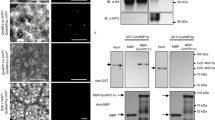
Abstract
Symbiotic root nodules are beneficial to leguminous host plants; however, excessive nodulation damages the host because it interferes with the distribution of nutrients in the plant. To keep a steady balance, the nodulation programme is regulated systemically in leguminous hosts1,2. Leguminous mutants that have lost this ability display a hypernodulating phenotype. Through the use of reciprocal and self-grafting studies using Lotus japonicus hypernodulating mutants, har1 (also known as sym78)3, we show that the shoot genotype is responsible for the negative regulation of nodule development. A map-based cloning strategy revealed that HAR1 encodes a protein with a relative molecular mass of 108,000, which contains 21 leucine-rich repeats, a single transmembrane domain and serine/threonine kinase domains. The har1 mutant phenotype was rescued by transfection of the HAR1 gene. In a comparison of Arabidopsis receptor-like kinases, HAR1 showed the highest level of similarity with CLAVATA1 (CLV1)4. CLV1 negatively regulates formation of the shoot and floral meristems through cell–cell communication involving the CLV3 peptide5. Identification of hypernodulation genes thus indicates that genes in leguminous plants bearing a close resemblance to CLV1 regulate nodule development systemically, by means of organ–organ communication.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Kosslak, R. M. & Bohlool, B. B. Suppression of nodule development of one side of a split-root system of soybeans caused by prior inoculation of the other side. Plant Physiol. 75, 125–130 (1984)
Gresshoff, P. M. & Caetano-Anollés, G. Current Topics in Plant Molecular Biology: Plant Biotechnology and Development (ed. Gresshoff, P. M.) 87–99 (CRC, Boca Raton, 1992)
Kawaguchi, M. et al. Root, root hair, and symbiotic mutants of the model legume Lotus japonicus. Mol. Plant-Microbe Interact. 15, 17–26 (2002)
Clark, S. E., Williams, R. W. & Meyerowitz, E. M. The CLAVATA1 gene encodes a putative receptor kinase that controls shoot and floral meristem size in Arabidopsis. Cell 89, 575–585 (1997)
DeYoung, B. J. & Clark, S. E. Signaling through the CLAVATA1 receptor complex. Plant Mol. Biol. 46, 505–513 (2001)
Penmetsa, R. V. & Cook, D. R. A legume ethylene-insensitive mutant hyperinfected by its rhizobial symbiont. Science 275, 527–530 (1997)
Nutman, P. S. Studies on the physiology of nodule formation. III. Experiments on the excision of root-tips and nodules. Ann. Bot. 16, 79–101 (1952)
Pierce, M. & Bauer, W. D. A rapid regulatory response governing nodulation in soybean. Plant Physiol. 73, 286–290 (1983)
van Brussel, A. A., Tak, T., Boot, K. J. & Kijne, J. W. Autoregulation of root nodule formation: signals of both symbiotic partners studied in a split-root system of Vicia sativa subsp. nigra. Mol. Plant-Microbe Interact. 15, 341–349 (2002)
Nishimura, R., Ohmori, M. & Kawaguchi, M. The novel symbiotic phenotype of enhanced-nodulating mutant of Lotus japonicus: astray is an early nodulating mutant with wider nodulation zone. Plant Cell Physiol. 43, 853–859 (2002)
Shauser, L. et al. Symbiotic mutants deficient in nodule establishment identified after T-DNA transformation of Lotus japonicus. Mol. Gen. Genet. 239, 414–423 (1998)
Szczyglowski, K. et al. Nodule organogenesis and symbiotic mutants of the model legume Lotus japonicus. Mol. Plant-Microbe Interact. 11, 684–697 (1998)
Wopereis, J. et al. Short root mutant of Lotus japonicus with a dramatically altered symbiotic phenotype. Plant J. 23, 97–114 (2000)
Solaiman, M. Z. et al. Characterization of mycorrhizas formed by Glomus sp. on root of hypernodulating mutants of Lotus japonicus. J. Plant Res. 113, 443–448 (2000)
Delves, A. C. et al. Regulation of the soybean-Rhizobium nodule symbiosis by shoot and root factors. Plant Physiol. 82, 588–590 (1986)
Hamaguchi, H., Kokubun, M. & Akao, S. Shoot control of nodulation is modified by the root in the supernodulating soybean mutant En6500 and its wild-type parent cultivar Enrei. Soil. Sci. Plant Nutr. 38, 771–774 (1992)
Sheng, C. & Harper, J. Shoot versus root signal involvement in nodulation and vegetative growth in wild-type and hypernodulating soybean genotypes. Plant Physiol. 113, 825–831 (1997)
Sagan, M. & Duc, G. Sym28 and Sym29, two new genes involved in autoregulation of nodulation in pea (Pisum sativum L.). Symbiosis 20, 229–245 (1996)
Olsson, J. E., Nakao, P., Bohlool, B. B. & Gresshoff, P. M. Lack of systemic suppression of nodulation in split root systems of supernodulating soybean (Glycine max [L.] Merr.) mutants. Plant Physiol. 90, 1347–1352 (1989)
Yamanaka, N. et al. An information linkage map of soybean reveals QTLs for flowering time, leaflet morphology and regions of segregation distortion. DNA Res. 8, 61–72 (2001)
Yamamoto, E., Karakaya, H. C. & Knap, H. T. Molecular characterization of two soybean homologues of Arabidopsis thaliana CLAVATA1 from the wild type and fasciation mutant. Biochem. Biophys. Acta. 1491, 333–340 (2000)
Akao, S. & Kouchi, H. A supernodulating mutant isolated from soybean cultivar Enrei. Soil Sci. Plant Nutr. 38, 183–187 (1992)
Matsubayashi, Y., Ogawa, M., Morita, A. & Sakagami, Y. An LRR receptor kinases involved in perception of a peptide plant hormone, phytosulfokine. Science 296, 1470–1472 (2002)
Scheer, J. M. & Ryan, C. A. The systemin receptor SR160 from Lycopersicon peruvianum is a member of the LRR receptor kinase family. Proc. Natl Acad. Sci. USA 99, 9585–9590 (2002)
Flemetakis, E. et al. Lotus japonicus contains two distinct ENOD40 genes that are expressed in symbiotic, nonsymbiotic, and embryonic tissues. Mol. Plant-Microbe Interact. 13, 987–994 (2000)
Kouchi, H., Takane, K., So, R. B., Ladha, J. K. & Reddy, P. M. Rice ENOD40: isolation and expression analysis in rice and transgenic soybean root nodules. Plant J. 18, 121–129 (1999)
Kawaguchi, M. et al. Providing the basis for genomics in Lotus japonicus: the accessions Miyakojima and Gifu are appropriate crossing partners for genetic analyses. Mol. Gen. Genomics 266, 157–166 (2001)
Hayashi, M. et al. Construction of a genetic linkage map of the model legume Lotus japonicus using an intraspecific F2 population. DNA Res. 8, 301–310 (2001)
Kawasaki, S. & Murakami, Y. Genome analysis of Lotus japonicus. J. Plant Res. 113, 497–506 (2000)
Stiller, J. et al. High frequency transformation and regeneration of transgenic plants in model legume Lotus japonicus. J. Exp. Bot. 48, 1357–1365 (1997)
Acknowledgements
We thank A. A. T. van den Brussel, J. Harper, P. M. Gresshoff and T. Ohyama for comments, and S. Sato, Y. Nakamura and S. Tabata for providing draft sequence data on TAC clones and for gene modelling. We also thank E. Kobayashi for horticultural services. This research was supported in part by Grants-in-Aid for Scientific Research on Priority Areas and Special Coordination Funds for Promoting Science and Technology to M.K. from the Ministry of Education, Culture, Sports, Science and Technology of the Japanese Government. This work was also partly supported by a grant from the programme for Promotion of Basic Research Activities for Innovative Biosciences to K.H.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Rights and permissions
About this article
Cite this article
Nishimura, R., Hayashi, M., Wu, GJ. et al. HAR1 mediates systemic regulation of symbiotic organ development. Nature 420, 426–429 (2002). https://doi.org/10.1038/nature01231
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature01231
This article is cited by
-
The developmental dynamics in cool season legumes with focus on chickpea
Plant Molecular Biology (2023)
-
Lotus japonicus regulates root nodulation and nitrogen fixation dependent on the molecular form of nitrogen fertilizer
Plant and Soil (2023)
-
Balancing nitrate acquisition strategies in symbiotic legumes
Planta (2023)
-
The B-type response regulator GmRR11d mediates systemic inhibition of symbiotic nodulation
Nature Communications (2022)
-
Lotus japonicus HAR1 regulates root morphology locally and systemically under a moderate nitrate condition in the absence of rhizobia
Planta (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.