Abstract
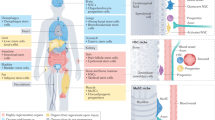
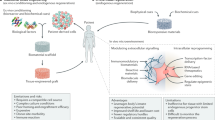
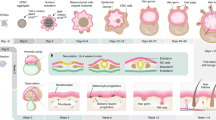
The concept of producing 'spare parts' of the body for replacement of damaged or lost organs lies at the core of the varied biotechnological practices referred to generally as tissue engineering. Use of postnatal stem cells has the potential to significantly alter the perspective of tissue engineering. Successful long-term restoration of continuously self-renewing tissues such as skin, for example, depends on the use of extensively self-renewing stem cells. The identification and isolation of stem cells from a number of tissues provides appropriate targets for prospective gene therapies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Stock, U. A. & Vacanti, J. P. Tissue engineering: current state and prospects. Annu. Rev. Med. 52, 443–451 (2001).
Lagasse, E., Shizuru, J. A., Uchida, N., Tsukamoto, A. & Weissman, I. L. Toward regenerative medicine. Immunity 14, 425–436 (2001).
Barrandon, Y. & Green, H. Three clonal types of keratinocyte with different capacities for multiplication. Proc. Natl Acad. Sci. USA 84, 2302–2306 (1987).
Pellegrini, G. et al. p63 identifies keratinocyte stem cells. Proc. Natl Acad. Sci. USA 98, 3156–3161 (2001).
Ruszczak, Z. & Schwartz, R. A. Modern aspects of wound healing: an update. Dermatol. Surg. 26, 219–229 (2000).
Pellegrini, G. et al. The control of epidermal stem cells (holoclones) in the treatment of massive full-thickness burns with autologous keratinocytes cultured on fibrin. Transplantation 68, 868–879 (1999).
Pellegrini, G. et al. Long-term restoration of damaged corneal surfaces with autologous cultivated corneal epithelium. Lancet 349, 990–993 (1997).
Tsai, R. J., Li, L. M. & Chen, J. K. Reconstruction of damaged corneas by transplantation of autologous limbal epithelial cells. N. Engl. J. Med. 343, 86–93 (2000).
Friedenstein, A. J., Piatetzky, S. II & Petrakova, K. V. Osteogenesis in transplants of bone marrow cells. J. Embryol. Exp. Morphol. 16, 381–390 (1966).
Bianco, P. & Gehron Robey, P. Marrow stromal stem cells. J. Clin. Invest. 105, 1663–1668 (2000).
Bianco, P., Riminucci, M., Gronthos, S. & Robey, P. G. Bone marrow stromal stem cells: nature, biology, and potential applications. Stem Cells 19, 180–192 (2001).
Langstaff, S. et al. Resorbable bioceramics based on stabilized calcium phosphates. Part I: rational design, sample preparation and material characterization. Biomaterials 20, 1727–1741. (1999).
Hutmacher, D. W. Scaffolds in tissue engineering bone and cartilage. Biomaterials 21, 2529–2543 (2000).
Bruder, S. P., Kraus, K. H., Goldberg, V. M. & Kadiyala, S. The effect of implants loaded with autologous mesenchymal stem cells on the healing of canine segmental bone defects. J. Bone Joint Surg. Am. 80, 985–996 (1998).
Krebsbach, P. H., Mankani, M. H., Satomura, K., Kuznetsov, S. A. & Robey, P. G. Repair of craniotomy defects using bone marrow stromal cells. Transplantation 66, 1272–1278 (1998).
Petite, H. et al. Tissue-engineered bone regeneration. Nature Biotechnol. 18, 959–963 (2000).
Kon, E. et al. Autologous bone marrow stromal cells loaded onto porous hydroxyapatite ceramic accelerate bone repair in critical-size defects of sheep long bones. J. Biomed. Mater. Res. 49, 328–337 (2000).
Quarto, R. et al. Repair of large bone defects with the use of autologous bone marrow stromal cells. N. Engl. J. Med. 344, 385–386 (2001).
Mankani, M. H. et al. Pedicled bone flap formation using transplanted bone marrow stromal cells. Arch. Surg. 136, 263–270 (2001).
Hou, Z. et al. Osteoblast-specific gene expression after transplantation of marrow cells: implications for skeletal gene therapy. Proc. Natl Acad. Sci. USA 96, 7294–7299 (1999).
Horwitz, E. M. et al. Transplantability and therapeutic effects of bone marrow-derived mesenchymal cells in children with osteogenesis imperfecta. Nature Med. 5, 309–313 (1999).
Liechty, K. W. et al. Human mesenchymal stem cells engraft and demonstrate site-specific differentiation after in utero transplantation in sheep. Nature Med. 6, 1282–1286 (2000).
Kocher, A. A. et al. Neovascularization of ischemic myocardium by human bone-marrow-derived angioblasts prevents cardiomyocyte apoptosis, reduces remodeling and improves cardiac function. Nature Med. 7, 430–436 (2001).
Ferrari, G. et al. Muscle regeneration by bone marrow-derived myogenic progenitors. Science 279, 1528–1530 (1998).
Orlic, D. et al. Bone marrow cells regenerate infarcted myocardium. Nature 410, 701–705 (2001).
Wakitani, S., Saito, T. & Caplan, A. I. Myogenic cells derived from rat bone marrow mesenchymal stem cells exposed to 5-azacytidine. Muscle Nerve 18, 1417–1426 (1995).
Gussoni, E. et al. Dystrophin expression in the mdx mouse restored by stem cell transplantation. Nature 401, 390–394 (1999).
Makino, S. et al. Cardiomyocytes can be generated from marrow stromal cells in vitro. J. Clin. Invest. 103, 697–705 (1999).
Jackson, K. A. et al. Regeneration of ischemic cardiac muscle and vascular endothelium by adult stem cells. J. Clin. Invest. 107, 1395–1402 (2001).
Azizi, S. A., Stokes, D., Augelli, B. J., DiGirolamo, C. & Prockop, D. J. Engraftment and migration of human bone marrow stromal cells implanted in the brains of albino rats—similarities to astrocyte grafts. Proc. Natl Acad. Sci. USA 95, 3908–3913 (1998).
Lagasse, E. et al. Purified hematopoietic stem cells can differentiate into hepatocytes in vivo. Nature Med. 6, 1229–1234 (2000).
Kuznetsov, S. A. et al. Circulating skeletal stem cells. J. Cell Biol. 153, 1133–1140 (2001).
Bonadio, J. Tissue engineering via local gene delivery: update and future prospects for enhancing the technology. Adv. Drug Deliv. Rev. 44, 185–194 (2000).
Asahara, T., Kalka, C. & Isner, J. M. Stem cell therapy and gene transfer for regeneration. Gene Ther. 7, 451–457 (2000).
Dellambra, E. et al. Toward epidermal stem cell-mediated ex vivo gene therapy of Junctional Epidermolysis Bullosa. Hum. Gene Ther. 11, 2283–2287 (2000).
Lanzov, V. A. Gene targeting for gene therapy: prospects. Mol. Genet. Metab. 68, 276–282 (1999).
Li, J. & Baker, M. D. Mechanisms involved in targeted gene replacement in mammalian cells. Genetics 156, 809–821 (2000).
Jen, K. Y. & Gewirtz, A. M. Suppression of gene expression by targeted disruption of messenger RNA: available options and current strategies. Stem Cells 18, 307–319 (2000).
Johnstone, B. & Yoo, J. U. Autologous mesenchymal progenitor cells in articular cartilage repair. Clin. Orthop. S156–S162 (1999).
Gronthos, S., Mankani, M., Brahim, J., Robey, P. G. & Shi, S. Postnatal human dental pulp stem cells (DPSCs) in vitro and in vivo. Proc. Natl Acad. Sci. USA 97, 13625–13630 (2000).
Ramiya, V. K. et al. Reversal of insulin-dependent diabetes using islets generated in vitro from pancreatic stem cells. Nature Med. 6, 278–282 (2000).
Bjorklund, A. Cell replacement strategies for neurodegenerative disorders. Novartis Found. Symp. 231, 7–15 (2000).
Acknowledgements
The support of Telethon Fondazione Onlus Grant (to P.B.) is gratefully acknowledged.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bianco, P., Robey, P. Stem cells in tissue engineering. Nature 414, 118–121 (2001). https://doi.org/10.1038/35102181
Issue Date:
DOI: https://doi.org/10.1038/35102181
This article is cited by
-
Long-term follow-up and exploration of the mechanism of stromal vascular fraction gel in chronic wounds
Stem Cell Research & Therapy (2023)
-
Beta-naphthoflavone increases the differentiation of osteoblasts and suppresses adipogenesis in human adipose derived stem cells involving STAT3 pathway
Molecular & Cellular Toxicology (2023)
-
Macrophage-derived apoptotic vesicles regulate fate commitment of mesenchymal stem cells via miR155
Stem Cell Research & Therapy (2022)
-
Effects of Biomimetic Materials on Stem Cells from Human Exfoliated Deciduous Teeth
Regenerative Engineering and Translational Medicine (2022)
-
Intracellular pH-responsive iron-catechin nanoparticles with osteogenic/anti-adipogenic and immunomodulatory effects for efficient bone repair
Nano Research (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.