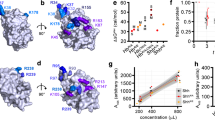
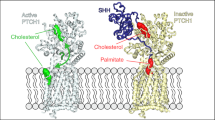
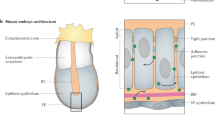
Abstract
Morphogens are ‘form-generating’ substances that spread from localized sites of production and specify distinct cellular outcomes at different concentrations. A cell's perception of morphogen concentration is thought to be determined by the number of active receptors, with inactive receptors making little if any contribution1. Patched (Ptc)2,3,4,5, the receptor for the morphogen Hedgehog (Hh)6,7,8,9,10,11,12, is active in the absence of ligand and blocks the expression of target genes by inhibiting Smoothened (Smo), an essential transducer of the Hh signal3,13,14,15,16. Hh binding to Ptc abrogates the ability of Ptc to inhibit Smo, thereby unleashing Smo activity and inducing target gene expression2,3,12,13,14,15,16. Here, we show that a cell's measure of ambient Hh concentration is not determined solely by the number of active (unliganded) Ptc molecules. Instead, we find that Hh-bound Ptc can titrate the inhibitory action of unbound Ptc. Furthermore, we demonstrate that this effect is sufficient to allow normal reading of the Hh gradient in the presence of a form of Ptc that cannot bind the ligand12 but retains its ability to inhibit Smo. These results support a model in which the ratio of bound to unbound Ptc molecules determines the cellular response to Hh.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Gurdon, J. B. & Bourillot, P. Y. Morphogen gradient interpretation. Nature 413, 797–803 (2001)
Ingham, P. W., Taylor, A. M. & Nakano, Y. Role of the Drosophila patched gene in positional signalling. Nature 353, 184–187 (1991)
Chen, Y. & Struhl, G. Dual roles for patched in sequestering and transducing Hedgehog. Cell 87, 553–563 (1996)
Stone, D. M. et al. The tumour-suppressor gene patched encodes a candidate receptor for Sonic hedgehog. Nature 384, 129–134 (1996)
Marigo, V., Davey, R. A., Zuo, Y., Cunningham, J. M. & Tabin, C. J. Biochemical evidence that patched is the Hedgehog receptor. Nature 384, 176–179 (1996)
Basler, K. & Struhl, G. Compartment boundaries and the control of Drosophila limb pattern by hedgehog protein. Nature 368, 208–214 (1994)
Tabata, T. & Kornberg, T. B. Hedgehog is a signaling protein with a key role in patterning Drosophila imaginal discs. Cell 76, 89–102 (1994)
Capdevila, J., Estrada, M. P., Sanchez-Herrero, E. & Guerrero, I. The Drosophila segment polarity gene patched interacts with decapentaplegic in wing development. EMBO J. 13, 71–82 (1994)
Struhl, G., Barbash, D. A. & Lawrence, P. A. Hedgehog organises the pattern and polarity of epidermal cells in the Drosophila abdomen. Development 124, 2143–2154 (1997)
Mullor, J. L., Calleja, M., Capdevila, J. & Guerrero, I. Hedgehog activity, independent of decapentaplegic, participates in wing disc patterning. Development 124, 1227–1237 (1997)
Strigini, M. & Cohen, S. M. A Hedgehog activity gradient contributes to AP axial patterning of the Drosophila wing. Development 124, 4697–4705 (1997)
Briscoe, J., Chen, Y., Jessell, T. M. & Struhl, G. A hedgehog-insensitive form of patched provides evidence for direct long-range morphogen activity of sonic hedgehog in the neural tube. Mol. Cell 7, 1279–1291 (2001)
Hooper, J. E. Distinct pathways for autocrine and paracrine Wingless signalling in Drosophila embryos. Nature 372, 461–464 (1994)
van den Heuvel, M. & Ingham, P. W. smoothened encodes a receptor-like serpentine protein required for hedgehog signalling. Nature 382, 547–551 (1996)
Alcedo, J., Ayzenzon, M., Von Ohlen, T., Noll, M. & Hooper, J. E. The Drosophila smoothened gene encodes a seven-pass membrane protein, a putative receptor for the hedgehog signal. Cell 86, 221–232 (1996)
Ingham, P. W. & McMahon, A. P. Hedgehog signaling in animal development: paradigms and principles. Genes Dev. 15, 3059–3087 (2001)
Taipale, J., Cooper, M. K., Maiti, T. & Beachy, P. A. Patched acts catalytically to suppress the activity of Smoothened. Nature 418, 892–897 (2002)
Lawrence, P. A. & Struhl, G. Morphogens, compartments, and pattern: lessons from Drosophila? Cell 85, 951–961 (1996)
Nakano, Y. et al. A protein with several possible membrane-spanning domains encoded by the Drosophila segment polarity gene patched. Nature 341, 508–513 (1989)
Hooper, J. E. & Scott, M. P. The Drosophila patched gene encodes a putative membrane protein required for segmental patterning. Cell 59, 751–765 (1989)
Vervoort, M., Crozatier, M., Valle, D. & Vincent, A. The COE transcription factor Collier is a mediator of short-range Hedgehog-induced patterning of the Drosophila wing. Curr. Biol. 9, 632–639 (1999)
Alcedo, J., Zou, Y. & Noll, M. Posttranscriptional regulation of smoothened is part of a self-correcting mechanism in the Hedgehog signaling system. Mol. Cell 6, 457–465 (2000)
Denef, N., Neubuser, D., Perez, L. & Cohen, S. M. Hedgehog induces opposite changes in turnover and subcellular localization of patched and smoothened. Cell 102, 521–531 (2000)
Ingham, P. W. et al. Patched represses the Hedgehog signalling pathway by promoting modification of the Smoothened protein. Curr. Biol. 10, 1315–1318 (2000)
Hepker, J., Wang, Q. T., Motzny, C. K., Holmgren, R. & Orenic, T. V. Drosophila cubitus interruptus forms a negative feedback loop with patched and regulates expression of Hedgehog target genes. Development 124, 549–558 (1997)
Aza-Blanc, P. & Kornberg, T. B. Ci: a complex transducer of the hedgehog signal. Trends Genet. 15, 458–462 (1999)
Methot, N. & Basler, K. Hedgehog controls limb development by regulating the activities of distinct transcriptional activator and repressor forms of Cubitus interruptus. Cell 96, 819–831 (1999)
Murakami, S., Nakashima, R., Yamashita, E. & Yamaguchi, A. Crystal structure of bacterial multidrug efflux transporter AcrB. Nature 419, 587–593 (2002)
Greenwood, S. & Struhl, G. Different levels of Ras activity can specify distinct transcriptional and morphological consequences in early Drosophila embryos. Development 124, 4879–4886 (1997)
Lee, T. & Luo, L. Mosaic analysis with a repressible cell marker for studies of gene function in neuronal morphogenesis. Neuron 22, 451–461 (1999)
Acknowledgements
We thank A. Adachi for generating transgenic flies; X.-J. Qiu for technical assistance; and R. Axel, E. Gouaux, I. Greenwald, T. Jessell, L. Johnston, R. Mann and A. Tomlinson for discussion and advice on the manuscript. A.C. is a Postdoctoral Associate and G.S. is an Investigator of the Howard Hughes Medical Institute.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Rights and permissions
About this article
Cite this article
Casali, A., Struhl, G. Reading the Hedgehog morphogen gradient by measuring the ratio of bound to unbound Patched protein. Nature 431, 76–80 (2004). https://doi.org/10.1038/nature02835
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature02835
This article is cited by
-
A systematic summary of survival and death signalling during the life of hair follicle stem cells
Stem Cell Research & Therapy (2021)
-
Hedgehog mediated degradation of Ihog adhesion proteins modulates cell segregation in Drosophila wing imaginal discs
Nature Communications (2017)
-
Capping Enzyme mRNA-cap/RNGTT Regulates Hedgehog Pathway Activity by Antagonizing Protein Kinase A
Scientific Reports (2017)
-
Smoothened regulation in response to Hedgehog stimulation
Frontiers in Biology (2015)
-
A model to explain specific cellular communications and cellular harmony:- a hypothesis of coupled cells and interactive coupling molecules
Theoretical Biology and Medical Modelling (2014)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.