Abstract
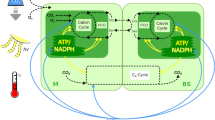
An important adaptation to CO2-limited photosynthesis in cyanobacteria, algae and some plants was development of CO2-concentrating mechanisms (CCM)1. Evolution of a CCM occurred many times in flowering plants, beginning at least 15–20 million years ago, in response to atmospheric CO2 reduction, climate change, geological trends, and evolutionary diversification of species2. In plants, this is achieved through a biochemical inorganic carbon pump called C4 photosynthesis, discovered 35 years ago3. C4 photosynthesis is advantageous when limitations on carbon acquisition are imposed by high temperature, drought and saline conditions. It has been thought that a specialized leaf anatomy, composed of two, distinctive photosynthetic cell types (Kranz anatomy), is required for C4 photosynthesis4. We provide evidence that C4 photosynthesis can function within a single photosynthetic cell in terrestrial plants. Borszczowia aralocaspica (Chenopodiaceae) has the photosynthetic features of C4 plants, yet lacks Kranz anatomy. This species accomplishes C4 photosynthesis through spatial compartmentation of photosynthetic enzymes, and by separation of two types of chloroplasts and other organelles in distinct positions within the chlorenchyma cell cytoplasm.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Badger, M. R. & Spalding, M H. in Photosynthesis: Physiology and Metabolism (eds Leegood, R. C., Sharkey, T. D. & von Caemmerer, S.) 369–397 (Kluwer Academic, Netherlands, 2000).
Sage, R. F. Environmental and evolutionary preconditions for the origin and diversification of the C4 photosynthetic syndrome. Plant Biol. 3, 202–213 (2001).
Hatch, M. D. in Photosynthesis and Photorespiration (eds Hatch, M. D., Osmond, C. B. & Slatyer, R. O.) 139–152 (Wiley-Interscience, New York, 1971).
Sage, R. F. & Monson, R. K. C4 Plant Biology (Academic, San Diego, 1999).
Edwards, G. E. & Walker, D. A. C3,C4: Mechanisms, and Cellular and Environmental Regulation, of Photosynthesis (Blackwell Scientific, Oxford, 1983).
Edwards, G. E., Furbank, R. T., Hatch, M. D. & Osmond, C. B. What does it take to be C4? Lessons from the evolution of C4 photosynthesis. Plant Physiol. 125, 46–49 (2001).
Winter, K. & Smith, J. A. C. Crassulacean Acid Metabolism (Springer, New York, 1996).
Freitag, H. & Stichler, W. A remarkable new leaf type with unusual photosynthetic tissue in a central asiatic genus of Chenopodiaceae. Plant Biol. 2, 154–160 (2000).
Carolin, R. C., Jacobs, S. W. L. & Vesk, M. The structure of the cells of the mesophyll and parenchymatous bundle sheath of the Gramineae. Bot. J. Linn. Soc. 66, 259–275 (1973).
Voznesenskaya, E. V., Franceschi, V. R., Pyankov, V. I. & Edwards, G. E. Anatomy, chloroplast structure and compartmentation of enzymes relative to photosynthetic mechanisms in leaves and cotyledons of species in the tribe Salsoleae (Chenopodiaceae). J. Exp. Bot. 50, 1779–1795 (1999).
Voznesenskaya, E. V. & Gamaley, Y. V. The ultrastructural characteristics of leaf types with Kranz-anatomy. Bot. Zh. 71, 1291–1307 (1986) (in Russian).
Pyankov, V. I. et al. Occurrence of C3 and C4 photosynthesis in cotyledons and leaves of Salsola species (Chenopodiaceae). Photosynth. Res. 63, 69–84 (2000).
Kanai, R. & Edwards, G. in C4 Plant Biology (eds Sage, R. F. & Monson, R. K.) 49–87 (Physiological Ecology series, Academic, San Diego, 1999).
Pyankov, V. I. et al. Features of photosynthesis in Haloxylon species of Chenopodiaceae that are dominant plants in Central Asian deserts. Plant Cell Physiol. 40, 125–134 (1999).
Farquhar, G. D. On the nature of carbon isotope discrimination in C4 species. Aust. J. Plant Physiol. 10, 205–226 (1983).
Laing, W. A., Ogren, W. L. & Hageman, R. H. Regulation of soybean net photosynthetic CO2 fixation by the interaction of CO2, O2, and ribulose 1,5-diphosphate carboxylase. Plant Physiol. 54, 678–685 (1974).
Maroco, J. P., Ku, M. S. B. & Edwards, G. E. Utilization of O2 in the metabolic optimization of C4 photosynthesis. Plant Cell Environ. 23, 115–121 (2000).
Cerling, T. E., Quade, J. & Wang, Y. Expansion and emergence of C4 plants. Nature 371, 112 (1994).
Pagani, M., Freeman, K. H. & Arthur, M. A. Late Miocene atmospheric CO2 concentrations and the expansion of C4 grasses. Science 285, 876–878 (1999).
Sheehy, J. E., Mitchell, P. L. & Hardy, B. Redesigning Rice Photosynthesis to Increase Yield (IRRI and Elsevier Science, Makati City, Philippines, 2000).
Matsuoka, M., Furbank, R. T., Fukayama, H. & Miyao, M. Molecular engineering of C4 photosynthesis. Annu. Rev. Plant Physiol. Plant Mol. Biol. 52, 297–314 (2001).
Maurino, V. G., Drincovich, M. F. & Andreo, C. S. NADP-malic enzyme isoforms in maize leaves. Biochem. Mol. Biol. Int. 38, 239–250 (1996).
Long, J. J., Wang, J.-L. & Berry, J. O. Cloning and analysis of the C4 photosynthetic NAD-dependent malic enzyme of amaranth mitochondria. J. Biol. Chem. 269, 2827–2833 (1994).
Voznesenskaya, E. V. et al. Salsola arbusculiformis, a C3–C4 intermediate in Salsoleae (Chenopodiaceae). Ann. Bot. 88, 337–348 (2001).
Akhani, H., Trimborn, P. & Ziegler, H. Photosynthetic pathways in Chenopodiaceae from Africa, Asia and Europe with their ecological, phytogeographical and taxonomical importance. Plant Syst. Evol. 206, 187–221 (1997).
Dai, Z., Ku, M. S. B. & Edwards, G. E. Oxygen sensitivity of photosynthesis in C3, C4, and C3–C4 intermediate species of Flaveria. Planta 198, 563–571 (1996).
Acknowledgements
We thank the Electron Microscope Center of Washington State University for use of facilities and staff assistance. We thank N. Ogar and H. Akhani for supply of viable seeds, and S. Huber, C. A. Ryan and T. Okita for comments on the manuscript. This work was supported by the National Science Foundation (G.E.E.) and the DFG (H.F.).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Voznesenskaya, E., Franceschi, V., Kiirats, O. et al. Kranz anatomy is not essential for terrestrial C4 plant photosynthesis. Nature 414, 543–546 (2001). https://doi.org/10.1038/35107073
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/35107073
This article is cited by
-
Improvement of photosynthesis in changing environment: approaches, achievements and prospects
Plant Biotechnology Reports (2024)
-
Morphological and antioxidant responses of Nopalea cochenillifera cv. Maya (edible Opuntia sp. “Kasugai Saboten”) to chilling acclimatization
Journal of Plant Research (2023)
-
Photosynthetic responses of oat to leaf blight disease caused by Pantoea agglomerans
Journal of Plant Pathology (2022)
-
Role of C4 photosynthetic enzyme isoforms in C3 plants and their potential applications in improving agronomic traits in crops
Photosynthesis Research (2022)
-
Molecular mechanism underlying the effect of maleic hydrazide treatment on starch accumulation in S. polyrrhiza 7498 fronds
Biotechnology for Biofuels (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.