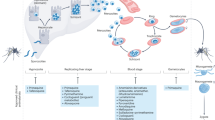
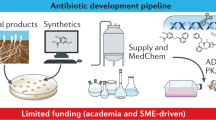
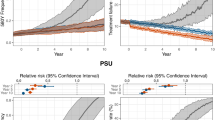
Abstract
Continued and sustainable improvements in antimalarial medicines through focused research and development are essential for the world's future ability to treat and control malaria. Unfortunately, malaria is a disease of poverty, and despite a wealth of scientific knowledge there is insufficient market incentive to generate the competitive, business-driven industrial antimalarial drug research and development that is normally needed to deliver new products. Mechanisms of partnering with industry have been established to overcome this obstacle and to open up and build on scientific opportunities for improved chemotherapy in the future.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Calabrese, L. & Fleischer, A. Thalidomide: current and potential clinical applications. Am. J. Med. 108, 487–495 (2000).
Wellems, T. & Plowe, C. Chloroquine-resistant malaria. J. Infect. Dis. 184, 770–776 (2001).
Sirawaraporn, W. Dihydrofolate reductase and antifolate resistance in malaria. Drug Resist. Update 1, 397–406 (1998).
Cowman, A. in Malaria Parasite Biology, Pathogenesis and Protection (ed. Sherman, I.) 317–330 (ASM, Washington, 1999).
Report No. WHO/CDS/RBM/2001.33 (World Health Organization, Geneva, 2001).
Report No. WHO/CDS/RBM/2001.35 (World Health Organization, Geneva, 2001).
Report No. WHO/MAL/98.1086 (World Health Organization, Geneva, 1998).
Rosenthal, P. (ed.) Antimalarial Chemotherapy. Mechanisms of Action, Resistance, and New Directions in Drug Discovery (Humana, Totowa, New Jersey, 2001).
Meshnick, S. & Dobson, M. in Antimalarial Chemotherapy. Mechanisms of Action, Resistance, and New Directions in Drug Discovery (ed. Rosenthal, P.) 15–26 (Humana, Totowa, New Jersey, 2001).
Ridley, R. G. & Hudson, A. T. Quinoline anti-malarials. Exp. Opin. Ther. Patents 8, 121–136 (1998).
O'Neill, P., Bray, P., Hawley, S., Ward, S. & Park, B. 4-Aminoquinolines—past, present, and future: a chemical perspective. Pharmacol. Ther. 77, 29–58 (1998).
Tilley, L., Loria, P. & Foley, M. in Antimalarial Chemotherapy. Mechanisms of Action, Resistance, and New Directions in Drug Discovery (ed. Rosenthal, P.) 87–122 (Humana, Totowa, New Jersey, 2001).
Dorsey, G., Fidock, D., Wellems, T. & Rosenthal, P. in Antimalarial Chemotherapy. Mechanisms of Action, Resistance and New Directions in Drug Discovery (ed. Rosenthal, P.) 153–172 (Humana, Totowa, New Jersey, 2001).
Li, Y. & Wu, Y.-L. How chinese scientists discovered qinghaosu (artemisinin) and developed its derivatives. What are the future perspectives? Med. Trop. 58, 9–12 (1998).
Price, R. Artemisinin drugs: novel antimalarial agents. Exp. Opin. Invest. Drugs 9, 1815–1827 (2000).
Haynes, R. Artemisinin and derivatives: the future for malaria treatment? Curr. Opin. Infect. Dis. 14, 719–726 (2001).
Meshnick, S. in Antimalarial Chemotherapy. Mechanisms of Action, Resistance, and New Directions in Drug Discovery (ed. Rosenthal, P.) 191–202 (Humana, Totowa, New Jersey, 2001).
White, N. Assessment of the pharmacodynamic properties of antimalarial drugs in vivo. Antimicrob. Agents Chemoth. 41, 1413–1422 (1997).
White, N. Delaying antimalarial drug resistance with combination chemotherapy. Parassitologia 41, 301–308 (1999).
Brockman, A. et al. Plasmodium falciparum antimalarial drug susceptibility on the north-western border of Thailand during five years of extensive use of artesunate-mefloquine. Trans. R. Soc. Trop. Med. Hyg. 94, 537–544 (2000).
Bloland, P. B., Ettling, M. & Meek, S. Combination therapy for malaria in Africa: hype or hope? Bull. World Health Organ. 78, 1378–88 (2000).
Petras, J. et al. Arteether-induced brain injury in Macaca mulatta. I. The precerebellar nuclei: the lateral reticular nuclei, paramedian reticular nuclei, and perihypoglossal nuclei. Anat. Embryol. (Berl.) 201, 383–397 (2000).
Dayan, A. Neurotoxicity and artemisinin compounds. Do the observations in animals justify limitation of clinical use? Med. Trop. 58, 32–37 (1998).
Kissinger, E. et al. Clinical and neurophysiological study of the effects of multiple doses of artemisinin on brain-stem function in Vietnamese patients. Am. J. Trop. Med. Hyg. 63, 2000 (2000).
Leonardi, E., Gilvary, G., White, N. & Nosten, F. Severe allergic reactions to oral artesunate: a report of two cases. Trans. R. Soc. Trop. Med. Hyg. 95, 182–183 (2001).
Skelton-Stroud, P. & Mull, R. Positioning, labelling, and medical information control of co-artemether tablets (CPG 56697): a fixed novel combination of artemether and benflumetol. Med. Trop. 58, 77–81 (1998).
Noedl, H., Allmendinger, T., Prajakwong, S., Wernsdorfer, G. & Wernsdorfer, W. Desbutyl-benflumetol, a novel antimalarial compound: in vitro activity in fresh isolates of Plasmodium falciparum from Thailand. Antimicrob. Agents Chemother. 45, 2106–2109 (2001).
Basco, L., Bickii, J. & Ringwald, P. In vitro activity of lumefantrine (benflumetol) against clinical isolates of Plasmodium falciparum in Yaounde, Cameroon. Antimicrob. Agents Chemother. 42, 2347–2351 (1998).
Ezzet, F., Vugt, M. v., Nosten, F., Looareesuwan, S. & White, N. Pharmacokinetics and pharmacodynamics of lumefantrine (benflumetol) in acute falciparum malaria. Antimicrob. Agents Chemother. 44, 697–704 (2000).
Sherman, I. in Malaria—Parasite Biology, Pathogenesis and Protection (ed. Sherman, I.) 177–184 (ASM, Washington DC, 1998).
Wolfe, E. et al. Cost-effectiveness of sulfadoxine-pyrimethamine for the prevention of malaria-associated low birth weight. Am. J. Trop. Med. Hyg. 64, 178–186 (2001).
Takechi, M. et al. Therapeutic efficacy of sulphadoxine/pyrimethamine and susceptibility in vitro of P. falciparum isolates to sulphadoxine-pyremethamine and other antimalarial drugs in Malawian children. Trop. Med. Int. Health 6, 429–434 (2001).
White, N. Drug resistance in malaria. Br. Med. Bull. 54, 703–715 (1998).
Looareesuwan, S., Chulay, J., Canfield, C. & Hutchinson, D. Malarone (atovaquone and proguanil hydrochloride): a review of its clinical development for treatment of malaria. Malarone Clinical Trials Study Group. Am. J. Trop. Med. Hyg. 60, 533–541 (1999).
Vaidya, A. in Antimalarial Chemotherapy. Mechanisms of Action, Resistance, and New Directions in Drug Discovery (ed. Rosenthal, P.) 203–218 (Humana, Totowa, New Jersey, 2001).
Fichera, M. & Roos, D. A plastid organelle as a drug target in apicomplexan parasites. Nature 390, 407–409 (1997).
Clough, B. & Wilson, R. in Antimalarial Chemotherapy. Mechanisms of Action, Resistance, and New Directions in Drug Discovery (ed. Rosenthal, P.) 265–286 (Humana, Totowa, New Jersey, 2001).
Macreadie, I., Ginsburg, H., Sirawaraporn, W. & Tilley, L. Antimalarial drug development and new targets. Parasitol. Today 16, 438–444 (2000).
Padmanaban, G. & Rangarajan, P. Emerging targets for antimalarial drugs. Expert Opin. Ther. Targets 5, 423–441 (2001).
Bannister, L. A brief illustrated guide to the ultrastructure of Plasmodium falciparum asexual blood stages. Parasitol. Today 16, 427–433 (2000).
Francis, S., Sullivan, D. J. & Goldberg, D. Hemoglobin metabolism in the malaria parasite Plasmodium falciparum. Annu. Rev. Microbiol. 51, 97–123 (1997).
Coombs, G. et al. Aspartic proteases of Plasmodium falciparum and other parasitic protozoa as drug targets. Trends Parasitol. 17, 532–537 (2001).
Shenai, B., Sijwali, P., Singh, A. & Rosenthal, P. Characterization of native and recombinant falcipain-2, a principal trophozoite cysteine protease and essential hemoglobinase of Plasmodium falciparum. J. Biol. Chem. 275, 29000–29010 (2000).
Eggleson, K., Duttin, K. & Goldberg, D. Identification and characterisation of Falcilysin, a metallopeptidase involved in hemoglobin catabolism within the malarial parasite Plasmodium falciparum. J. Biol. Chem. 274, 32411–32417 (1999).
Pagola, S., Stephens, P., Bohle, D., Kosar, A. & Madsen, S. The structure of malaria pigment β-haematin. Nature 16, 307–310 (2000).
Chou, A., Cherli, R. & Fitch, C. Ferriprotoporphyrin IX fulfills the criteria for identification as the chloroquine receptor of malaria parasites. Biochemistry 19, 1543–1549 (1980).
Sullivan, D. J., Gluzman, I., Russell, D. & Goldberg, D. On the molecular mechanism of chloroquine's antimalarial action. Proc. Natl Acad. Sci. USA 93, 11865–11870 (1996).
Bray, P., Mungthin, M., Ridley, R. & Ward, S. Access to hematin: the basis of chloroquine resistance. Mol. Pharmacol. 54, 170–179 (1998).
Mungthin, M., Bray, P., Ridley, R. & Ward, S. Central role of hemoglobin degradation in mechanisms of action of 4-aminoquinolines, quinoline methanols, and phenanthrene methanols. Antimicrob. Agents Chemother. 42, 2973–2977 (1998).
Sullivan, D. J., Matile, H., Ridley, R. & Goldberg, D. A common mechanism for blockade of heme polymerization by antimalarial quinolines. J. Biol. Chem. 273, 31103–31107 (1998).
Meshnick, S., Taylor, T. & Kamchonwongpaisan, S. Artemisinin and the antimalarial endoperoxides: From herbal remedy to targeted chemotherapy. Microbiol. Rev. 60, 301–315 (1996).
Ridley, R., Dorn, A., Vippagunta, S. & Vennerstrom, J. Haematin (haem) polymerisation and its inhibition by quinoline antimalarials. Ann. Trop. Med. Parasitol. 91, 559–566 (1997).
Sugioka, Y. & Suzuki, M. The chemical basis for the ferriprotoporphyrin IX–chloroquine complex induced lipid peroxidation. Biochim. Biophys. Acta 1074, 19–24 (1991).
Ginsburg, H. & Krugliak, M. Chloroquine—some open questions on its antimalarial mode of action and resistance. Drug Resist. Update 2, 63–70 (1999).
Olliaro, P., Haynes, R., Meunier, B. & Yuthavong, Y. Possible modes of action of the artemisinin-type compounds. Trends Parasitol. 17, 122–126 (2001).
Fidock, D. A. et al. Mutations in the P. falciparum digestive vacuole transmembrane protein PfCRT and evidence for their role in chloroquine resistance. Mol. Cell 6, 861–871 (2000).
Nomura, T. et al. Evidence for different mechanisms of chloroquine resistance in 2 Plasmodium species that cause human malaria. J. Infect. Dis. 183, 1653–1661 (2001).
Duraisingh, M., Roper, C., Walliker, D. & Warhurst, D. Increased sensitivity to the antimalarials mefloquine and artemisinin is conferred by mutations in the pfmdr1 gene of Plasmodium falciparum. Mol. Microbiol. 36, 955–961 (2000).
Reed, M. B., Saliba, K. J., Caruana, S. R., Kirk, K. & Cowman, A. F. Pgh1 modulates sensitivity and resistance to multiple antimalarials in Plasmodium falciparum. Nature 403, 906–909 (2000).
Cowman, A., Galatis, D. & Thompson, J. Selection for mefloquine resistance in Plasmodium falciparum is linked to amplification of the pfmdr 1 gene and cross-resistance to halofantrine and quinine. Proc. Natl Acad. Sci. USA 91, 1143–1147 (1994).
Stocks, P. A., Raynes, K. & Ward, S. in Antimalarial Chemotherapy. Mechanisms of Action, Resistance, and New Directions in Drug Discovery (ed. Rosenthal, P.) 235–253 (Humana, Totowa, New Jersey, 2001).
Kurosawa, Y. et al. Hematin polymerization assay as a high-throughput screen for identification of new antimalarial pharmacophores. Antimicrob. Agents Chemother. 44, 2638–2644 (2000).
Martin, S., Oduola, A. & Milhous, W. Reversal of chloroquine resistance in Plasmodium falciparum by verapamil. Science 235, 899–901 (1987).
Bray, P. & Ward, S. A comparison of the phenomenology and genetics of multidrug resistance in cancer cells and quinoline resistance in Plasmodium falciparum. Pharmacol. Ther. 77, 1–28 (1998).
Sowunmi, A. et al. Enhanced efficacy of chloroquine-chlorpheniramine combination in acute uncomplicated falciparum malaria in children. Trans. R. Soc. Trop. Med. Hyg. 91, 63–67 (1997).
Posner, G. Antimalarial peroxides in the qinghaosu (artemisinin) and yinghaosu families. Expert Opin. Ther. Patents 8, 1487–1493 (1998).
Expert Opinion on: Water soluble trioxanes as potent and safe antimalarial agents. Exp. Opin. Ther. Patents 11, 1351–1354 (2001).
Vroman, J., Alvim-Gaston, M. & Avery, M. Current progress in the chemistry, medicinal chemistry and drug design of artemisinin-based antimalarials. Curr. Pharmaceut. Des. 5, 101–108 (1999).
Dong, Y. & Vennerstrom, J. Peroxidic antimalarials. Exp. Opin. Ther. Patents 11, 1753–1760 (2001).
Kohler, S. et al. A plastid of probable green algal origin in Apicomplexan parasites. Science 275, 1485–1489 (1997).
Wilson, R. et al. Complete gene map of the plastid-like DNA of the malaria parasite Plasmodium falciparum. J. Mol. Biol. 261, 155–172 (1996).
Waller, R. et al. Nuclear encoded proteins target the plastid in Toxoplasma gondii and Plasmodium falciparum. Proc. Natl Acad. Sci. USA 95, 12352–12357 (1998).
Surolia, N. & Surolia, A. Triclosan offers protection against blood stages of malaria by inhibiting enoyl-ACP reductase of Plasmodium falciparum. Nature Med. 7, 167–173 (2001).
Jomaa, H. et al. Inhibitors of the nonmevalonate pathway of isoprenoid biosynthesis as antimalarial drugs. Science 285, 1573–1576 (1999).
Ohkanda, J. et al. Peptidomimetic inhibnitors of protein farnesyltransferases show antimalarial activity. Bioorg. Med. Chem. Lett. 11, 761–764 (2001).
Fry, M. & Beesley, J. Mitochondria of mammalian Plasmodium spp. Parasitol. Today 102, 17–26 (1991).
Alzeer, J. et al. Phenyl beta-methoxyacrylates: a new antimalarial pharmacophore. J. Med. Chem. 43, 560–568 (2000).
Hyde, J. Mechanisms of resistance of Plasmodium falciparum to antimalarial drugs. Microb. Infect. (in the press).
Winstanley, P. Chemotherapy for falciparum malaria: the armoury, the problems and the prospects. Parasitol. Today 16, 146–153 (2000).
Kinyanjui, S., Mberu, E., Winstanley, P., Jacobus, D. & Watkins, W. The antimalarial triazine WR99210 and the prodrug PS-15: folate reversal of in vitro activity against Plasmodium falciparum and a non-antifolate mode of action of the prodrug. Am. J. Trop. Med. Hyg. 60, 943–947 (1999).
Olliaro, P. & Yuthavong, Y. An overview of chemotherapeutic targets for antimalarial drug discovery. Pharmacol. Ther. 81, 91–110 (1999).
Dunn, C. et al. The structure of lactate dehydrogenase from Plasmodium falciparum reveals a new target for anti-malarial design. Nature Struct. Biol. 3, 912–915 (1996).
Woodrow, C., Penny, J. & Krishna, S. Intraerythrocytic Plasmodium falciparum expresses a high affinity facilitative hexose transporter. J. Biol. Chem. 274, 7272–7277 (1999).
Kirk, K. Membrane transport in the malaria-infected erythrocyte. Physiol. Rev. 81, 495–537 (2001).
Desai, S., Bezrukov, S. & Zimmerberg, J. A voltage-dependent channel involved in nutrient uptake by red blood cells infected with the malaria parasite. Nature 406, 1001–1005 (2000).
Blackman, M. Proteases involved in erythrocyte invasion by the malaria parasite: function and potential as drug targets. Curr. Drug Targets 1, 59–83 (2000).
Vial, H. et al. Transport of phospholipid synthesis precursors and lipid trafficking into malaria-infected erythrocytes. Novartis Found. Symp. 226, 82–88 (1999).
Kappes, B., Doerig, C. & Graeser, R. An overview of Plasmodium protein kinases. Parasitol. Today 15, 449–454 (1999).
Horrocks, P., Bowman, S., Kyes, S., Waters, A. & Craig, A. Entering the post-genomic era of malaria research. Bull. World Health Organ. 78, 1424–1437 (2000).
Buysse, J. The role of genomics in antibacterial target discovery. Curr. Med. Chem. 8, 1763–1776 (2001).
Teichman, S., Chothia, C. & Gerstein, M. Advances in structural genomics. Curr. Opin. Struct. Biol. 9, 390–399 (1999).
Wu, Y., Kirkman, L. & Wellems, T. Transformation of Plasmodium falciparum malaria parasites by homologous integration of plasmids that confer resistance to pyrimethamine. Proc. Natl Acad. Sci. USA 93, 1130–1134 (1996).
Waters, A. et al. Transfection of malaria parasites. Methods 13, 134–147 (1997).
Ridley, R. Antimalarial drug discovery and development — an industrial perspective. Exp. Parasitol. 87, 293–304 (1997).
Venkatesh, S. & Lipper, R. Role of the development scientist in compound lead selection and optimization. J. Pharm. Sci. 89, 145–154 (2000).
Lipinski, C., Lombardo, F., Dominy, B. & Feeney, P. Experimental and computational approaches to estimate solubility in drug discovery and development settings. Adv. Drug Deliv. Rev. 23, 3–25 (1997).
Alnwick, D. Roll back malaria—what are the prospects? Bull. World Health Organ. 78, 1377 (2000).
Sachs, J. A new global commitment to disease control in Africa. Nature Med. 7, 521–523 (2001).
Acknowledgements
I thank W. Jacobs and D. Fidock for allowing me to describe their concept for transfecting genes from human Plasmodium species into rodent Plasmodium species; T. Sukwa for discussions; and D. Fidock for reading the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ridley, R. Medical need, scientific opportunity and the drive for antimalarial drugs. Nature 415, 686–693 (2002). https://doi.org/10.1038/415686a
Issue Date:
DOI: https://doi.org/10.1038/415686a
This article is cited by
-
Preparation, characterization, and evaluation of eosin B–loaded nano-liposomes for growth inhibition of Plasmodium falciparum
Parasitology Research (2022)
-
Histone acetyltransferase PfGCN5 regulates stress responsive and artemisinin resistance related genes in Plasmodium falciparum
Scientific Reports (2021)
-
Plant-Based Synthesis of Silver Nanoparticles Using Aqueous Leaf Extract of Salvia officinalis: Characterization and its Antiplasmodial Activity
Journal of Cluster Science (2021)
-
Diagnosing the drug resistance signature in Plasmodium falciparum: a review from contemporary methods to novel approaches
Journal of Parasitic Diseases (2021)
-
Hantzsch-like synthesis of bis(sulfanediyl)bis(tetrahydropyrimido[4,5-b]quinoline-4,6-diones) linked to arene or heteroarene cores utilizing bis(sulfanediyl)bis(6-aminopyrimidin-4-ones) as precursors
Monatshefte für Chemie - Chemical Monthly (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.