Abstract
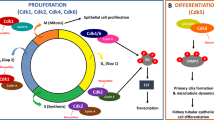
Polycystic kidney diseases (PKDs) are primarily characterized by the growth of fluid-filled cysts in renal tubules leading to end-stage renal disease1,2,3. Mutations in the PKD1 or PKD2 genes lead to autosomal dominant PKD (ADPKD), a slowly developing adult form4,5. Autosomal recessive polycystic kidney disease results from mutations in the PKHD1 gene, affects newborn infants and progresses very rapidly6,7. No effective treatment is currently available for PKD. A previously unrecognized site of subcellular localization was recently discovered for all proteins known to be disrupted in PKD: primary cilia8,9. Because ciliary functions seem to be involved in cell cycle regulation, disruption of proteins associated with cilia or centrioles may directly affect the cell cycle and proliferation, resulting in cystic disease10,11,12. We therefore reasoned that the dysregulated cell cycle may be the most proximal cause of cystogenesis, and that intervention targeted at this point could provide significant therapeutic benefit for PKD. Here we show that treatment with the cyclin-dependent kinase (CDK) inhibitor (R)-roscovitine does indeed yield effective arrest of cystic disease in jck and cpk mouse models of PKD. Continuous daily administration of the drug is not required to achieve efficacy; pulse treatment provides a robust, long-lasting effect, indicating potential clinical benefits for a lifelong therapy. Molecular studies of the mechanism of action reveal effective cell-cycle arrest, transcriptional inhibition and attenuation of apoptosis. We found that roscovitine is active against cysts originating from different parts of the nephron, a desirable feature for the treatment of ADPKD, in which cysts form in multiple nephron segments. Our results indicate that inhibition of CDK is a new and effective approach to the treatment of PKD.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Welling, L. W. & Grantham, J. J. in Renal Pathology (eds Tisher, C. & Brenner, B.) 1828–1863 (Lippincott, Philadelphia, 1996)
Ecder, T. & Schrier, R. W. Hypertension and left ventricular hypertrophy in autosomal dominant polycystic kidney disease. Expert Rev. Cardiovasc. Ther. 2, 369–374 (2004)
Wilson, P. D. Polycystic kidney disease: new understanding in the pathogenesis. Int. J. Biochem. Cell Biol. 36, 1868–1873 (2004)
Hughes, J. et al. The polycystic kidney disease 1 (PKD1) gene encodes a novel protein with multiple cell recognition domains. Nature Genet. 10, 151–160 (1995)
Mochizuki, T. et al. PKD2, a gene for polycystic kidney disease that encodes an integral membrane protein. Science 272, 1339–1342 (1996)
Onuchic, L. F. et al. PKHD1, the polycystic kidney and hepatic disease 1 gene, encodes a novel large protein containing multiple immunoglobulin-like plexin-transcription-factor domains and parallel β-helix 1 repeats. Am. J. Hum. Genet. 70, 1305–1317 (2002)
Ward, C. J. et al. The gene mutated in autosomal recessive polycystic kidney disease encodes a large, receptor-like protein. Nature Genet. 30, 259–269 (2002)
Pazour, G. J. Intraflagellar transport and cilia-dependent renal disease: the ciliary hypothesis of polycystic kidney disease. J. Am. Soc. Nephrol. 15, 2528–2536 (2004)
Pan, J., Wang, Q. & Snell, W. J. Cilium-generated signaling and cilia-related disorders. Lab. Invest. 85, 452–463 (2005)
Bhunia, A. K. et al. PKD1 induces p21waf1 and regulation of the cell cycle via direct activation of the JAK-STAT signaling pathway in a process requiring PKD2. Cell 109, 157–168 (2002)
Quarmby, L. M. & Parker, J. D. Cilia and the cell cycle? J. Cell Biol. 169, 707–710 (2005)
Woo, D. D., Miao, S. Y., Pelayo, J. C. & Woolf, A. S. Taxol inhibits progression of congenital polycystic kidney disease. Nature 368, 750–753 (1994)
Meijer, L. et al. Biochemical and cellular effects of roscovitine, a potent and selective inhibitor of the cyclin-dependent kinases cdc2, cdk2 and cdk5. Eur. J. Biochem. 243, 527–536 (1997)
McClue, S. J. et al. In vitro and in vivo antitumor properties of the cyclin dependent kinase inhibitor CYC202 (R-roscovitine). Int. J. Cancer 102, 463–468 (2002)
MacCallum, D. E. et al. Seliciclib (CYC202, R-Roscovitine) induces cell death in multiple myeloma cells by inhibition of RNA polymerase II-dependent transcription and down-regulation of Mcl-1. Cancer Res. 65, 5399–5407 (2005)
Guzi, T. CYC-202 Cyclacel. Curr. Opin. Investig. Drugs 5, 1311–1318 (2004)
Smith, L. A. et al. Development of polycystic kidney disease in juvenile cystic kidney mice: insights into pathogenesis, ciliary abnormalities, and common features with human disease. J. Am. Soc. Nephrol. 17, 2821–2831 (2006)
Diehl, J. A., Zindy, F. & Sherr, C. J. Inhibition of cyclin D1 phosphorylation on threonine-286 prevents its rapid degradation via the ubiquitin–proteasome pathway. Genes Dev. 11, 957–972 (1997)
Bach, S. et al. Roscovitine targets, protein kinases and pyridoxal kinase. J. Biol. Chem. 280, 31208–31219 (2005)
Omori, S. et al. Extracellular signal-regulated kinase inhibition slows disease progression in mice with polycystic kidney disease. J. Am. Soc. Nephrol. 17, 1604–1614 (2006)
Lin, L., Ye, Y. & Zakeri, Z. p53, Apaf-1, caspase-3, and -9 are dispensable for Cdk5 activation during cell death. Cell Death Differ. 13, 141–150 (2006)
Ecder, T. et al. Caspases, Bcl-2 proteins and apoptosis in autosomal-dominant polycystic kidney disease. Kidney Int. 61, 1220–1230 (2002)
Tao, Y. et al. Caspase inhibition reduces tubular apoptosis and proliferation and slows disease progression in polycystic kidney disease. Proc. Natl Acad. Sci. USA 102, 6954–6959 (2005)
Di Giovanni, S. et al. Cell cycle inhibition provides neuroprotection and reduces glial proliferation and scar formation after traumatic brain injury. Proc. Natl Acad. Sci. USA 102, 8333–8338 (2005)
Verdaguer, E. et al. Antiapoptotic effects of roscovitine in cerebellar granule cells deprived of serum and potassium: a cell cycle-related mechanism. Neurochem. Int. 44, 251–261 (2004)
Tirado, O. M., Mateo-Lozano, S. & Notario, V. Roscovitine is an effective inducer of apoptosis of Ewing’s sarcoma family tumor cells in vitro and in vivo.. Cancer Res. 65, 9320–9327 (2005)
Sweeney, W. E. et al. Combination treatment of PKD utilizing dual inhibition of EGF-receptor activity and ligand bioavailability. Kidney Int. 64, 1310–1319 (2003)
Tao, Y., Kim, J., Schrier, R. W. & Edelstein, C. L. Rapamycin markedly slows disease progression in a rat model of polycystic kidney disease. J. Am. Soc. Nephrol. 16, 46–51 (2005)
Shillingford, J. M. et al. The mTOR pathway is regulated by polycystin-1, and its inhibition reverses renal cystogenesis in polycystic kidney disease. Proc. Natl Acad. Sci. USA 103, 5466–5471 (2006)
Torres, V. E. et al. Effective treatment of an orthologous model of autosomal dominant polycystic kidney disease. Nature Med. 10, 363–364 (2004)
Acknowledgements
We thank R. Russo, T. Barry and A. Taylor for expert technical assistance, the staff of the Genzyme Department of Comparative Medicine and Histology unit for help with in vivo studies and sample preparations. We are grateful to L. Meijer and H. Galons (CNRS, Roscoff) for providing N6-methyl-(R)-roscovitine. We thank A. Smith, R. Gregory, T. Natoli, H. Husson and J. Leonard for helpful discussions and comments on this manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Reprints and permissions information is available at www.nature.com/reprints. The authors declare no competing financial interests.
Supplementary information
Supplementary Notes
This file contains Supplementary Methods, Supplementary Figures 2 and 5 with legends and additional references. (DOC 3723 kb)
Supplementary Table 1
Anti-cystic effect of Roscovitine in jck mouse model of PKD. (DOC 100 kb)
Supplementary Table 2
Anti-cystic effect of Roscovitine in cpk mouse model of PKD. (DOC 38 kb)
Rights and permissions
About this article
Cite this article
Bukanov, N., Smith, L., Klinger, K. et al. Long-lasting arrest of murine polycystic kidney disease with CDK inhibitor roscovitine. Nature 444, 949–952 (2006). https://doi.org/10.1038/nature05348
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature05348
This article is cited by
-
Pathological consequences of DNA damage in the kidney
Nature Reviews Nephrology (2023)
-
Role of cyclin-dependent kinase 2 in the progression of mouse juvenile cystic kidney disease
Laboratory Investigation (2020)
-
Disruption of CUL3-mediated ubiquitination causes proximal tubule injury and kidney fibrosis
Scientific Reports (2019)
-
CCDC114 is mutated in patient with a complex phenotype combining primary ciliary dyskinesia, sensorineural deafness, and renal disease
Journal of Human Genetics (2019)
-
Roscovitine effectively enhances antitumor activity of temozolomide in vitro and in vivo mediated by increased autophagy and Caspase-3 dependent apoptosis
Scientific Reports (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.