Abstract
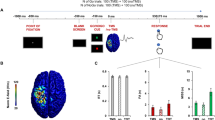
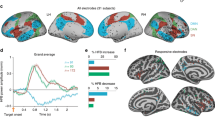
By using the (14C)2-deoxyglucose method1, inhibition has been shown to be a metabolically active process at the level of the synapse2,3. This is supported by recent results from magnetic resonance spectroscopy that related the changes in neuroenergetics occurring with functional activation to neurotransmitter cycling4. However, inhibitory synapses are less numerous and strategically better located than excitatory synapses, indicating that inhibition may be more efficient, and therefore less energy-consuming, than excitation. Here we test this hypothesis using event-related functional magnetic resonance imaging in volunteers whose motor cortex was inhibited during the no-go condition of a go/no-go task, as demonstrated by transcranial magnetic stimulation. Unlike excitation, inhibition evoked no measurable change in the blood-oxygenation-level-dependent signal in the motor cortex, indicating that inhibition is less metabolically demanding. Therefore, the ‘activation’ seen in functional imaging studies probably results from excitation rather than inhibition.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Sokoloff, L. et al. The [14C]deoxyglucose method for the measurement of local cerebral glucose utilization: theory, procedure, and normal values in the conscious and anesthetized albino rat. J. Neurochem. 28, 897–916 ( 1977).
Ackermann, R. F., Finch, D. M., Babb, T. L. & Engel, J. Jr Increased glucose metabolism during long-duration recurrent inhibition of hippocampal pyramidal cells. J. Neurosci. 4, 251–264 (1984).
Nudo, R. J. & Masterton, R. B. Stimulation-induced [14C]2-deoxyglucose labeling of synaptic activity in the central auditory system. J. Comp. Neurol. 245, 553– 565 (1986).
Rothman, D. L. et al. In vivo nuclear magnetic resonance spectroscopy studies of the relationship between the glutamate–glutamine neurotransmitter cycle and functional neuroenergetics. Phil. Trans. R. Soc. Lond. B 354, 1165–1177 (1999).
Fairen, J.D. & Regodor, J. in Cerebral Cortex (eds Peters, A. & Jones, E.) 201–253 (Plenum, New York, 1984).
DeFelipe, J. & Farinas, I. The pyramidal neuron of the cerebral cortex: morphological and chemical characteristics of the synaptic inputs. Prog. Neurobiol. 39, 563– 607 (1992).
Beaulieu, C. & Colonnier, M. A laminar analysis of the number of round-asymmetrical and flat- symmetrical synapses on spines, dendritic trunks, and cell bodies in area 17 of the cat. J. Comp. Neurol. 231, 180–189 ( 1985).
Peters, A., Oalay, S. & Webster, H. The Fine Structure of the Nervous System. Neurons and their Supporting Cells (Oxford Univ. Press, New York, 1991).
Koos, T. & Tepper, J. M. Inhibitory control of neostriatal projection neurons by GABAergic interneurons. Nature Neurosci. 2, 467–472 ( 1999).
Buckner, R. L. et al. Detection of cortical activation during averaged single trials of a cognitive task using functional magnetic resonance imaging. Proc. Natl Acad. Sci. USA 93, 14878– 14883 (1996).
Leocani, L., Cohen, L., Wassermann, E., Ikome, K. & Hallett, M. Human corticospinal excitability evaluated with transcranial magnetic stimulation during different reaction time paradigms. Brain 123, 1161– 1173 (2000).
Rosler, K. M., Etter, C., Truffert, A., Hess, C. W. & Magistris, M. R. Rapid cortical motor output map changes assessed by the triple stimulation technique. NeuroReport 10 , 579–583 (1999).
Kujirai, T. et al. Corticocortical inhibition in human motor cortex. J. Physiol. (Lond.) 471, 501–519 (1993).
Ziemann, U., Rothwell, J. C. & Ridding, M. C. Interaction between intracortical inhibition and facilitation in human motor cortex. J. Physiol. (Lond.) 496, 873–881 (1996).
van Gelderen, P. et al. Three-dimensional functional magnetic resonance imaging of human brain on a clinical 1.5-T scanner. Proc. Natl Acad. Sci. USA 92, 6906–6910 ( 1995).
Deiber, M. P. et al. Cortical areas and the selection of movement: a study with positron emission tomography. Exp. Brain Res. 84, 393–402 (1991).
Shima, K., Mushiake, H., Saito, N. & Tanji, J. Role for cells in the presupplementary motor area in updating motor plans. Proc. Natl Acad. Sci. USA 93, 8694–8698 (1996).
Picard, N. & Strick, P. L. Motor areas of the medial wall: a review of their location and functional activation. Cereb. Cortex 6, 342–353 ( 1996).
Humberstone, M. et al. Functional magnetic resonance imaging of single motor events reveals human presupplementary motor area. Ann. Neurol. 42, 632–637 (1997).
Frackowiak, R. S., Lenzi, G. L., Jones, T. & Heather, J. D. Quantitative measurement of regional cerebral blood flow and oxygen metabolism in man using 15O and positron emission tomography: theory, procedure, and normal values. J. Comput. Assist. Tomogr. 4, 727 –736 (1980).
Raichle, M. E. Measurement of local cerebral blood flow and metabolism in man with positron emission tomography. Federation Proceedings 40, 2331–2334 (1981).
Fox, P. T. & Raichle, M. E. Focal physiological uncoupling of cerebral blood flow and oxidative metabolism during somatosensory stimulation in human subjects. Proc. Natl Acad. Sci. USA 83, 1140–1144 (1986).
Ogawa, S. et al. Intrinsic signal changes accompanying sensory stimulation: functional brain mapping with magnetic resonance imaging. Proc. Natl Acad. Sci. USA 89, 5951–5955 ( 1992).
Silva, A. C., Lee, S. P., Yang, G., Iadecola, C. & Kim, S. G. Simultaneous blood oxygenation level-dependent and cerebral blood flow functional magnetic resonance imaging during forepaw stimulation in the rat. J. Cereb. Blood Flow Metab. 19, 871–879 (1999).
Rees, G. et al. Characterizing the relationship between BOLD contrast and regional cerebral blood flow measurements by varying the stimulus presentation rate. Neuroimage 6, 270–278 (1997).
Raichle, M. E. Behind the scenes of functional brain imaging: a historical and physiological perspective. Proc. Natl Acad. Sci. USA 95, 765–772 (1998).
Weeks, R. A., Gerloff, C., Dalakas, M. & Hallett, M. PET study of visually and non-visually guided finger movements in patients with severe pan-sensory neuropathies and healthy controls. Exp. Brain Res. 128, 291–302 (1999).
Mima, T. et al. Brain structures related to active and passive finger movements in man. Brain 122, 1989– 1997 (1999).
Hallett, M., Shahani, B. T. & Young, R. R. EMG analysis of stereotyped voluntary movements in man. J. Neurol. Neurosurg. Psychiatry 38, 1154–1162 (1975).
Thavenaz, P., Ruttimann, U. E. & Unser, M. Iterative multi-scale registration without landmarks. Proc. IEEE Int. Conf. on Image Processing Vol. III 228–231 (IEEE Computer Society Press, Los Alamitos, CA, 1995).
Acknowledgements
We thank D.G. Schoenberg for help with the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Waldvogel, D., van Gelderen, P., Muellbacher, W. et al. The relative metabolic demand of inhibition and excitation. Nature 406, 995–998 (2000). https://doi.org/10.1038/35023171
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/35023171
This article is cited by
-
Diversity of neuronal activity is provided by hybrid synapses
Nonlinear Dynamics (2021)
-
PET imaging of metabolic changes after neural stem cells and GABA progenitor cells transplantation in a rat model of temporal lobe epilepsy
European Journal of Nuclear Medicine and Molecular Imaging (2019)
-
Preparation and execution of teeth clenching and foot muscle contraction influence on corticospinal hand-muscle excitability
Scientific Reports (2017)
-
Brief Report: The Go/No-Go Task Online: Inhibitory Control Deficits in Autism in a Large Sample
Journal of Autism and Developmental Disorders (2016)
-
Predictive coding for motion stimuli in human early visual cortex
Brain Structure and Function (2016)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.