Abstract
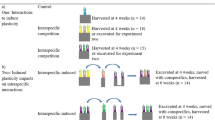
Explaining mutualistic cooperation between species remains one of the greatest problems for evolutionary biology1,2,3,4. Why do symbionts provide costly services to a host, indirectly benefiting competitors sharing the same individual host? Host monitoring of symbiont performance and the imposition of sanctions on ‘cheats’ could stabilize mutualism5,6. Here we show that soybeans penalize rhizobia that fail to fix N2 inside their root nodules. We prevented a normally mutualistic rhizobium strain from cooperating (fixing N2) by replacing air with an N2-free atmosphere (Ar:O2). A series of experiments at three spatial scales (whole plants, half root systems and individual nodules) demonstrated that forcing non-cooperation (analogous to cheating) decreased the reproductive success of rhizobia by about 50%. Non-invasive monitoring implicated decreased O2 supply as a possible mechanism for sanctions against cheating rhizobia. More generally, such sanctions by one or both partners may be important in stabilizing a wide range of mutualistic symbioses.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Herre, E. A., Knowlton, N., Mueller, U. G. & Rehner, S. A. The evolution of mutualisms: exploring the paths between conflict and cooperation. Trends Ecol. Evol. 14, 49–53 (1999)
Frank, S. A. Foundations of Social Evolution (Princeton Univ. Press, Princeton, 1998)
Yu, D. W. Parasites of mutualisms. Biol. J. Linn. Soc. 72, 529–546 (2001)
Axelrod, R. & Hamilton, W. D. The evolution of cooperation. Science 211, 1390–1396 (1981)
Denison, R. F. Legume sanctions and the evolution of symbiotic cooperation by rhizobia. Am. Nat. 156, 567–576 (2000)
West, S. A., Kiers, E. T., Simms, E. L. & Denison, R. F. Sanctions and mutualism stability: why do rhizobia fix nitrogen? Proc. R. Soc. Lond. B 269, 685–694 (2002)
Crespi, B. J. The evolution of social behavior in microorganisms. Trends Ecol. Evol. 16, 178–183 (2001)
West, S. A., Kiers, E. T., Pen, I. & Denison, R. F. Sanctions and mutualism stability: when should less beneficial mutualists be tolerated? J. Evol. Biol. 15, 830–837 (2002)
Pellmyr, O. & Huth, C. J. Evolutionary stability of mutualism between yuccas and yucca moths. Nature 372, 257–260 (1994)
Frank, S. A. Mutual policing and repression of competition in the evolution of cooperative groups. Nature 377, 520–522 (1995)
Ratnieks, F. L. W., Monnin, T. & Foster, K. R. Inclusive fitness theory: novel predictions and tests in social Hymenoptera. Ann. Zool. Fennici 38, 201–214 (2001)
Burdon, J. J., Gibson, A. H., Searle, S. D., Woods, M. J. & Brockwell, J. Variation in the effectiveness of symbiotic associations between native rhizobia and temperate Australian Acacia: within-species interactions. J. Appl. Ecol. 36, 398–408 (1999)
Singleton, P. W. & Stockinger, K. R. Compensation against ineffective nodulation in soybean. Crop Sci. 23, 69–72 (1983)
Ferriere, R., Bronstein, J. L., Rinaldi, S., Law, R. & Gauduchon, M. Cheating and the evolutionary stability of mutualisms. Proc. R. Soc. Lond. B 269, 773–780 (2001)
Amarger, N. Competition for nodule formation between effective and ineffective strains of Rhizobium meliloti. Soil Biol. Biochem. 13, 475–480 (1981)
Hahn, M. & Studer, D. Competitiveness of a nif- Bradyrhizobium japonicum mutant against the wild-type strain. FEMS Microbiol. Lett. 33, 143–148 (1986)
Rasche, M. E. & Arp, D. J. Hydrogen inhibition of nitrogen reduction by nitrogenase in isolated soybean nodule bacteroids. Plant Physiol. 91, 663–668 (1989)
Singleton, P. W. & van Kessel, C. Effect of localized nitrogen availability to soybean half-root systems on photosynthate partitioning to roots and nodules. Plant Physiol. 83, 552–556 (1987)
Udvardi, M. K. & Kahn, M. L. Evolution of the (Brady)Rhizobium–legume symbiosis: why do bacteroids fix nitrogen? Symbiosis 14, 87–101 (1993)
King, B. J. & Layzell, D. B. Effect of increases in oxygen concentration during the argon-induced decline in nitrogenase activity in root nodules of soybean. Plant Physiol. 96, 376–381 (1991)
Sheehy, J. E., Minchin, F. R. & Witty, J. F. Biological control of the resistance to oxygen flux in nodules. Ann. Bot. 52, 565–571 (1983)
Hartwig, U., Boller, B. & Nösberger, J. Oxygen supply limits nitrogenase activity of clover nodules after defoliation. Ann. Bot. 59, 285–291 (1987)
Denison, R. F. & Harter, B. L. Nitrate effects on nodule oxygen permeability and leghemoglobin. Nodule oximetry and computer modeling. Plant Physiol. 107, 1355–1364 (1995)
Layzell, D. B., Rainbird, R. M., Atkins, C. A. & Pate, J. S. Economy of photosynthetic use in nitrogen-fixing legume nodules. Plant Physiol. 64, 888–891 (1979)
Sen, D. & Weaver, R. W. Nitrogen fixing activity of rhizobial strain 32H1 in peanut and cowpea nodules. Plant Sci. Lett. 18, 315–318 (1980)
Lodwig, E. M. et al. Amino-acid cycling drives nitrogen fixation in the legume–Rhizobium symbiosis. Nature 422, 722–726 (2003)
Kijne, J. W. The fine structure of pea root nodules. 2. Senescence and disintegration of the bacteroid tissue. Physiol. Plant Pathol. 7, 17–21 (1975)
Sprent, J. I. & Raven, J. A. Evolution of nitrogen-fixing symbioses. Proc. R. Soc. Edinb. B 85, 215–237 (1985)
Layzell, D. B., Hunt, S., King, B. J., Walsh, K. B. & Weagle, G. E. in Applications of Continuous and Steady-State Methods to Root Biology (eds Torrey, J. G. & Winship, L. J.) 1–28 (Kluwer Academic, Dordrecht, 1989)
Denison, R. F. & Layzell, D. B. Measurement of legume nodule respiration and O2 permeability by noninvasive spectrophotometry of leghemoglobin. Plant Physiol. 96, 137–143 (1991)
Acknowledgements
We thank R. Grosberg, S. Nee, A. Griffin, D. Shuker and M. Stanton for comments, P. Graham, D. Phillips and M. King for advice on methods, R. Nelson and D. Smith for soybean cultivars ‘T243’ and ‘S0066’, and P. van Berkum for B. japonicum 110ARS. This work was supported by the NSF (grant to R.F.D. and graduate fellowship to E.T.K.), the California Agricultural Experiment Station, the Land Institute, the Royal Society, the BBSRC and the NERC.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Rights and permissions
About this article
Cite this article
Kiers, E., Rousseau, R., West, S. et al. Host sanctions and the legume–rhizobium mutualism. Nature 425, 78–81 (2003). https://doi.org/10.1038/nature01931
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature01931
This article is cited by
-
Evidence that the domesticated fungus Leucoagaricus gongylophorus recycles its cytoplasmic contents as nutritional rewards to feed its leafcutter ant farmers
IMA Fungus (2023)
-
Lotus japonicus regulates root nodulation and nitrogen fixation dependent on the molecular form of nitrogen fertilizer
Plant and Soil (2023)
-
Eco-evolutionary Logic of Mutualisms
Dynamic Games and Applications (2023)
-
On Holobionts, Holospecies, and Holoniches: the Role of Microbial Symbioses in Ecology and Evolution
Microbial Ecology (2023)
-
Impacts of domestication on the rhizobial mutualism of five legumes across a gradient of nitrogen-fertilisation
Plant and Soil (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.