. 136, 229–240 (1997)
Acknowledgements
We thank B. McEwen, C. Rieder and V. Magidson for fruitful discussions and critical reading of the manuscript. This work was supported by grants from the National Institutes of Health grants (to A.K. and T.M.K.). Assembly of our laser microsurgery system was supported in part by a Nikon/MBL fellowship (A.K.). We acknowledge the use of Wadsworth Centre’s electron microscopy core facility.
Author information
Authors and Affiliations
Corresponding authors
Supplementary information
Supplementary Figures
The file contains Supplementary Figures S1-S6 with Legends. (PDF 2537 kb)
Rights and permissions
About this article
Cite this article
Lončarek, J., Kisurina-Evgenieva, O., Vinogradova, T. et al. The centromere geometry essential for keeping mitosis error free is controlled by spindle forces. Nature 450, 745–749 (2007). https://doi.org/10.1038/nature06344
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature06344
This article is cited by
-
Defining a core configuration for human centromeres during mitosis
Nature Communications (2023)
-
Mechanical Torque Promotes Bipolarity of the Mitotic Spindle Through Multi-centrosomal Clustering
Bulletin of Mathematical Biology (2022)
-
Prc1-rich kinetochores are required for error-free acentrosomal spindle bipolarization during meiosis I in mouse oocytes
Nature Communications (2020)
-
Spindle tubulin and MTOC asymmetries may explain meiotic drive in oocytes
Nature Communications (2018)
-
Chromosome missegregation during anaphase triggers p53 cell cycle arrest through histone H3.3 Ser31 phosphorylation
Nature Cell Biology (2016)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.