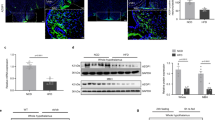
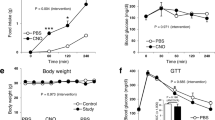
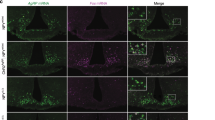
Abstract
A subset of neurons in the brain, known as ‘glucose-excited’ neurons, depolarize and increase their firing rate in response to increases in extracellular glucose. Similar to insulin secretion by pancreatic β-cells1, glucose excitation of neurons is driven by ATP-mediated closure of ATP-sensitive potassium (KATP) channels2,3,4,5. Although β-cell-like glucose sensing in neurons is well established, its physiological relevance and contribution to disease states such as type 2 diabetes remain unknown. To address these issues, we disrupted glucose sensing in glucose-excited pro-opiomelanocortin (POMC) neurons5 via transgenic expression of a mutant Kir6.2 subunit (encoded by the Kcnj11 gene) that prevents ATP-mediated closure of KATP channels6,7. Here we show that this genetic manipulation impaired the whole-body response to a systemic glucose load, demonstrating a role for glucose sensing by POMC neurons in the overall physiological control of blood glucose. We also found that glucose sensing by POMC neurons became defective in obese mice on a high-fat diet, suggesting that loss of glucose sensing by neurons has a role in the development of type 2 diabetes. The mechanism for obesity-induced loss of glucose sensing in POMC neurons involves uncoupling protein 2 (UCP2), a mitochondrial protein that impairs glucose-stimulated ATP production8. UCP2 negatively regulates glucose sensing in POMC neurons. We found that genetic deletion of Ucp2 prevents obesity-induced loss of glucose sensing, and that acute pharmacological inhibition of UCP2 reverses loss of glucose sensing. We conclude that obesity-induced, UCP2-mediated loss of glucose sensing in glucose-excited neurons might have a pathogenic role in the development of type 2 diabetes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Ashcroft, F. M., Harrison, D. E. & Ashcroft, S. J. Glucose induces closure of single potassium channels in isolated rat pancreatic beta-cells. Nature 312, 446–448 (1984)
Ashford, M. L., Boden, P. R. & Treherne, J. M. Glucose-induced excitation of hypothalamic neurones is mediated by ATP-sensitive K+ channels. Pflugers Arch. 415, 479–483 (1990)
Miki, T. et al. ATP-sensitive K+ channels in the hypothalamus are essential for the maintenance of glucose homeostasis. Nature Neurosci. 4, 507–512 (2001)
Kang, L., Routh, V. H., Kuzhikandathil, E. V., Gaspers, L. D. & Levin, B. E. Physiological and molecular characteristics of rat hypothalamic ventromedial nucleus glucosensing neurons. Diabetes 53, 549–559 (2004)
Ibrahim, N. et al. Hypothalamic proopiomelanocortin neurons are glucose responsive and express KATP channels. Endocrinology 144, 1331–1340 (2003)
Koster, J. C., Marshall, B. A., Ensor, N., Corbett, J. A. & Nichols, C. G. Targeted overactivity of β cell KATP channels induces profound neonatal diabetes. Cell 100, 645–654 (2000)
Koster, J. C., Sha, Q., Shyng, S. & Nichols, C. G. ATP inhibition of KATP channels: control of nucleotide sensitivity by the N-terminal domain of the Kir6.2 subunit. J. Physiol. (Lond.) 515, 19–30 (1999)
Zhang, C. Y. et al. Uncoupling protein-2 negatively regulates insulin secretion and is a major link between obesity, β cell dysfunction, and type 2 diabetes. Cell 105, 745–755 (2001)
Balthasar, N. et al. Leptin receptor signaling in POMC neurons is required for normal body weight homeostasis. Neuron 42, 983–991 (2004)
Silver, I. A. & Erecinska, M. Extracellular glucose concentration in mammalian brain: continuous monitoring of changes during increased neuronal activity and upon limitation in oxygen supply in normo-, hypo-, and hyperglycemic animals. J. Neurosci. 14, 5068–5076 (1994)
Enriori, P. J. et al. Diet-induced obesity causes severe but reversible leptin resistance in arcuate melanocortin neurons. Cell Metab. 5, 181–194 (2007)
Poitout, V. & Robertson, R. P. Minireview: Secondary β-cell failure in type 2 diabetes–a convergence of glucotoxicity and lipotoxicity. Endocrinology 143, 339–342 (2002)
Lowell, B. B. & Shulman, G. I. Mitochondrial dysfunction and type 2 diabetes. Science 307, 384–387 (2005)
Echtay, K. S. et al. Superoxide activates mitochondrial uncoupling proteins. Nature 415, 96–99 (2002)
Krauss, S., Zhang, C. Y. & Lowell, B. B. A significant portion of mitochondrial proton leak in intact thymocytes depends on expression of UCP2. Proc. Natl Acad. Sci. USA 99, 118–122 (2002)
Laybutt, D. R. et al. Genetic regulation of metabolic pathways in β-cells disrupted by hyperglycemia. J. Biol. Chem. 277, 10912–10921 (2002)
Krauss, S. et al. Superoxide-mediated activation of uncoupling protein 2 causes pancreatic β cell dysfunction. J. Clin. Invest. 112, 1831–1842 (2003)
Joseph, J. W. et al. Uncoupling protein 2 knockout mice have enhanced insulin secretory capacity after a high-fat diet. Diabetes 51, 3211–3219 (2002)
Zhang, C. Y. et al. Genipin inhibits UCP2-mediated proton leak and acutely reverses obesity- and high glucose-induced β cell dysfunction in isolated pancreatic islets. Cell Metab. 3, 417–427 (2006)
Joseph, J. W. et al. Free fatty acid-induced β -cell defects are dependent on uncoupling protein 2 expression. J. Biol. Chem. 279, 51049–51056 (2004)
Horvath, T. L. et al. Brain uncoupling protein 2: uncoupled neuronal mitochondria predict thermal synapses in homeostatic centers. J. Neurosci. 19, 10417–10427 (1999)
Richard, D., Clavel, S., Huang, Q., Sanchis, D. & Ricquier, D. Uncoupling protein 2 in the brain: distribution and function. Biochem. Soc. Trans. 29, 812–817 (2001)
Richard, D. et al. Distribution of the uncoupling protein 2 mRNA in the mouse brain. J. Comp. Neurol. 397, 549–560 (1998)
Mountjoy, P. D., Bailey, S. J. & Rutter, G. A. Inhibition by glucose or leptin of hypothalamic neurons expressing neuropeptide Y requires changes in AMP-activated protein kinase activity. Diabetologia 50, 168–177 (2007)
Burdakov, D., Gerasimenko, O. & Verkhratsky, A. Physiological changes in glucose differentially modulate the excitability of hypothalamic melanin-concentrating hormone and orexin neurons in situ. J. Neurosci. 25, 2429–2433 (2005)
Routh, V. H. Glucosensing neurons in the ventromedial hypothalamic nucleus (VMN) and hypoglycemia-associated autonomic failure (HAAF). Diabetes Metab. Res. Rev. 19, 348–356 (2003)
Dallaporta, M., Perrin, J. & Orsini, J. C. Involvement of adenosine triphosphate-sensitive K+ channels in glucose-sensing in the rat solitary tract nucleus. Neurosci. Lett. 278, 77–80 (2000)
Cowley, M. A. et al. Leptin activates anorexigenic POMC neurons through a neural network in the arcuate nucleus. Nature 411, 480–484 (2001)
Dhillon, H. et al. Leptin directly activates SF1 neurons in the VMH, and this action by leptin is required for normal body-weight homeostasis. Neuron 49, 191–203 (2006)
Perkins, K. L. Cell-attached voltage-clamp and current-clamp recording and stimulation techniques in brain slices. J. Neurosci. Methods 154, 1–18 (2006)
Acknowledgements
We would like to thank J. Koster and C. Nichols for donation of the Kir6.2[ΔN2–30,K185Q]–GFP construct; Z. Yang, L. Christiansen, M. Kramer and S. Skowronek for animal care and technical assistance; and B. Bean for discussions, guidance with electrophysiological experiments and for critical reading of this manuscript. This work was supported by NIH grants (B.B.L., J.K.E., M.A.C., P.J.E.); a Smith Family Pinnacle Award from the American Diabetes Association (J.K.E.); a National Natural Science Foundation of China Outstanding Young Scientist Award; the National Basic Research Program of China (973 Program); the ‘111’ Project; and the Natural Science Foundation of Jiangsu Province (C.-Y.Z.).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
Reprints and permissions information is available at www.nature.com/reprints. The authors declare no competing financial interests.
Supplementary information
Supplementary Information
This file contains Supplementary Data with a full description of studies used to characterize the ATP sensitivity of both wild-type and mutKir6.2 POMC neurons in addition to whole-cell recordings showing effects of glucose, genipin and leptin. Also, Supplementary Figure S1 displays RT-PCR results showing tissue specific expression of mutKir6.2 and characterization of KATP channels in mutKir6.2 POMC neurons; and Supplementary Figure S2 displays whole cell electrophysiological recordings of wild-type and mutKir6.2 POMC neurons showing effects of glucose, genipin and leptin. (PDF 692 kb)
Rights and permissions
About this article
Cite this article
Parton, L., Ye, C., Coppari, R. et al. Glucose sensing by POMC neurons regulates glucose homeostasis and is impaired in obesity. Nature 449, 228–232 (2007). https://doi.org/10.1038/nature06098
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature06098
This article is cited by
-
Association between resting energy expenditure, diet and uncoupling protein 2 in obese women with normal and low resting energy expenditure
Nutrire (2023)
-
Relevance and consequence of chronic inflammation for obesity development
Molecular and Cellular Pediatrics (2023)
-
A lncRNA-encoded mitochondrial micropeptide exacerbates microglia-mediated neuroinflammation in retinal ischemia/reperfusion injury
Cell Death & Disease (2023)
-
Inflammatory Milieu Induces Mitochondrial Alterations and Neuronal Activations in Hypothalamic POMC Neurons in a Time-Dependent Manner
Molecular Neurobiology (2023)
-
The ventromedial hypothalamic nucleus: watchdog of whole-body glucose homeostasis
Cell & Bioscience (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.