Abstract
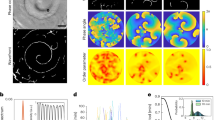
Nutrient-deprived Dictyostelium amoebae aggregate to form a multicellular structure by chemotaxis, moving towards propagating waves of cyclic AMP that are relayed from cell to cell. Organizing centres are not formed by founder cells, but are dynamic entities consisting of cores of outwardly rotating spiral waves1,2,3,4 that self-organize in a homogeneous cell population. Spiral waves are ubiquitously observed in chemical reactions as well as in biological systems5,6,7,8. Although feedback control of spiral waves in spatially extended chemical reactions has been demonstrated in recent years9,10, the mechanism by which control is achieved in living systems is unknown. Here we show that mutants of the cyclic AMP/protein kinase A pathway show periodic signalling, but fail to organize coherent long-range wave territories, owing to the appearance of numerous spiral cores. A theoretical model suggests that autoregulation of cell excitability mediated by protein kinase A acts to optimize the number of signalling centres.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Palsson, E. et al. Selection for spiral waves in the social amoebae Dictyostelium . Proc. Natl Acad. Sci. USA 94, 13719–13723 (1997)
Lauzeral, J., Halloy, J. & Goldbeter, A. Desynchronization of cells on the developmental path triggers the formation of spiral waves of cAMP during Dictyostelium aggregation. Proc. Natl Acad. Sci. USA 94, 9153–9158 (1997)
Foerster, P., Muller, S. & Hess, B. Curvature and spiral geometry in aggregation patterns of Dictyostelium discoideum . Development 109, 11–16 (1990)
Siegert, F. & Weijer, C. Digital image processing of optical density wave propagation in Dictyostelium discoideum and analysis of the effects of caffeine and ammonia. J. Cell Sci. 93, 325–335 (1989)
Lechleiter, J., Girard, S., Peralta, E. & Clapham, D. Spiral calcium wave propagation and annihilation in Xenopus laevis oocytes. Science 252, 123–126 (1991)
Gorelova, N. A. & Bures, J. Spiral waves of spreading depression in the isolated chicken retina. J. Neurobiol. 14, 353–363 (1983)
Davidenko, J. M., Pertsov, A. V., Salomonsz, R., Baxter, W. & Jalife, J. Stationary and drifting spiral waves of excitation in isolated cardiac muscle. Nature 355, 349–351 (1992)
Gray, R. A., Pertsov, A. M. & Jalife, J. Spatial and temporal organization during cardiac fibrillation. Nature 392, 75–78 (1998)
Kim, M. et al. Controlling chemical turbulence by global delayed feedback: pattern formation in catalytic CO oxidation on Pt(110). Science 292, 1357–1360 (2001)
Tung, C. K. & Chan, C. K. Dynamics of spiral waves under phase feedback control in a Belousov–Zhabotinsky reaction. Phys. Rev. Lett. 89, 248302 (2002)
Saran, S. et al. cAMP signaling in Dictyostelium—Complexity of cAMP synthesis, degradation and detection. J. Muscle Res. Cell Motil. 23, 793–802 (2002)
Iranfar, N., Fuller, D. & Loomis, W. F. Genome-wide expression analyses of gene regulation during early development of Dictyostelium discoideum . Euk. Cell 2, 664–670 (2003)
Gross, J. D., Peacey, M. J. & Trevan, D. J. Signal emission and signal propagation during early aggregation in Dictyostelium discoideum . J. Cell Sci. 22, 645–656 (1976)
Devreotes, P. N., Potel, M. J. & MacKay, S. A. Quantitative analysis of cyclic AMP waves mediating aggregation in Dictyostelium discoideum . Dev. Biol. 96, 405–415 (1983)
Torrence, C. & Compo, C. P. A practical guide to wavelet analysis. Bull. Am. Meteorol. Soc. 79, 61–78 (1998)
Thomason, P. A. et al. An intersection of the cAMP/PKA and two-component signal transduction systems in Dictyostelium . EMBO J. 17, 2838–2845 (1998)
Wessels, D. J. et al. The internal phosphodiesterase RegA is essential for the suppression of lateral pseudopods during Dictyostelium chemotaxis. Mol. Biol. Cell 11, 2803–2820 (2000)
Harwood, A. J. et al. Culmination in Dictyostelium is regulated by the cAMP-dependent protein kinase. Cell 69, 615–624 (1992)
Patel, H. et al. A temperature-sensitive adenylyl cyclase mutant of Dictyostelium . EMBO J. 19, 2247–2256 (2000)
Segall, J. E. et al. A MAP kinase necessary for receptor-mediated activation of adenylyl cyclase in Dictyostelium . J. Cell Biol. 128, 405–413 (1995)
Maeda, M. et al. Periodic signaling controlled by an oscillatory circuit that includes protein kinases ERK2 and PKA. Science 304, 875–878 (2004)
Sucgang, R., Weijer, C. J., Siegert, F., Franke, J. & Kessin, R. H. Null mutations of the Dictyostelium cyclic nucleotide phosphodiesterase gene block chemotactic cell movement in developing aggregates. Dev. Biol. 192, 181–192 (1997)
Laub, M. T. & Loomis, W. F. A molecular network that produces spontaneous oscillations in excitable cells of Dictyostelium . Mol. Biol. Cell 9, 3521–3532 (1998)
Martiel, J.-L. & Goldbeter, A. A model based on receptor desensitization for cyclic AMP signaling in Dictyostelium cells. Biophys. J. 52, 807–828 (1987)
Levine, H., Aranson, I., Tsimring, L. & Truong, T. V. Positive genetic feedback governs cAMP spiral wave formation in Dictyostelium . Proc. Natl Acad. Sci. USA 93, 6382–6386 (1996)
Lee, K. J., Goldstein, R. E. & Cox, E. C. Resetting wave forms in Dictyostelium territories. Phys. Rev. Lett. 87, 066101 (2001)
Fey, P., Compton, K. & Cox, E. C. Green fluorescent protein production in the cellular slime molds Polysphondylium pallidum and Dictyostelium discoideum . Gene 165, 127–130 (1995)
Simon, M. N., Pelegrini, O., Veron, M. & Kay, R. R. Mutation of protein kinase-A causes heterochronic development of Dictyostelium . Nature 356, 171–172 (1992)
Acknowledgements
The authors thank X. J. Guan and other members of the Cox laboratory for assistance, J. E. Segall for HS174 and HS174-A1, A. Kuspa for AK1045, H. Kuwayama for the pkaC-null strain, A. Harwood for the pka-Rm and Rc expression vectors, M. Veron for the anti-PKA-R monoclonal antibody, J. T. Bonner and M. Sano for discussions and K. McQuade, R. Segev and G. Weeks for detailed comments on our earlier manuscript. The authors also thank the Dictyostelium Stock Centre for providing HTY506. P.A.T. received a fellowship from the Wellcome Trust. This work was supported by a grant to E.C.C. from the NIH.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Supplementary information
Supplementary Notes
This file contains the legends for Supplementary Figures S4 and S5, Supplementary Discussion, Supplementary Equations, Supplementary Methods and additional references. (DOC 88 kb)
Supplementary Movie 1
This movie is a time-lapse recording of the optical density waves shown in Fig. 1b. Each panel is 29 mm 2 29 mm. The time label is added only to show the frame speed of the movie and does not represent the time elapsed after starvation. The movie clip is edited so that each panel starts 4-5 h after starvation. The movie of the erkB mutant starts approximately 10 h after starvation. (MPG 2130 kb)
Supplementary Movie 2
This movie shows the angular variable representation of the optical density waves and phase singularities (snapshots shown in Fig. 3a and Supplementary Fig. S4). Each panel is 12 mm 2 12 mm. The frame speed is the same as Supplementary Movie 1. (MPG 2423 kb)
Supplementary Movie 3
This movie shows the effect of starvation treatment on waveform and the simulations using initial conditions with higher excitability (Fig. 4 c-f). (MPG 5534 kb)
Supplementary Figure S4
A time-delay embedding technique reveals abnormal numbers of phase singularities in the mutants. (PDF 664 kb)
Supplementary Figure S5
Application of a mist of cAMP resets the phase of the optical density oscillations in the field of erkB mutants. (PDF 740 kb)
Rights and permissions
About this article
Cite this article
Sawai, S., Thomason, P. & Cox, E. An autoregulatory circuit for long-range self-organization in Dictyostelium cell populations. Nature 433, 323–326 (2005). https://doi.org/10.1038/nature03228
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature03228
This article is cited by
-
Exploring rare cellular activity in more than one million cells by a transscale scope
Scientific Reports (2021)
-
Topological turbulence in the membrane of a living cell
Nature Physics (2020)
-
Oscillatory cAMP cell-cell signalling persists during multicellular Dictyostelium development
Communications Biology (2019)
-
Spontaneous center formation in Dictyostelium discoideum
Scientific Reports (2019)
-
Connectivity and complex systems: learning from a multi-disciplinary perspective
Applied Network Science (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.