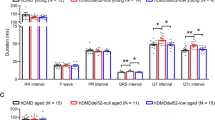
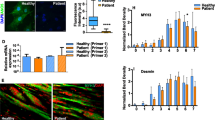
Abstract
Dystrophin deficiency causes Duchenne muscular dystrophy (DMD) in humans, an inherited and progressive disease of striated muscle deterioration that frequently involves pronounced cardiomyopathy1. Heart failure is the second leading cause of fatalities in DMD1,2. Progress towards defining the molecular basis of disease in DMD has mostly come from studies on skeletal muscle, with comparatively little attention directed to cardiac muscle. The pathophysiological mechanisms involved in cardiac myocytes may differ significantly from skeletal myofibres; this is underscored by the presence of significant cardiac disease in patients with truncated or reduced levels of dystrophin but without skeletal muscle disease3. Here we show that intact, isolated dystrophin-deficient cardiac myocytes have reduced compliance and increased susceptibility to stretch-mediated calcium overload, leading to cell contracture and death, and that application of the membrane sealant poloxamer 188 corrects these defects in vitro. In vivo administration of poloxamer 188 to dystrophic mice instantly improved ventricular geometry and blocked the development of acute cardiac failure during a dobutamine-mediated stress protocol. Once issues relating to optimal dosing and long-term effects of poloxamer 188 in humans have been resolved, chemical-based membrane sealants could represent a new therapeutic approach for preventing or reversing the progression of cardiomyopathy and heart failure in muscular dystrophy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Muntoni, F. Cardiomyopathy in muscular dystrophies. Curr. Opin. Neurol. 16, 577–583 (2003)
Emery, A. E. H. in Duchenne Muscular Dystrophy (ed. Emery, A. E. H.) 26–45 (Oxford Univ. Press, Oxford, 2003)
Finsterer, J. & Stollberger, C. The heart in human dystrophinopathies. Cardiology 99, 1–19 (2003)
Rodriguez, E. K. et al. A method to reconstruct myocardial sarcomere lengths and orientations at transmural sites in beating canine hearts. Am. J. Physiol. 263, H293–H306 (1992)
Pasternak, C., Wong, S. & Elson, E. L. Mechanical function of dystrophin in muscle cells. J. Cell Biol. 128, 355–361 (1995)
Wu, G. et al. Lipid corralling and poloxamer squeeze-out in membranes. Phys. Rev. Lett. 93, 028101 (2004)
Lee, R. C., River, L. P., Pan, F. S., Ji, L. & Wollmann, R. L. Surfactant-induced sealing of electropermeabilized skeletal muscle membranes in vivo. Proc. Natl Acad. Sci. USA 89, 4524–4528 (1992)
Lee, R. C., Hannig, J., Matthews, K. L., Myerov, A. & Chen, C. T. Pharmaceutical therapies for sealing of permeabilized cell membranes in electrical injuries. Ann. NY Acad. Sci. 888, 266–273 (1999)
Turner, P. R., Fong, P. Y., Denetclaw, W. F. & Steinhardt, R. A. Increased calcium influx in dystrophic muscle. J. Cell Biol. 115, 1701–1712 (1991)
Togo, T., Alderton, J. M., Bi, G. Q. & Steinhardt, R. A. The mechanism of facilitated cell membrane resealing. J. Cell Sci. 112, 719–731 (1999)
Bansal, D. et al. Defective membrane repair in dysferlin-deficient muscular dystrophy. Nature 423, 168–172 (2003)
Danialou, G. et al. Dystrophin-deficient cardiomyocytes are abnormally vulnerable to mechanical stress-induced contractile failure and injury. FASEB J. 15, 1655–1657 (2001)
Davis, D. B., Delmonte, A. J., Ly, C. T. & McNally, E. M. Myoferlin, a candidate gene and potential modifier of muscular dystrophy. Hum. Mol. Genet. 9, 217–226 (2000)
Gregorevic, P. et al. Systemic delivery of genes to striated muscles using adeno-associated viral vectors. Nature Med. 10, 828–834 (2004)
Squire, S. et al. Prevention of pathology in mdx mice by expression of utrophin: analysis using an inducible transgenic expression system. Hum. Mol. Genet. 11, 3333–3344 (2002)
Torrente, Y. et al. Human circulating AC133+ stem cells restore dystrophin expression and ameliorate function in dystrophic skeletal muscle. J. Clin. Invest. 114, 182–195 (2004)
Goyenvalle, A. et al. Rescue of dystrophic muscle through U7 snRNA-mediated exon skipping. Science 306, 1796–1799 (2004)
Adams-Graves, P. et al. RheothRx (poloxamer 188) injection for the acute painful episode of sickle cell disease: a pilot study. Blood 90, 2041–2046 (1997)
Jewell, R. C., Khor, S. P., Kisor, D. F., LaCroix, K. A. & Wargin, W. A. Pharmacokinetics of RheothRx injection in healthy male volunteers. J. Pharm. Sci. 86, 808–812 (1997)
Straub, V. & Campbell, K. P. Muscular dystrophies and the dystrophin-glycoprotein complex. Curr. Opin. Neurol. 10, 168–175 (1997)
Coutu, P., Bennett, C. N., Favre, E. G., Day, S. M. & Metzger, J. M. Parvalbumin corrects slowed relaxation in adult cardiac myocytes expressing hypertrophic cardiomyopathy-linked α-tropomyosin mutations. Circ. Res. 94, 1235–1241 (2004)
Yasuda, S. I. et al. A novel method to study contraction characteristics of a single cardiac myocyte using carbon fibers. Am. J. Physiol. Heart Circ. Physiol. 281, H1442–H1446 (2001)
Grynkiewicz, G., Poenie, M. & Tsien, R. Y. A new generation of Ca2+ indicators with greatly improved fluorescence properties. J. Biol. Chem. 260, 3440–3450 (1985)
Michele, D. E., Gomez, C. A., Hong, K. E., Westfall, M. V. & Metzger, J. M. Cardiac dysfunction in hypertrophic cardiomyopathy mutant tropomyosin mice is transgene-dependent, hypertrophy-independent, and improved by β-blockade. Circ. Res. 91, 255–262 (2002)
Burger, A. J., Notarianni, M. P. & Aronson, D. Safety and efficacy of an accelerated dobutamine stress echocardiography protocol in the evaluation of coronary artery disease. Am. J. Cardiol. 86, 825–829 (2000)
Acknowledgements
This work was supported by a grant from the National Institute on Aging (J.M.M.). D.T. was supported by an NIH National Research Service Award, and S.M.D. was supported by an American Heart Association Fellow-to-Faculty Transition Award.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Reprints and permissions information is available at npg.nature.com/reprintsandpermissions. The authors declare no competing financial interests.
Supplementary information
Supplementary Figure S1
Schematic of experimental apparatus.
Supplementary Figure S2
Cardiac myocyte force decline upon single stretch during active contractions
Supplementary Figures Legends
Legends to accompany the above Supplementary Figures.
Supplementary Table S1
Summary of baseline haemodynamic function.
Supplementary Video S1
Single cardiac myocyte passive stretch video.
Supplementary Video S2
Mdx myocyte hypercontracture video.
Supplementary Video S3
Mdx myocyte fibrillation video.
Rights and permissions
About this article
Cite this article
Yasuda, S., Townsend, D., Michele, D. et al. Dystrophic heart failure blocked by membrane sealant poloxamer. Nature 436, 1025–1029 (2005). https://doi.org/10.1038/nature03844
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature03844
This article is cited by
-
Rapid restitution of contractile dysfunction by synthetic copolymers in dystrophin-deficient single live skeletal muscle fibers
Skeletal Muscle (2023)
-
Remote ischemic preconditioning prevents sarcolemmal-associated proteolysis by MMP-2 inhibition
Molecular and Cellular Biochemistry (2023)
-
The role of the dystrophin glycoprotein complex in muscle cell mechanotransduction
Communications Biology (2022)
-
Assessment of left ventricular dyssynchrony by speckle tracking echocardiography in children with duchenne muscular dystrophy
The International Journal of Cardiovascular Imaging (2022)
-
Effects of Intravenous Infusion of Vepoloxamer on Left Ventricular Function in Dogs with Advanced Heart Failure
Cardiovascular Drugs and Therapy (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.