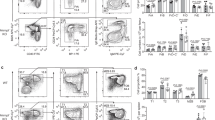
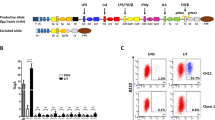
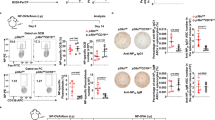
Abstract
Activated B cells differentiate to plasma cells to secrete IgM or, after undergoing class switch recombination (CSR), to secrete other classes of immunoglobulins1,2,3,4. Diversification of antibody function by CSR is important for humoral immunity. However, it remains unclear how the decision for the bifurcation is made. Bach2 is a B-cell-specific transcription repressor interacting with the small Maf proteins whose expression is high only before the plasma cell stage5,6,7. Here we show that Bach2 is critical for CSR and somatic hypermutation (SHM)2,4,8 of immunoglobulin genes. Genetic ablation of Bach2 in mice revealed that Bach2 was required for both T-cell-independent and T-cell-dependent IgG responses and SHM. When stimulated in vitro, Bach2-deficient B cells produced IgM, as did wild-type cells, and abundantly expressed Blimp-1 (refs 9, 10) and XBP-1 (ref. 11), critical regulators of the plasmacytic differentiation12, indicating that Bach2 was not required for the plasmacytic differentiation itself. However, they failed to undergo efficient CSR. These findings define Bach2 as a key regulator of antibody response and provide an insight into the orchestration of CSR and SHM during plasma cell differentiation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Snapper, C., Marcu, K. & Zelazowski, P. The immunoglobulin class switch: beyond ‘accessibility’. Immunity 6, 217–223 (1997)
Kinoshita, K. & Honjo, T. Linking class-switch recombination with somatic hypermutation. Nature Rev. Mol. Cell Biol. 2, 493–503 (2001)
Manis, J., Tian, M. & Alt, F. Mechanism and control of class-switch recombination. Trends Immunol. 23, 31–39 (2002)
Honjo, T., Kinoshita, K. & Muramatsu, M. Molecular mechanism of class switch recombination: linkage with somatic hypermutation. Annu. Rev. Immunol. 20, 165–196 (2002)
Igarashi, K. et al. Regulation of transcription by dimerization of erythroid factor NF-E2 p45 with small Maf proteins. Nature 367, 568–572 (1994)
Oyake, T. et al. Bach proteins belong to a novel family of BTB-basic leucine zipper transcription factors that interact with MafK and regulate transcription through the NF-E2 site. Mol. Cell. Biol. 16, 6083–6095 (1996)
Muto, A. et al. Identification of Bach2 as a B-cell-specific partner for small maf proteins that negatively regulate the immunoglobulin heavy chain gene 3′ enhancer. EMBO J. 17, 5734–5743 (1998)
Papavasiliou, F. & Schatz, D. Somatic hypermutation of immunoglobulin genes: merging mechanisms for genetic diversity. Cell 109, S35–S44 (2002)
Shaffer, A. et al. Blimp-1 orchestrates plasma cell differentiation by extinguishing the mature B cell gene expression program. Immunity 17, 51–62 (2002)
Shapiro-Shelef, M. et al. Blimp-1 is required for the formation of immunoglobulin secreting plasma cells and pre-plasma memory B cells. Immunity 19, 607–620 (2003)
Reimold, A. et al. Plasma cell differentiation requires the transcription factor XBP-1. Nature 412, 300–307 (2001)
Calame, K., Lin, K. & Tunyaplin, C. Regulatory mechanisms that determine the development and function of plasma cells. Annu. Rev. Immunol. 21, 205–230 (2003)
Durandy, A. & Honjo, T. Human genetic defects in class-switch recombination (hyper-IgM syndromes). Curr. Opin. Immunol. 13, 543–548 (2001)
Allen, D., Simon, T., Sablitzky, F., Rajewsky, K. & Cumano, A. Antibody engineering for the analysis of affinity maturation of an anti-hapten response. EMBO J. 7, 1995–2001 (1989)
Jacob, J., Kelsoe, G., Rajewsky, K. & Weiss, U. Intraclonal generation of antibody mutants in germinal centres. Nature 354, 389–392 (1991)
Hasbold, J., Lyons, A., Kehry, M. & Hodgkin, P. Cell division number regulates IgG1 and IgE switching of B cells following stimulation by CD40 ligand and IL-4. Eur. J. Immunol. 28, 1040–1051 (1998)
Muramatsu, M. et al. Class switch recombination and hypermutation require activation-induced cytidine deaminase (AID), a potential RNA editing enzyme. Cell 102, 553–563 (2000)
Mittrucker, H. et al. Requirement for the transcription factor LSIRF/IRF4 for mature B and T lymphocyte function. Science 275, 540–543 (1997)
Ye, B. et al. The BCL-6 proto-oncogene controls germinal-centre formation and Th2-type inflammation. Nature Genet. 16, 161–170 (1997)
Schubart, D., Rolink, A., Kosco-Vilbois, M., Botteri, F. & Matthias, P. B-cell-specific coactivator OBF-1/OCA-B/Bob1 required for immune response and germinal centre formation. Nature 383, 538–542 (1996)
Kim, U. et al. The B-cell-specific transcription coactivator OCA-B/OBF-1/Bob-1 is essential for normal production of immunoglobulin isotypes. Nature 383, 542–547 (1996)
Barberis, A., Widenhorn, K., Vitelli, L. & Busslinger, M. A novel B-cell lineage-specific transcription factor present at early but not late stages of differentiation. Genes Dev. 4, 849–859 (1990)
Lin, Y., Wong, K. & Calame, K. Repression of c-myc transcription by Blimp-1, an inducer of terminal B cell differentiation. Science 276, 596–599 (1997)
Monroe, J. Up-regulation of c-fos expression is a component of the mIg signal transduction mechanism but is not indicative of competence for proliferation. J. Immunol. 140, 1454–1460 (1988)
Sun, J. et al. Heme regulates the dynamic exchange of Bach1 and NF-E2-related factors in the Maf transcription factor network. Proc. Natl Acad. Sci. USA 101, 1461–1466 (2004)
Brand, M. et al. Dynamic changes in transcription factor complexes during erythroid differentiation revealed by quantitative proteomics. Nature Struct. Mol. Biol. 11, 73–80 (2004)
Muto, A. et al. Activation of Maf/AP-1 repressor Bach2 by oxidative stress promotes apoptosis and its interaction with promyelocytic leukemia nuclear bodies. J. Biol. Chem. 277, 20724–20733 (2002)
Sun, J. et al. The promoter of mouse transcription repressor bach1 is regulated by Sp1 and trans-activated by Bach1. J. Biochem. (Tokyo) 130, 385–392 (2001)
Acknowledgements
We thank M. Kobayashi, D. Engel, M. Muramatsu and T. Honjo for discussion and comments on the manuscript, M. Busslinger for information on RT–PCR analysis, N. Kaneko for the preparation of tissue sections, and M. Kanno for the use of FACS machinery. This work was supported by Grants-in-aid from the Ministry of Education, Culture, Sport, Science and Technology of Japan and Hiroshima University 21st century COE programme. A.M. was initially supported by the Research Fellowships for Young Scientists from the Japanese Society for Promotion of Science.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Supplementary information
Supplementary Figure 1
a, Targeting strategy of mouse bach2 gene. b, Southern blot analysis of DNA prepared from the control and targeted embryonic stem (ES) cell clones. c, Western blot analysis of bone marrow B cell extracts of each genotype with anti-Bach2.
Supplementary Figure 2a
Nucleotide sequences of Vh186.2 mRNAs from MLN of bach2+/+ mice 28 days after primary immunization with NP-CGG.
Supplementary Figure 2b
Nucleotide sequences of Vh186.2 mRNAs from MLN of bach2-/- mice 28 days after primary immunization with NP-CGG.
Supplementary Figure 2c
Nucleotide sequence of Vh186.2, DH and JH joints amplified from the mRNA of MLN cells of the NP-CGG immunized bach2+/+ and bach2-/- mice.
Supplementary Figure 3
Purified spleen B cells from wild-type and bach2-/- mice were cultured with LPS for 48 hrs and BrdU for last 6 hrs. BrdU incorporations were measured.
Rights and permissions
About this article
Cite this article
Muto, A., Tashiro, S., Nakajima, O. et al. The transcriptional programme of antibody class switching involves the repressor Bach2. Nature 429, 566–571 (2004). https://doi.org/10.1038/nature02596
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature02596
This article is cited by
-
BACH2-mediated CD28 and CD40LG axes contribute to pathogenesis and progression of T-cell lymphoblastic leukemia
Cell Death & Disease (2024)
-
The genetics of Graves’ disease
Reviews in Endocrine and Metabolic Disorders (2024)
-
An early-onset SLE patient with a novel paternal inherited BACH2 mutation
Journal of Clinical Immunology (2023)
-
Up-regulated circBACH2 contributes to cell proliferation, invasion, and migration of triple-negative breast cancer
Cell Death & Disease (2021)
-
Attenuation of TCR-induced transcription by Bach2 controls regulatory T cell differentiation and homeostasis
Nature Communications (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.