Abstract
Adult honeybees have sleep-like states1,2 and, like human infants3, bees develop their own endogenous circadian rhythms as they mature4,5. But whereas disruption of our sleep cycles and synchronized internal rhythms may adversely affect our physiology and performance3, we show here that honeybees can revert to certain arrhythmic behaviours when necessary. To our knowledge, this chronobiological plasticity is the first example in any animal of a socially mediated reversal in activity rhythms.
Similar content being viewed by others
Main
Circadian rhythms in honeybees are an important component of the social behaviour development process that underlies the colony's division of labour. Larvae must be fed around the clock and are 'nursed' in the hive by young bees (5–15 days old) that work without any overt behavioural rhythms6. At about three weeks of age, however, a bee begins to forage outside the hive for pollen and nectar, an activity that calls for an internal circadian clock for timing visits to flowers and for sun-compass navigation7.
Honeybees show great plasticity during their behavioural development, with their hive-to-field transition being accelerated, delayed, or even reversed in response to changing colony conditions8. We therefore investigated whether this plasticity extends to the bees' behavioural rhythms, focusing on the reversion from foraging to nursing as a particularly compelling challenge to the clock. This reversion occurs in response to a severe shortage of nurse bees and is associated with changes in exocrine, endocrine and neurochemical processes8,9. Do foragers induced to return to nursing also revert to an arrhythmic behavioural state?
We established three unrelated colonies, each composed initially of 2,000–2,500 foragers, their queen and young (sib) brood. Colonies composed only of foragers are known to induce behavioural reversion8 , and indeed the division of labour was reorganized in these colonies: many bees continued to forage, participating in little or no nursing behaviour; some foragers reverted to nursing and stopped foraging completely, or almost completely (Fig. 1a).
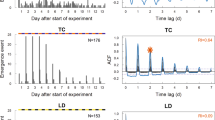
Brood care was observed under dim red light (invisible to bees7) every 3 h for three days. Observations of brood care6: six 10-min visual scans of individually tagged bees in the vicinity of the brood. Foraging observations were made as described6. a, Reorganization of division of labour in reversion colonies: frequency distributions of brood care differed significantly (chi-square test, P < 0.05) for bees never observed foraging (0, left plot), observed foraging once (1, middle), or observed foraging more than once (> 1, right). b, Colonial analysis. Mean (± s.e.) number of brood care events per observation period during the day (white bars) and night (black bars) (n = 6 scans per observation). To test comprehensively for diurnal rhythms, we pooled the data into two half-day categories and compared the amount of brood-care activity between them; this analysis was repeated for eight different half-day combinations. No behavioural rhythms were detected (P > 0.05, chi-square tests with Bonferonni correction). Results were similar for two other colonies (data not shown). Foragers and reverted nurses did not differ in age (29.9 ± 0.2 days, n = 26, and 29.8 ± 0.4, n = 8, respectively; P = 0.76, unpaired t-test). c, Individual analyses. Number of scans with brood care (days double-plotted). Bars at the bottom right show the light–dark regime outside: black, night; white, day). Sixty-six reverted nurses were analysed individually. Y44: example of a bee active around the clock and showing no diurnal rhythm in brood care (P > 0.05; statistical analyses as above); this behaviour was seen in 80.3% of reverted nurses. Y4: example of a bee active around the clock and with a weak diurnal rhythm in brood care (P > 0.05); this behaviour was seen in 15.2% of reverted nurses. W53: one of only three bees (4.5%) showing clear diurnal rhythms (P < 0.05).
As in typical colonies with young nurses6, brood care in our experimental colonies was performed around the clock, with no diurnal oscillations (Fig. 1b). The uninterrupted nursing occurred because individual bees had reverted to arrhythmic activity: analysis of individually tagged reverted nurses (n = 66) revealed that brood care was performed by arrhythmic bees nursing day and night, rather than by rhythmic bees working in shifts (Fig. 1c). We found that reversion also affected the activity–rest cycle: 21 reverted bees (31.8%) cared for the brood in seven or more consecutive observations for 21 hours or longer; foragers, in contrast, rest daily for periods of seven hours or more2.
The underlying cellular and molecular basis of this striking natural behavioural plasticity is unknown. There may be task-dependent changes in a central clock mechanism, uncoupling of nursing activity from the clock, or an effect resulting from nursing behaviour that overrides the clock output. Comparing these possibilities should help to clarify the nature of the cellular and molecular4 bases of chronobiological plasticity.
Reverted nurses were able to rear the brood to maturity in all three colonies. Although we did not test other possible consequences of reversion, our findings may have wider implications, given the conservation of some molecular components of biological clocks10 and of sleep regulation11,12.
References
Kaiser, W. & Steiner-Kaiser, J. Nature 301, 707–709 (1983).
Kaiser, W. J. Comp. Physiol. 163, 565–584 (1988).
Turek, F.W. & Zee, P. C. Regulation of Sleep and Circadian Rhythms (Dekker, New York, 1999).
Toma, D. P., Bloch, G., Moore, D. & Robinson, G.E. Proc. Natl Acad. Sci. USA 97, 6914–6919 (2000).
Stussi, T. & Harmelin, M. L. C. R. Acad. Sci. Hebd. Seances. Acad. Sci. D 262, 2066–2069 (1966).
Moore, D., Angel, J. E., Cheeseman, I. M., Fahrbach, S. E. & Robinson, G. E. Behav. Ecol. Sociobiol. 43, 147–160 (1998).
Von Firsch, K. The Dance Language and Orientation of Bees (Harvard University Press, Cambridge, 1967).
Huang, Z. Y. & Robinson, G. E. Behav. Ecol. Sociobiol. 39, 147–158 (1996).
Schulz, D. J. & Robinson, G. E. J. Comp. Physiol. A 184, 481–488 (1999).
Dunlap, J. C. Cell 96, 271–290 (1999).
Shaw, P. J., Cirelli, C., Greenspan, R. J. & Tononi, G. Science 287, 1834–1837 (2000).
Hendricks, J. C. et al. Neuron 25, 129–138 (2000).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bloch, G., Robinson, G. Reversal of honeybee behavioural rhythms. Nature 410, 1048 (2001). https://doi.org/10.1038/35074183
Issue Date:
DOI: https://doi.org/10.1038/35074183
This article is cited by
-
Targeted worker removal reveals a lack of flexibility in brood transport specialisation with no compensatory gain in efficiency
Scientific Reports (2024)
-
Time-course RNASeq of Camponotus floridanus forager and nurse ant brains indicate links between plasticity in the biological clock and behavioral division of labor
BMC Genomics (2022)
-
Locomotor activity patterns in three spider species suggest relaxed selection on endogenous circadian period and novel features of chronotype
Journal of Comparative Physiology A (2020)
-
The evolutionary ecology of circadian rhythms in infection
Nature Ecology & Evolution (2019)
-
Environmental enrichment influences spatial learning ability in captive-reared intertidal gobies (Bathygobius cocosensis)
Animal Cognition (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.